Stevanto Group tackling flexible drug containment packages and filling solutions
Outsourcing Pharma
JANUARY 30, 2024
Two new offerings have been unveiled by drug delivery and solutions provider, Stevanto Group S.p.A.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Outsourcing Pharma
JANUARY 30, 2024
Two new offerings have been unveiled by drug delivery and solutions provider, Stevanto Group S.p.A.

Drug Discovery World
OCTOBER 19, 2022
Ashley Rein , Global Market Manager for Topical Drug Delivery at Lubrizol Life Science Health (LLS Health), explains how by using mucoadhesive drug delivery, developers can bring novel drugs to market and maximise the efficacy of pre-existing APIs. . Consider drug delivery systems early on .
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Outsourcing Pharma
JANUARY 30, 2024
Two new offerings have been unveiled by drug delivery and solutions provider, Stevanto Group S.p.A.

Drug Discovery World
AUGUST 4, 2022
It may seem counterintuitive to spend time and money on planning for containment and delivery systems for a drug in the earliest stages of discovery when the chances of that molecule making it to market are still quite low. The increasing complexity of drug products has raised concern among pharmaceutical developers.

Pharmaceutical Technology
FEBRUARY 27, 2023
According to GlobalData, Phase I drugs for Adrenal Insufficiency have a 67% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. GlobalData’s report assesses how Hydrocortisone’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks.
Scienmag
JANUARY 8, 2021
Dill/NIST In a technique known as DNA origami, researchers fold long strands of DNA over and over again to construct a variety of tiny 3D structures, including miniature biosensors and drug-delivery containers.

Drug Discovery World
AUGUST 9, 2023
Darren Mercer, Chief Executive of CSP, said: “We are thrilled to acquire AxeroVision, and in particular the pipeline product AXR-270 which contains a novel selective glucocorticoid receptor agonist formulated in a cutting-edge drug delivery system that overcomes the issues of poor compliance and inefficient delivery of conventional eye drops.”

pharmaphorum
NOVEMBER 17, 2021
SMi Group’s 14 th Annual Conference and Exhibition: Pre-Filled Syringes and Injectable Drug Devices 2022 – 12 th – 13 th January 2022. Sustainability for Drug Devices Focus Day – 14 th January 2022. The Future of Drug Delivery and Combination Product Device Design.

Pharmaceutical Technology
SEPTEMBER 20, 2022
The information contained within the download document is intended for pharmaceutical manufacturers, wholesalers, retailers and distributors, pharmaceutical executives, medical representatives, business development managers, retail salesmen, sales managers, pharmacy executives, and any other individual involved in pharmaceutical marketing.

Pharmaceutical Technology
SEPTEMBER 8, 2022
Pharmaceutical solid dosage forms are the most popular in the pharmaceutical industry and are one of the most used drug delivery methods across patient groups. The download contains detailed information on the providers and their services and solutions, alongside contact details to aid your purchasing or hiring decision.

XTalks
JUNE 14, 2023
percent received approval from the US Food and Drug Administration (FDA) to treat dry eye disease. Attendees will understand best practices for novel drug delivery design and development. Attendees will understand best practices for novel drug delivery design and development. What Is Dry Eye Disease?

FDA Law Blog
MARCH 12, 2024
Koblitz — After years of silence from FDA on whether certain patents could be listed in the Orange Book, some manufacturers of drug and device combination products have had a rude awakening lately.

BioTech 365
AUGUST 17, 2021
the “Company”), a leading global provider of drug containment, drug delivery and diagnostic solutions to the … Continue reading →

BioTech 365
JULY 7, 2021
the “Company”), a leading global provider of drug containment, drug delivery and diagnostic solutions to the … Continue reading →
Drug Discovery World
JULY 31, 2023
Darren Mercer, Chief Executive of CSP, said: “We are thrilled to acquire AxeroVision, and in particular the pipeline product AXR-270 which contains a novel selective glucocorticoid receptor agonist formulated in a cutting-edge drug delivery system that overcomes the issues of poor compliance and inefficient delivery of conventional eye drops. “On
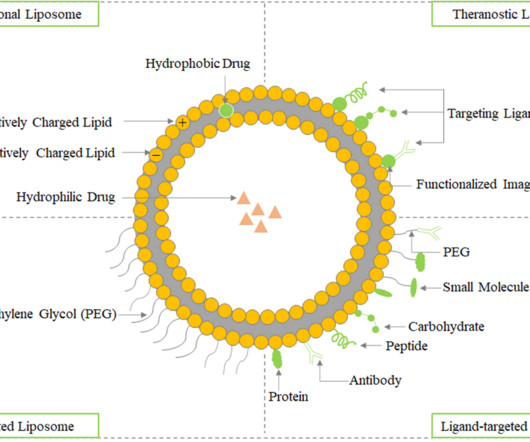
Roots Analysis
AUGUST 8, 2023
The hydrophobic portion contains two fatty acid chains with 10-24 carbon atoms, while the hydrophilic part primarily consists of phosphoric acid linked to a water-soluble molecule. Therapeutic Applications of Liposomes in Drug Delivery In comparison to existing formulations, liposomes offer greater therapeutic efficacy and safety.

XTalks
JANUARY 17, 2023
The US Food and Drug Administration (FDA) granted approval to AstraZeneca’s Airsupra (albuterol/budesonide), formerly known as PT027, for the as-needed treatment or prevention of bronchoconstriction and to reduce the risk of asthma attacks in individuals aged 18 years and older who have asthma.
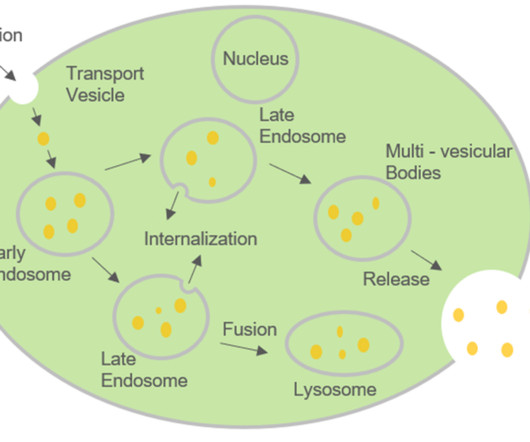
Roots Analysis
MARCH 8, 2023
Over time, various research studies have demonstrated the potential of exosomes ( membrane bound extracellular vesicles) in disease diagnosis, drug delivery and therapeutic applications. Targeted Drug Delivery: Ability to cross blood brain barrier cause lesser systemic effects and low toxicity.

Delveinsight
JANUARY 22, 2021
The launch of a novel drug in the market is not less than a celebration in the pharmaceutical and healthcare market; however, it also brings the real challenge of sustenance in the market. Similar is the picture of the Thrombocytopenia Drugs Market. Drug-Induced Thrombocytopenia, Gian Paolo Visentin, Chao Yan Liu).

The Pharma Data
NOVEMBER 9, 2020
Dr. Löbenberg holds Health Canada licences for research and analytical testing of a wide range of psychedelic compounds under the Controlled Drugs and Substance Act and research and analytical testing licences for cannabis under the Cannabis Act. . DMT (N,N-dimethyltryptamine). Prof. Dr. Löbenberg. ” Prof.

Pharmaceutical Technology
OCTOBER 26, 2022
Nano-based delivery systems are on the rise, as they enable manufacturers to deliver therapeutic agents to specific targeted tissue in a more controlled manner. Data indicates that the global nanopharmaceutical drugs market size reached USD 53.85 Billion in 2021 and is expected to reach USD 102.4 Billion in 2030.

The Pharma Data
DECEMBER 20, 2020
With this acquisition we are expanding our product pipeline to include psychedelic therapeutics, incorporating elements of our IP around drug delivery technology in which we already have prototypes developed, which we believe will propel us towards clinical studies relatively quickly. Ahmad Doroudian, CEO of BetterLife.

The Pharma Data
JANUARY 31, 2021
(CSI ® ) (NASDAQ: CSII) announced today that it has partnered with Chansu Vascular Technologies, LLC (CVT) to develop novel peripheral and coronary everolimus drug-coated balloons (DCBs). Dr. Marco started working on drug delivery cardiovascular devices while in charge of Medical Affairs for Abbott Vascular where, following the Perclose Inc.

The Pharma Data
NOVEMBER 2, 2020
The US Food and Drug Administration has twice designated psilocybin (4-phosphoryloxy-N,N-dimethyltryptamine) a “breakthrough therapy” for the treatment of severe and treatment-resistant depression due to its potential to provide a major improvement over currently available therapeutics for an unmet medical need.

XTalks
JANUARY 18, 2023
Ayesha also talked about the FDA approval of AstraZeneca’s asthma inhaler Airsupra, the first-in-class drug that contains both a beta agonist and corticosteroid. Hear more about the new subvariant and why scientists are keeping a close eye on it.

The Pharma Data
OCTOBER 27, 2020
On April 20, 2020, XPhyto announced a definitive development, technology purchase and licence agreement with 3a-Diagnostics for the development and commercialization of real-time, low-cost and easy-to-use screening tests using 3a’s pathogen specific biosensors and XPhyto’s oral dissolvable drug delivery platform.
Roots Analysis
FEBRUARY 18, 2022
Myeloid cells represent a group of cells containing granulocytes, monocytes, macrophages, mast cells and dendritic cells, which play a key role in innate immunity. In the past decade, more than 1,500 clinical trials have been registered for evaluating drug therapies targeting myeloid cells. Our Social Media Platform. Web: [link].

The Pharma Data
DECEMBER 28, 2020
is an emerging biotechnology company engaged in the development and commercialization of therapeutic pharmaceuticals as well as drug delivery platform technologies. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement. BetterLife Pharma Inc. Source link.

The Pharma Data
OCTOBER 20, 2020
” Mr. Bliss joins TLC following interim roles in cellular therapy, drug delivery and combination therapy where he led efforts to confirm research outcomes and partner the various technologies. This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995.

The Pharma Data
DECEMBER 14, 2020
While the medicalization of psychedelics is evolving and gaining momentum at an extraordinary pace, such therapeutics still face regulatory hurdles in the areas of manufacturing, formulating, and ultimately dispensing or the rescheduling of Schedule I drugs to patients. BetterLife Pharma Inc.

The Pharma Data
JANUARY 18, 2021
According to Health Canada, “the information currently available at the Controlled Substances Directorate, 2-bromo-LSD is NOT CONTROLLED under the Schedules to the Controlled Drugs and Substances Act.”. Forward-looking statements contained in this news release are expressly qualified by this cautionary statement. BetterLife Pharma Inc.

The Pharma Data
JANUARY 3, 2021
The strategic move to partition lung disease programs from TLC will allow TLC to maintain focus on its current pipeline of liposomal injectable formulation programs, such as TLC599 for osteoarthritis pain and TLC590 for postsurgical pain, while instilling to InspirMed funding designated for use in the development of inhalable drugs.

Druggist
NOVEMBER 19, 2020
Formoterol, which belongs to a group of drugs called long-acting beta2-agonist ( LABA ). As we already learned from the above combination inhaler in asthma contains two ingredients, one of which is inhaled corticosteroid and the other LABA. A spacer may offer drug delivery benefits in a wider patient group. Able Spacer®.

The Pharma Data
NOVEMBER 26, 2020
As drug molecules become more complex so do the options to deliver them. There are significant challenges in drug development in this space and we look forward to working closely with pharma and biotech partners to discover how we can add value to their programs, provide patient benefit and competitive product differentiation.”

Roots Analysis
FEBRUARY 16, 2022
In earlier times, container closure integrity testing was not regarded as an essential step in the process of manufacturing. The company developing drug product through such an intense process, use to consider container closure system only for packaging of the product. [1] Container Closure Integrity Testing Services Market.

The Pharma Data
JANUARY 3, 2021
The Company focuses on reducing attrition in clinical trials and on enhancing drug molecules’ formulation performance through its nanoforming services. This press release contains forward-looking statements, including, without limitation, statements regarding Nanoform’s strategy, business plans and focus.
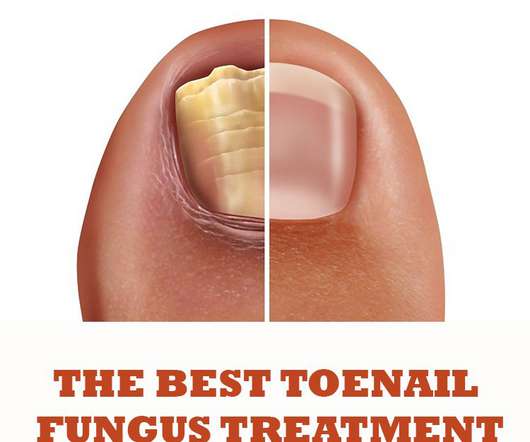
Druggist
OCTOBER 21, 2020
Terbinafine is recommended as the first-line oral antifungal drug. Topical products for toenail fungus treatment are recommended as the first option due to a better safety profile as compared to treatment with oral antifungal drugs. Amorolfine is classified as antimycotics drugs, which kills the fungus. Buy on Amazon Price incl.

The Pharma Data
JANUARY 17, 2021
Clinical validation of transdermal and sublingual drug formulations. Psychedelic API production, drug formulation and clinical integration. Drug Formulations and Delivery. Commercialization of rapid COVID-19 PCR test. Diagnostics. “2020 was a very productive year for XPhyto. .
Drug Discovery World
AUGUST 3, 2022
Emerging as a new treatment option in rare and orphan disease areas, oligonucleotide therapeutics have matured into a drug class with a broad indication spectrum. Oligonucleotides act on the RNA level through different molecular pathways, but there is a major drawback when using them as drugs: their poor bioavailability and cellular uptake.

The Pharma Data
NOVEMBER 4, 2020
is a bioscience accelerator focused on next-generation drug delivery, diagnostic, and phyto-therapeutic investment opportunities: precision transdermal and oral dissolvable drug dosage forms; rapid, low-cost infectious disease and oral health screening tools; and emerging plant-based therapeutics focused on European markets.
The Pharma Data
JUNE 26, 2021
Food and Drug Administration (FDA) has accepted the company’s Biologics License Application (BLA), under Priority Review, for Port Delivery System with ranibizumab (PDS) for the treatment of neovascular or “wet” age-related macular degeneration (nAMD). Roche (SIX: RO, ROG; OTCQX: RHHBY), today announced that the U.S.

The Pharma Data
NOVEMBER 4, 2020
. Juyou will be responsible for the overall clinical development and regulatory filings for Pliaglis with the National Medical Products Administration (the “NMPA”, formerly the China State Food and Drug Administration). million and US$1.8 million. . Juyou Bio-Technology Co.,

The Pharma Data
OCTOBER 27, 2020
DermSafe is listed by Health Canada under its approved products “ Disinfectants and hand sanitizers accepted under COVID-19 interim measure ” and has two Drug Identification Numbers (DIN) for personal use and personal commercial use. Statements have not been evaluated by Health Canada. Visit our website www.ovationscience.com.

The Pharma Data
DECEMBER 2, 2020
The company applies a multi-omics approach, combining genomics, epigenomics, proteomics, and metabolomics to its molecular testing products and to the development of a suite of investigational ingestible devices designed to provide precise diagnostic sampling and drug delivery solutions. Forward Looking Statements.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content