UK MHRA publishes guidance on medicines containing valproate
Pharmaceutical Technology
OCTOBER 12, 2023
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has published updated guidance on valproate-containing medicines.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
OCTOBER 12, 2023
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has published updated guidance on valproate-containing medicines.

Drug Patent Watch
MARCH 6, 2024
GELATIN-Free Medicines, 2024 is part of DrugPatentWatch’s deep library of business intelligence on biopharmaceutical drugs. Buy Gelatin-Free Medicines on Amazon This guide is designed to provide information for healthcare providers to… The post GELATIN-Free Medicines: Which Drugs Contain GELATIN?
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Patent Watch
MARCH 7, 2024
LACTOSE-Free Medicines, 2024 is part of DrugPatentWatch’s deep library of business intelligence on biopharmaceutical drugs. Buy Lactose-Free Medicines from Amazon This guide is designed to provide information for healthcare providers to… The post LACTOSE-Free Medicines: Which Drugs Contain LACTOSE?

Pharmaceutical Technology
APRIL 24, 2023
Traditional medicine refers to the knowledge, skills and practices that indigenous and different cultures use to maintain health, encompassing forms such as herbal medicine, Traditional Chinese Medicine (TCM) and acupuncture.

Drug Discovery World
AUGUST 4, 2022
It may seem counterintuitive to spend time and money on planning for containment and delivery systems for a drug in the earliest stages of discovery when the chances of that molecule making it to market are still quite low. However, starting early can greatly improve the efficiency and speed of the development process. Testing compatibility.

Velocity Clinical Research
SEPTEMBER 28, 2023
Six Velocity Principal Investigators coauthored an article recently published in Nature Medicine. 5-containing bivalent mRNA-1273.222 vaccine with the ancestral Wuhan-Hu-1 mRNA-1273 as booster doses. The article covers the results of a Phase 2/3 trial of the Omicron BA.4/BA.5-containing

Scienmag
MARCH 29, 2021
No FDA regulations in place to alert patients when medication is derived from animals More physicians and pharmacists are advocating for patients to be made aware of animal byproducts contained in common medications, according to new research in the Journal of Osteopathic Medicine.

pharmaphorum
SEPTEMBER 2, 2020
The UK’s medicines regulator has published long-awaited guidance on regulation of medicines and medical devices as the UK approaches the end of its Brexit transition period at the end of the year. The concern is that UK patients will face delays to receiving the latest medicines because of the changes to the regulatory system.

Rethinking Clinical Trials
AUGUST 3, 2023
Bergstrom, PhD Professor, Department of Biology University of Washington Slides Keywords Research, Misinformation, Media, Social Media Key Points Misinformation abounds in medicine and about medicine. Tags #pctGR, @Collaboratory1 The post Grand Rounds July 28, 2023: How Can Researchers Fight Misinformation About Medicine?

The Pharma Data
SEPTEMBER 24, 2020
Some supplements billed as ‘smart drugs’ may contain unapproved drugs in potentially dangerous combinations and doses, according to a new study. The team looked through the National Institute of Health’s Dietary Supplement Label Database and the Natural Medicines Database for products containing unapproved drugs. Conor Kavanagh.

Drug Discovery World
SEPTEMBER 4, 2023
In the third and final part of this series of articles focused on plant-based medicines, DDW’s Megan Thomas evaluates therapeutic breakthroughs using plant-based products in drug discovery. References [link] [link] [link] [link] The post Where is the promise for plant-based medicines?

Pharma in Brief
DECEMBER 23, 2021
Changes to the Patented Medicine Prices Review Board ( PMPRB ) regime, contained in pending amendments to the Patented Medicines Regulations , have been delayed until July 1, 2022. The amendments to the Regulations contain a number of changes affecting the PMPRB’s review of patented medicine prices. The amendments.

Pharma in Brief
APRIL 25, 2021
These amendments permit patents claiming different forms of a medicinal ingredient (e.g., As previously reported , the Proposed FDR Amendments were intended to clarify the regulatory requirements under the Abbreviated New Drug Submission (“ ANDS ”) pathway for generic drug products that contain different forms (e.g.,

Drug Discovery World
FEBRUARY 12, 2024
Why has there been so little progress in medicine? 7 Human biology-based research At Safer Medicines Trust, we believe that using human biology-based research is the best way to develop safe and effective treatments for patients. Rat Trap: The Capture of Medicine by Animal Research – and How to Break Free. BMJ Open Sci.

Scienmag
MARCH 18, 2021
Vaccination alone is unlikely to contain COVID-19 infections in the UK but gradual reopening and high vaccine uptake could minimize future waves of infection COVID-19 vaccines being rolled out in the UK are effective in preventing severe disease, but the extent to which they prevent against infection is still unclear.

FDA Law Blog
OCTOBER 19, 2022
Houck — If, as Albert Einstein supposedly quipped, a cluttered desk is the sign of a cluttered mind, what then does a cluttered medicine cabinet signify? More than a mere sign, a cluttered medicine cabinet can be hazardous if it contains unneeded and expired prescription medication susceptible to theft, misuse and abuse.
Drug Discovery World
FEBRUARY 6, 2023
It contains new long-term survival data of children enrolled in a Phase III trial showing durability of treatment effect through at least four years. “There is an urgent need for a therapy that improves the dismal survival outcome in children with SR-aGVHD” said Dr Silviu Itescu, Chief Executive of Mesoblast.
Pharmaceutical Technology
NOVEMBER 9, 2022
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has approved Pfizer and BioNTech’s bivalent Covid-19 booster vaccine that targets the SARS-CoV-2 virus’ original strain and the Omicron BA.4 Each booster dosage contains 15?g 5 sub-variants.

Pharma in Brief
JULY 7, 2021
Changes to the Patented Medicine Prices Review Board ( PMPRB ) regime, contained in pending amendments to the Patented Medicines Regulations , have been delayed until January 1, 2022. The amendments contain a number of changes affecting the PMPRB’s review of patented medicine prices.

Pharmaceutical Technology
SEPTEMBER 15, 2022
The list includes suppliers of a wide range of pharmaceutical packaging, containers and containment solutions, including plastic, glass and other customised packaging solutions, as well as labelling and printing solutions. Pharmaceutical packaging formats and materials. Trends in pharmaceutical contract packaging.

Pharma in Brief
AUGUST 5, 2020
DESCOVY contains a combination of tenofovir alafenamide ( TAF ) hemifumarate and emtricitabine. Because DESCOVY contains TAF, it benefits from the same data protection term granted to GENVOYA. GENVOYA contains a combination of four medicinal ingredients including TAF. Minister of Health’s Decision.

Drug Discovery World
AUGUST 3, 2023
In the first part of this series of articles focused on plant-based medicines, DDW’s Megan Thomas evaluates the use of medical cannabis in drug discovery. Medical cannabis is often a first-thought when regarding plant-based medicines, but there remains public scepticism, arguably due to the regulations relating to the substance.

Pharma in Brief
DECEMBER 3, 2023
This year marks the 30-year anniversary of the Patented Medicines (Notice of Compliance) Regulations ( Regulations ), introduced in 1993 to prevent patent infringement by linking the regulatory approval of generic or biosimilar drugs with the patent rights of innovators. Evolution of the Regulations Coming into force (1993–1997).

Medical Xpress
DECEMBER 2, 2022
Cough medicines containing the chemical pholcodine should be banned due to the risk of potentially deadly allergic reactions in people under general anaesthetic, the European Union's drug regulator said Friday.

STAT News
JANUARY 9, 2023
Cough medicine tainted with ethylene glycol that killed at least 19 children in Uzbekistan in late December 2022 has once again revealed lax oversight and regulation of pharmaceutical companies based in India. That preventable tragedy, which involves products made by Marion Biotech, based in Noida, India, echoes earlier cases.

Scienmag
AUGUST 6, 2020
First EEG measure of REM sleep allows scientists to distinguish dreaming from wakefulness When we dream, our brains are filled with noisy electrical activity that looks nearly identical to that of the awake brain.

Medical Xpress
MARCH 13, 2023
Financial disruption as a result of pandemic containment policies in the United States adversely influenced children's mental health, according to a new study co-led by Weill Cornell Medicine and Columbia University investigators.
Roots Analysis
MARCH 8, 2023
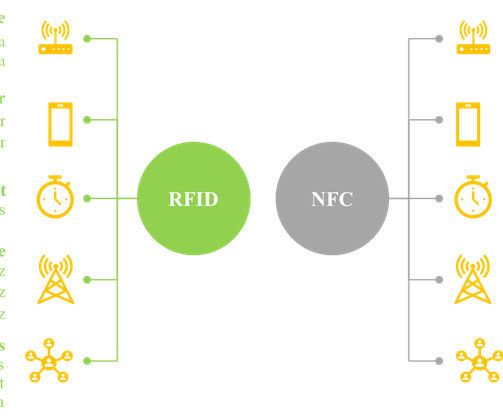
It is worth noting that smart labels contain a transponder code which can be read by sophisticated devices, including radio frequency identification device (RFID) tags and near-field communication (NFC) chips. The post Smart pharmaceutical and healthcare labels: Lets trace medicines from its origin appeared first on Blog.

Drug Discovery World
FEBRUARY 1, 2023
Michael Sassano , CEO at SOMAÍ Pharmaceuticals looks at what’s needed throughout Europe to help increase access to medicinal cannabis and the current state of pharmaceutical research in this area. The use of medicinal cannabis across much of the world is still in its infancy.

Scienmag
MARCH 19, 2021
Genome test for consanguineous couples makes sense For the first time researchers from Hebrew University of Jerusalem, Radboudumc, Maastricht UMC+ and international colleagues have gained insight into the “hidden genetic defects” of the general European population.

Pharmaceutical Technology
SEPTEMBER 5, 2022
Pharmaceutical wholesalers act as intermediaries between pharmaceutical manufacturers and retailers and facilitate the delivery of the right medicines in a timely, efficient, and secure manner. Worldwide sourcing and drop-ship of orphan medicines, and. GDP and GMP advisory. Temperature control facilities and supply solutions.

Pharmaceutical Technology
JUNE 8, 2023
Arexvy contains a combination of recombinant glycoprotein F stabilised in the prefusion conformation (RSVPreF3) and GSK’s AS01E adjuvant. The EC approval follows a positive opinion from the European Medicines Agency’s committee on medicinal products for human use (CHMP) in April 2023.

Scienmag
SEPTEMBER 21, 2020
Credit: Tumisu from Pixabay New research suggests that the impact of natural and vaccine-induced immunity will be key factors in shaping the future trajectory of the global coronavirus pandemic, known as COVID-19.
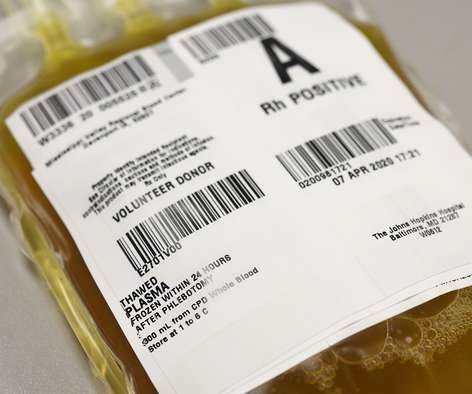
Scienmag
MARCH 30, 2022
The New England Journal of Medicine (NEJM) today published final results of a nationwide multicenter study led by researchers at Johns Hopkins Medicine and the Johns Hopkins Bloomberg School of Public Health that show plasma from patients who have recovered from COVID-19 and whose blood contains antibodies against SARS-CoV-2, the causative virus, is (..)

Scienmag
MARCH 15, 2021
Embargoed News from Annals of Internal Medicine Below please find summaries of new articles that will be published in the next issue of Annals of Internal Medicine. The summaries are not intended to substitute for the full articles as a source of information.

Pharmaceutical Technology
MAY 25, 2023
The results from a trial, published in The New England Journal of Medicine , found the vaccine was associated with a strong immune response and good safety profile. The London School of Hygiene and Tropical Medicine (LSHTM) also highlights the urgent need for a vaccine against the meningococcal X strain.
Pharmaceutical Technology
DECEMBER 27, 2022
Indian pharmaceutical company Hetero has received the World Health Organization Prequalification of Medicines Program (WHO PQ) approval for its Nirmacom (nirmatrelvir). Hetero’s Nirmacom combi-pack will contain nirmatrelvir 150 mg (two tablets) and ritonavir 100mg (one tablet). “We
The Pharma Data
MAY 8, 2021
The study aims to confirm that the proposed biosimilar has equivalent efficacy and comparable safety to the reference medicine* in patients with nAMD 1. Sandoz biosimilars help patients to access advanced biologic medicines more sustainably and affordably. Disclaimer. About Sandoz. source Link.

JAMA Internal Medicine
APRIL 5, 2021
This comparative effectiveness research study examines case-based and population-based interventions and their association with COVID-19 control in Taiwan.
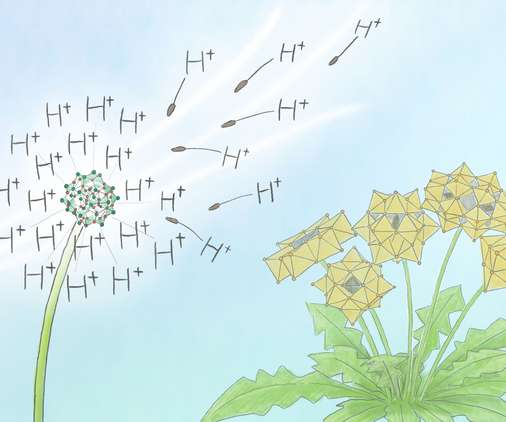
Scienmag
JUNE 17, 2022
Compounds, called polyoxometalates, contain a large metal-oxide cluster carry a negative charge. They are found everywhere, from anti-viral medicines to rechargeable batteries and flash memory devices. Credit: Mindy Takamiya/Kyoto University iCeMS Scientists at Kyoto […].

Pharma in Brief
JULY 7, 2020
The Patented Medicine Prices Review Board ( PMPRB or Board ) has found that a patent can pertain to a medicine based on clinical similarities to the invention—even if the patent does not actually encompass the medicine. Galderma ), markets two adapalene products in Canada: Differin ® , which contains 0.1% Background.
Pharmaceutical Technology
MARCH 31, 2023
The European Medicines Agency (EMA) has recommended HIPRA’s Covid-19 vaccine , Bimervax, as a Covid-19 booster. Available to people ages 16 years and above who have been vaccinated with a Covid-19 mRNA vaccine, EMA’s Human Medicines Committee concluded the vaccine is ready for marketing authorization in the EU, on 30 March.
STAT News
DECEMBER 12, 2022
The new medicine, a pill called Krazati, was cleared to treat patients with advanced non-small cell lung cancer containing a specific type of KRAS alteration called G12C. The mutation is found in approximately 13% of lung tumors. Continue to STAT+ to read the full story…

The Pharma Data
MAY 27, 2022
ViiV Healthcare has today announced that it is committing to license its patents relating to cabotegravir long-acting (LA) for HIV pre-exposure prophylaxis (PrEP) and is entering into negotiation on voluntary licensing terms with the UN-backed Medicines Patent Pool (MPP). Shionogi B.V. joined in October 2012.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content