FDA Action Alert: Sol-Gel, Eton and Pacira
BioSpace
AUGUST 1, 2021
The U.S. FDA is starting off August with a mix of PDUFA dates for acne products, seizure medications and a new manufacturing process for a non-opioid pain killer. Here’s a look.

BioSpace
AUGUST 1, 2021
The U.S. FDA is starting off August with a mix of PDUFA dates for acne products, seizure medications and a new manufacturing process for a non-opioid pain killer. Here’s a look.

JAMA Internal Medicine
AUGUST 1, 2021
This randomized clinical trial examines a decision support tool that provides lower back pain information and self-management recommendations that are specific to an individual’s characteristics, symptoms, and symptom progression.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioSpace
AUGUST 1, 2021
The company saw global net revenue of $13.9 billion during the second quarter, an increase of 33.9%. Sales were primarily driven by its immunology portfolio, which generated $6.12 billion.

JAMA Internal Medicine
AUGUST 1, 2021
This quality improvement study examines the continued citations of a widely publicized retracted article on COVID-19.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
BioSpace
AUGUST 1, 2021
Late-phase trial data show Servier’s TIBSOVO, in conjunction with chemotherapy azacitidine, prolonged event-free survival in adult patients with previously untreated IDH1-mutated acute myeloid leukemia.

ECRG Media's Clinical Research Podcast
AUGUST 1, 2021
Difference Between a CTA and a CRC Don't forget to Subscribe for new content! Subscribe: [link] arch Call or Text: 910-502-3732 Email: eliteclinicalgroup@gmail.com Podcast: [link] Steemit: [link] Advertise: eliteclinicalgroup@gmail.com Watch: » Industry News: [link] » All Videos: [link] » Interview Recaps: [link] » Glassdoor Reviews: [link] We do: » Insider Interviews » Resume Reviews » Question and Answer » Discuss all things Clinical Research » News and Product Reviews » Education Elite Clinic
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

ECRG Media's Clinical Research Podcast
AUGUST 1, 2021
Difference Between a CTA and a CRC. Don't forget to Subscribe for new content! Subscribe: [link] arch Call or Text: 910-502-3732 Email: eliteclinicalgroup@gmail.com Podcast: [link] Steemit: [link] Advertise: eliteclinicalgroup@gmail.com Watch: » Industry News: [link] » All Videos: [link] » Interview Recaps: [link] » Glassdoor Reviews: [link] We do: » Insider Interviews » Resume Reviews » Question and Answer » Discuss all things Clinical Research » News and Product Reviews » Education.

BioSpace
AUGUST 1, 2021
With the Alzheimer’s Association International Conference conference last week, there were plenty of clinical trial news and updates. Here’s a look.
The Pharma Data
AUGUST 1, 2021
In a sample of over 400 older adults in Argentina who had recovered from COVID-19, more than 60% displayed some degree of cognitive impairment, a researcher from The University of Texas Health Science Center at San Antonio reported July 29 at the Alzheimer’s Association International Conference. It is not known whether the impairment, such as forgetfulness and language difficulty, will be progressive, said Gabriel de Erausquin, MD, PhD, a neurologist with the health science center’s Glenn Biggs

BioSpace
AUGUST 1, 2021
The three companies will use their proceeds to support the development of their pipelines through additional business development opportunities.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

The Pharma Data
AUGUST 1, 2021
Saphnelo is a first-in-class type I interferon receptor antibody and the only new medicine in over a decade for patients with systemic lupus erythematosus . AstraZeneca’s Saphnelo (anifrolumab-fnia) has been approved in the US for the treatment of adult patients with moderate to severe systemic lupus erythematosus (SLE) who are receiving standard therapy. 1.

BioSpace
AUGUST 1, 2021
Seventeen years after buying global rights, AstraZeneca is finally announcing a win for the lupus community with an FDA approval for Saphnelo.

The Pharma Data
AUGUST 1, 2021
Amgen today announced that its Board of Directors declared a $1.76 per share dividend for the third quarter of 2021. The dividend will be paid on September 8, 2021, to all stockholders of record as of the close of business on August 17, 2021. About Amgen Amgen is committed to unlocking the potential of biology for patients suffering from serious illnesses by discovering, developing, manufacturing and delivering innovative human therapeutics.

BioSpace
AUGUST 1, 2021
The financing will be used to advance the company’s IMVT-1401 candidate across several different autoimmune disorder indications.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

The Pharma Data
AUGUST 1, 2021
– Takeda Intends to Challenge This Outcome through All Available Legal Means – First Quarter FY2021 Reported IRFS-based Financial Results Will Be Updated to Reflect the Impact of the Decision with No Impact on Core and Underlying Financial Results. Takeda Pharmaceutical Company Limited today announced the receipt of a decision by the Irish Tax Appeals Commission on July 30, 2021 (IST) to uphold the Irish Revenue Commissioners’ position related to the treatment of a break fee received

BioSpace
AUGUST 1, 2021
?The deal will see Ipsen and Exicure collaborating to research, develop, and commercialize Spherical Nucleic Acids to treat AS and HD.
Drug Patent Watch
AUGUST 1, 2021
BREO ELLIPTA (fluticasone furoate; vilanterol trifenatate) Glaxo grp ltd Patent: 6,537,983 Expiration: Aug 3, 2021 See More … For more information on how DrugPatentWatch can help with your pharmaceutical business…. The post Drug Patent Expirations for the Week of August 1, 2021 appeared first on DrugPatentWatch - Make Better Decisions.

Clinical Trial Gurus
AUGUST 1, 2021
In a year like 2021 in clinical research, site owners and principal investigators should be positioning themselves to convert opportunities into positive outcomes. In this webinar we share some strategies for being able to successfully execute on this type of a year!

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

BioSpace
AUGUST 1, 2021
It is offering 17.7 million shares with an expected price between $16 and $18 per share. It will trade on the Nasdaq under the ADGI ticker symbol.

BioTech 365
AUGUST 1, 2021
Ipsen and Exicure Enter Into Exclusive Collaboration Targeting Rare Neurodegenerative Disorders Ipsen and Exicure Enter Into Exclusive Collaboration Targeting Rare Neurodegenerative Disorders Ipsen obtains exclusive options to Spherical Nucleic Acids (SNAs™) currently under discovery evaluation for Huntington’s disease and Angelman … Continue reading →
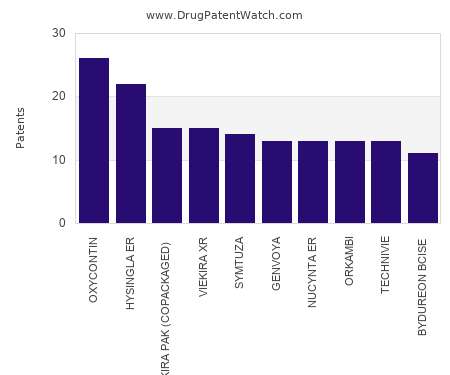
Drug Patent Watch
AUGUST 1, 2021
This chart shows the drugs with the most patents in Slovenia. Patents must be filed in each country (or, in some cases regional patent office) where patent protection is desired.…. The post Which pharmaceutical drugs have the most drug patents in Slovenia? appeared first on DrugPatentWatch - Make Better Decisions.

BioTech 365
AUGUST 1, 2021
Cross Medical Group Introduces QWO® Injections for Cellulite Reduction Cross Medical Group Introduces QWO® Injections for Cellulite Reduction Board-Certified Plastic Surgeon Dr. Kevin J. Cross and the Philadelphia-Based Team Now Offer the Leading-Edge Cellulite Treatment PHILADELPHIA, Aug.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

The Pharma Data
AUGUST 1, 2021
Reports Second-Quarter Diluted EPS of $0.42 on a GAAP Basis; Adjusted Diluted EPS of $3.11 – Delivers Second-Quarter Net Revenues of $13.959 Billion, an Increase of 33.9 Percent on a Reported Basis – Second-Quarter Global Net Revenues from the Immunology Portfolio Were $6.120 Billion, an Increase of 15.1 Percent on a Reported Basis, or 13.8 Percent on an Operational Basis; U.S.

BioTech 365
AUGUST 1, 2021
Press news Biocartis Group NV: Update on Fire Incident at Biocartis Warehouse Press news Biocartis Group NV: Update on Fire Incident at Biocartis Warehouse PRESS RELEASE 2 August 2021, 07:00 CET Update on Fire Incident at Biocartis Warehouse Mechelen, Belgium, … Continue reading →
World of DTC Marketing
AUGUST 1, 2021
SUMMARY: It’s almost impossible for the average person to get accurate and reliable information from the media. Even vaccine booster shots are being debated by HCPs and Pfizer. We need to follow the science. Physiican trust in the FDA is decling as well. The best way to get accurate health information may be to follow noted physicians on social media.

BioTech 365
AUGUST 1, 2021
Addex Appoints Seasoned Pharma Drug Researcher Mikhail Kalinichev as Head of Translational Science Addex Appoints Seasoned Pharma Drug Researcher Mikhail Kalinichev as Head of Translational Science Geneva, Switzerland, August 2, 2021 (GLOBE NEWSWIRE) – Ad Hoc Announcement Pursuant to Art.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

BioTech 365
AUGUST 1, 2021
Addex Therapeutics to Release Half-Year 2021 Financial Results and Host Conference Call on August 5, 2021 Addex Therapeutics to Release Half-Year 2021 Financial Results and Host Conference Call on August 5, 2021 Geneva, Switzerland, August 2, 2021 – Addex Therapeutics Ltd … Continue reading →

BioTech 365
AUGUST 1, 2021
GC Pharma Reports Q2 2021 Results GC Pharma Reports Q2 2021 Results YONGIN, South Korea–(BUSINESS WIRE)–GC Pharma (formerly known as Green Cross Corporation) (KRX: 006280), a South Korean biopharmaceutical company, today announced unaudited consolidated financial results for the three months … Continue reading →

BioTech 365
AUGUST 1, 2021
Leicester’s Hospitals Awards Deenova a Unit Dose Pharmacy Automation 15 Year Contract Leicester’s Hospitals Awards Deenova a Unit Dose Pharmacy Automation 15 Year Contract MILAN–(BUSINESS WIRE)–#Deenova—Deenova reported today it signed its first ever contract in the United Kingdom, and established … Continue reading →

BioTech 365
AUGUST 1, 2021
IGEA Pharma N.V. / Key word(s): Miscellaneous 02-Aug-2021 / 00:47 CET/CEST Release of an ad hoc announcement pursuant to Art. 53 LR The issuer is solely responsible for the content of this announcement.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content