Why the FDA is making a test case of a cancer drug from China
Bio Pharma Dive
FEBRUARY 8, 2022
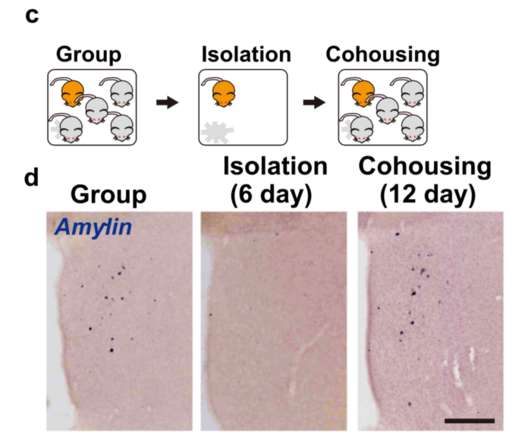
In documents released Tuesday, FDA staff gave a cool review to an immunotherapy developed by Eli Lilly and Innovent Biologics. An advisory meeting this week to review it could have broader implications for cancer drug development in the U.S.



































Let's personalize your content