Intellia cleared to start key CRISPR drug study in UK
Bio Pharma Dive
OCTOBER 19, 2020
The trial would be the first test of a systemically administered gene editing treatment that uses the Nobel Prize-winning technology to alter DNA.

Bio Pharma Dive
OCTOBER 19, 2020
The trial would be the first test of a systemically administered gene editing treatment that uses the Nobel Prize-winning technology to alter DNA.

World of DTC Marketing
OCTOBER 19, 2020
GOOD MORNING: eMarketer is promoting a report on pharma’s projected digital ad spending in the coming year, and from my conversations with pharma insiders, it’s wrong. There will be an investment in new digital platforms but display and search spending, I believe, are going to decline. There is no doubt that more people are turning to the Internet to help sort through the conflicting health information but that does not translate into more online ads.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
OCTOBER 19, 2020
The findings have implications for how pharma teams should think about their DTC and HCP efforts — they are no longer two separate marketing channels.

pharmaphorum
OCTOBER 19, 2020
Regular fliers are used to long queues to clear security – but a rigorous COVID-19 testing regime could be added to pre-flight checks backed by a digital “health passport” to prove passengers are free from the coronavirus. The UK is the latest country to test the scheme that allows passengers to demonstrate their coronavirus status as part of a global framework for COVID-safe air travel.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
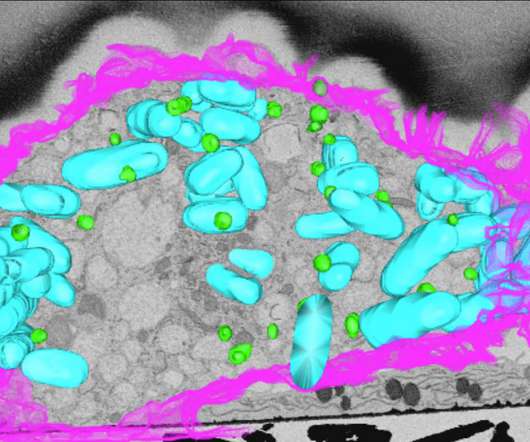
Scienmag
OCTOBER 19, 2020
Droplets of fat inside our cells are helping the body’s own defence system fight back against infection, University of Queensland researchers have discovered. Credit: Institute for Molecular Bioscience, University of Queensland Droplets of fat inside our cells are helping the body’s own defence system fight back against infection, University of Queensland researchers have discovered.

pharmaphorum
OCTOBER 19, 2020
As part of our series of opinion pieces from leaders at Janssen, the company’s Maria Raad looks at how we can embrace tech and data science to overcome increasing pressures on healthcare systems. For the next generation born in the western world, living to be 100 will be the norm. While this seems like a desirable aspiration for our grandchildren, it adds new pressures on our healthcare systems.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

pharmaphorum
OCTOBER 19, 2020
Gilead Sciences’ Kite Pharma unit is closing on approval of its second European approval for a CAR-T for cancer, after the CHMP backed its Tecartus therapy for mantle cell lymphoma. The EMA’s human drugs advisory committee recommended approval of Tecartus (brexucabtagene autoleucel; formerly KTE-X19 ) for relapsed or refractory MCL setting up a formal approval by the regulator in the coming weeks.

Intouch Solutions
OCTOBER 19, 2020
Brands can help patients and physicians by building and strengthening their relationships with NPs and PAs. Engaging them helps the entire care team perform at a higher level. Learn how here! The post HCP > MD: How Brands Can Help NPs and PAs Improve Healthcare appeared first on Intouch Solutions.
Outsourcing Pharma
OCTOBER 19, 2020
The agencyâs Solidarity Therapeutics Trial examined a range of drugs repurposed for COVID-19 treatment, finding nearly no effect on patient mortality.

JAMA Internal Medicine
OCTOBER 19, 2020
In this issue of JAMA Internal Medicine, 3 important articles explore the use of tocilizumab in coronavirus disease 2019 (COVID-19) pneumonia. Tocilizumab is a humanized monoclonal antibody that binds human interleukin 6 (IL-6) receptors. It is used routinely in inflammatory arthritis, giant cell arteritis, and cytokine release syndrome after chimeric antigen receptor T-cell therapy.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
BioSpace
OCTOBER 19, 2020
Under terms of the deal, Connecticut-based Cara Therapeutics will receive $100 million in an upfront payment, as well as an equity investment of $50 million. Additional milestone payments could bring the total of the deal to about $290 million.
BioPharma Reporter
OCTOBER 19, 2020
Edesa Biotech, Inc. has received clearance from the U.S. Food and Drug Administration (FDA) to begin the Phase 2 portion of its Phase 2/3 clinical study of its investigational drug, EB05, for the treatment of hospitalized COVID-19 patients.

XTalks
OCTOBER 19, 2020
PepsiCo snack and beverages, Mtn Dew, Mtn Dew Game Fuel and Doritos are joining forces with Activision’s blockbuster Call of Duty game franchise for the launch of Call of Duty: Black Ops Cold War. The program gives players access to bonus in-game rewards and prizes. With every purchase comes a chance to win special gaming swag and scoring double XP.
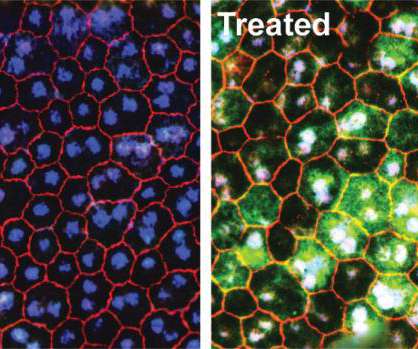
Scienmag
OCTOBER 19, 2020
New generation CRISPR technology lays foundation for therapeutics to treat a wide range of inherited ocular diseases Credit: Nature Biomedical Engineering Irvine, CA – October 19, 2020 – A breakthrough study, led by researchers from the University of California, Irvine, results in the restoration of retinal and visual functions of mice models suffering from inherited […].

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

pharmaphorum
OCTOBER 19, 2020
Novartis’ near-$10 billion takeover of The Medicines Company last year was focused mainly on one asset – cholesterol-lowering drug inclisiran – and the Swiss pharma is now a step closer to getting a return on its investment. Inclisiran – now given the trade name Leqvio – has been recommended for approval by the CHMP as a treatment for hypercholesterolaemia or mixed dyslipidaemia, two common forms of elevated cholesterol linked to increased risk of cardiovascular disease.

Pharma Times
OCTOBER 19, 2020
Following completion of acquisition, site will undergo refurbishment

BioSpace
OCTOBER 19, 2020
INCOG BioPharma Services, a biopharmaceutical manufacturing services startup, announced on Oct. 15 that it will be establishing its first manufacturing facility and global headquarters in Fishers, Indiana.

Scienmag
OCTOBER 19, 2020
The latest report from the Khaled bin Sultan Living Oceans Foundation finds Palau’s reefs had the highest coral cover observed on the Global Reef Expedition–the largest coral reef survey and mapping expedition in history. Credit: ©Keith Ellenbogen/iLCP Scientists at the Khaled bin Sultan Living Oceans Foundation (KSLOF) have released their findings on the state of […].

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

Pharma Times
OCTOBER 19, 2020
Chief executive of trade body warns no-deal could affect medicines supply
BioPharma Reporter
OCTOBER 19, 2020
Moderna is engaging with international regulators to accelerate approval of its COVID-19 vaccine candidate.
BioSpace
OCTOBER 19, 2020
Intellia has received approval to launch their Phase I trial of NTLA-2001 in hereditary ATTR.
BioPharma Reporter
OCTOBER 19, 2020
US company, Dyno Therapeutics, has inked a deal with Roche to develop next-generation AAV gene therapy vectors for central nervous system (CNS) diseases and liver-directed therapies.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

BioSpace
OCTOBER 19, 2020
In a challenge trial, people are given a vaccine then intentionally dosed with the virus or bacteria that causes the disease.

Pharma Times
OCTOBER 19, 2020
Dupixent, Forxiga and Opdivo were also recommended for therapeutic extensions

JAMA Internal Medicine
OCTOBER 19, 2020
This randomized clinical trial evaluates the effect of early tocilizumab administration vs standard therapy in preventing clinical worsening in patients hospitalized with COVID-19.

Outsourcing Pharma
OCTOBER 19, 2020
The CRO and life-sciences tech firm collaborated on a solution capable of meeting challenges tied to starting and conducting global clinical trials.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Scienmag
OCTOBER 19, 2020
Silk so robust potential new genetic material touted Credit: Professor Mark Elgar, the University of Melbourne An international collaboration has provided the first insights into a new type of silk produced by the very unusual Australian basket-web spider, which uses it to build a lobster pot web that protects its eggs and trap prey. The […].

BioSpace
OCTOBER 19, 2020
Shares of Aptinyx skyrocketed more than 75% after the company reported its mid-stage experimental treatment for post-traumatic stress disorder demonstrated clinically meaningful and statistically significant results that will set up a pivotal study next year.

Scienmag
OCTOBER 19, 2020
12-week double-blind control trial in 148 Air Force airmen Credit: Photo by L. Brian Stauffer CHAMPAIGN, Ill. — Researchers studied the effects of a 12-week exercise regimen on 148 active-duty Air Force airmen, half of whom also received a twice-daily nutrient beverage that included protein; the omega-3 fatty acid, DHA; lutein; phospholipids; vitamin D; B […].

JAMA Internal Medicine
OCTOBER 19, 2020
This cohort study assesses mortality rates among patients with COVID-19 admitted to intensive care who were treated with tocilizumab.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content