How Gilead finally spent its money
Bio Pharma Dive
NOVEMBER 24, 2020
This year, the biotech spent about $27 billion trying to become a leader in cancer research. Executives who spoke to BioPharma Dive said there's still work to be done.

Bio Pharma Dive
NOVEMBER 24, 2020
This year, the biotech spent about $27 billion trying to become a leader in cancer research. Executives who spoke to BioPharma Dive said there's still work to be done.

World of DTC Marketing
NOVEMBER 24, 2020
SUMMARY : In a 2019 Pew Research Center poll, more Americans said they consider made-up news a bigger problem than terrorism, illegal immigration, racism, and sexism. And 68% said disinformation greatly impacts people’s confidence in government institutions. No matter the form, inaccurate health information is a danger to public health. While psychological factors leave people unguarded against misinformation, people in the U.S. are particularly vulnerable to health misinformation because the l
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
NOVEMBER 24, 2020
The drug, now known as Oxlumo, is the first approved treatment for patients with a potentially life-threatening condition that can cause kidney failure. Like other Alnylam drugs, it comes at a six-figure list price.

Camargo
NOVEMBER 24, 2020
Most are familiar with, know someone who uses, or take “pumps” themselves to prevent or relieve shortness of breath after an effort or during allergy season. Medicine has been delivered by inhalation for decades to treat human respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), cystic fibrosis (CF), and airway infection.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
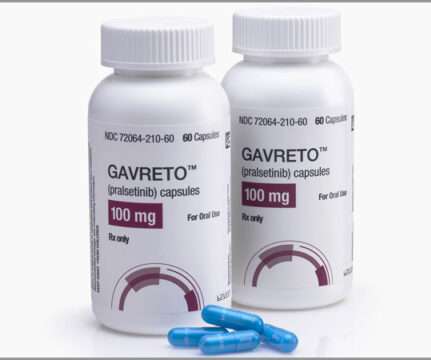
Pharma Mirror
NOVEMBER 24, 2020
Catalent, the leading global provider of advanced delivery technologies, development, and manufacturing solutions for drugs, biologics, cell and gene therapies, and consumer health products, today announced that it had entered into a commercial supply agreement with Blueprint Medicines following FDA approval of GAVRETO™ (pralsetinib). Developed by Blueprint Medicines, GAVRETO is a new therapy indicated for the treatment of adults with metastatic RET fusion-positive non-small cell lung cancer (NS

pharmaphorum
NOVEMBER 24, 2020
A merger involving two US digital health specialists and a blank cheque company has created a telemedicine player, called UpHealth, that is valued at more than $1.3 billion. The three-way deal combines UpHealth – which provides patient care management, telemedicine and digital pharmacy services and gives its name to the new group – with CloudBreak, which provides a video consultation platform for doctors and patients.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

pharmaphorum
NOVEMBER 24, 2020
Pharma sales is set to change forever. To keep afloat in the current climate, teams need to embrace flexibility and remember that traditional content won’t work in new contexts, say experts from Syneos Health. Selling in a socially-distanced world is about more than simply taking conversations online – it requires a wholesale change in how sales teams operate and the kinds of content they use.

XTalks
NOVEMBER 24, 2020
For years, Beyond Meat and Impossible Foods have had a stronghold on the US plant-based meat market, a market that is expected to reach $27.9 billion by 2025. But with growing environmental and safety concerns over meat production and more people opting to eat less meat, new entrants are being welcomed with open arms in the growing market. Enter Modern Meat , a Vancouver, Canada-based meat alternative company that launched around two years ago and recently went public.
pharmaphorum
NOVEMBER 24, 2020
The developer of Russia’s COVID-19 vaccine Sputnik V has pledged to make it available for less than $10 per dose – or $20 per two-dose course – on the same day as revealing new clinical data with the shot. Sputnik V ’s efficacy has remained high at 91.4% according to the latest analysis, which comes from almost 19,000 subjects who were assessed seven days after the second dose of the vaccine, administered 28 days after the first.

BioSpace
NOVEMBER 24, 2020
Manufacturing capacity is often decried as a major bottleneck in administering COVID-19 vaccines to a global population of 7.8 billion. For mRNA vaccines, the bottleneck is the availability of raw materials, not bioreactors.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
BioPharma Reporter
NOVEMBER 24, 2020
Up to 10% of vaccines can be reportedly lost in transit due to breakage or fluctuations in sub-zero temperatures required for preservation, but a manufacturer of data-driven temperature-controlled smart containers says it can cut that rate to just 0.1%.

Scienmag
NOVEMBER 24, 2020
Glass tuning-fork-shaped tubing gives accurate, continuous measurements of drug dissolution Credit: William Grover When you take a time-release drug, you count on it doing what the package says: release the drug slowly into your bloodstream to provide benefits over the specified period of time. When the drug dissolves too slowly or too quickly, the results […].
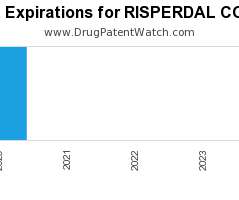
Drug Patent Watch
NOVEMBER 24, 2020
Annual Drug Patent Expirations for RISPERDAL+CONSTA Risperdal Consta is a drug marketed by Janssen Pharms and is included in one NDA. It is available from one supplier. There is one…. The post New patent expiration for Janssen Pharms drug RISPERDAL CONSTA appeared first on DrugPatentWatch - Make Better Decisions.
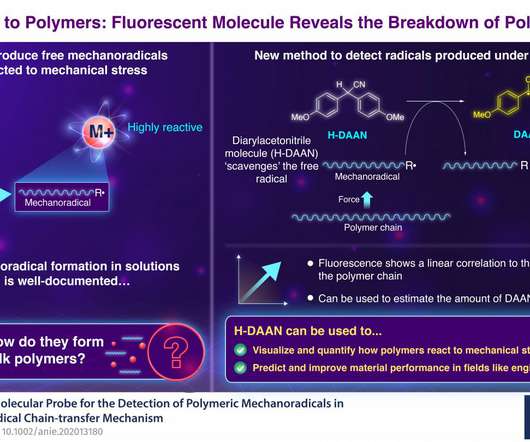
Scienmag
NOVEMBER 24, 2020
Credit: Tokyo Tech Nylon, rubber, silicone, Teflon, PVC – these are all examples of man-made polymers – long chains of repeated molecular units that we call monomers. While polymers also exist in nature (think wool, silk, or even hair), the invention of synthetic polymers, the most famous of which is plastic, revolutionized the industry.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
BioSpace
NOVEMBER 24, 2020
There has been a high level of movement in COVID-19 vaccine news this week, with Pfizer and BioNTech, Moderna, and AstraZeneca and the University of Oxford all having positive clinical trial announcements. Here’s some follow-up news.

Scienmag
NOVEMBER 24, 2020
Credit: Janusz Pawliszyn A common insecticide that is a major hazard for honeybees is now effectively detected in honey thanks to a simple new method. Researchers at the University of Waterloo developed an environmentally friendly, fully automated technique that extracts pyrethroids from the honey. Pyrethroids are one of two main groups of pesticides that contribute […].
BioSpace
NOVEMBER 24, 2020
Convalescent plasma as a treatment for COVID-19 infections has demonstrated mixed results at best in terms of efficacy, and a recent trial in Argentina provides additional fuel to that conversation.

Scienmag
NOVEMBER 24, 2020
Credit: UPMC PITTSBURGH, Nov. 24, 2020 – In today’s issue of the Annals of Surgery, epidemiologists from the University of Pittsburgh published two separate analyses that could help guide clinicians and policymakers in counseling bariatric surgery patients to improve their quality of life for many years to come. A study led by Gretchen White, Ph.D., […].

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

BioPharma Reporter
NOVEMBER 24, 2020
Biopharma company, CureVac, and Wacker Chemie have signed a contract for the manufacturing of CureVacâs mRNA-based COVID-19 vaccine candidate - CVnCoV.
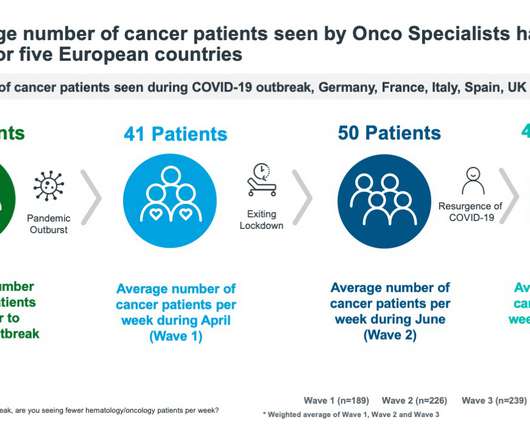
pharmaphorum
NOVEMBER 24, 2020
IQVIA’s Sarah Rickwood explores how pharma can help healthcare systems address treatment backlogs caused by the COVID-19 pandemic. As lockdowns started in the West during March 2020, there was, inevitably, much punditry on exit scenarios – for economies, populations, and healthcare systems. Considerable time was devoted to the discussion of V- , U- and L-shaped recoveries.

BioSpace
NOVEMBER 24, 2020
Find out which biopharma companies are raking in cash this week, as companies from around the globe provide updates on their financing rounds and IPOs.

Scienmag
NOVEMBER 24, 2020
People of color & those with lower incomes or education levels less likely to say they’ll seek vaccination against a disease that’s high risk for older adults Credit: University of Michigan Once enough people receive effective vaccines against the novel coronavirus, experts say, the end of the pandemic may be in sight. But a new […].

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Outsourcing Pharma
NOVEMBER 24, 2020
The two companies will collaborate on a Phase III clinical trial investigating a potential treatment for patients hospitalized with COVID-19.

Scienmag
NOVEMBER 24, 2020
Credit: RIKEN A research team at the RIKEN Center for Biosystems Dynamics Research (BDR) in Japan has succeeded in experimentally evolving the common bacteria Escherichia coli under pressure from a large number of individual antibiotics. In doing so, they were able to identify the mechanisms and constraints underlying evolved drug resistance. Their findings, published in the scientific […].

Pharma Times
NOVEMBER 24, 2020
MenQuadfi approved for vaccination in individuals 12 months of age or older

BioPharma Reporter
NOVEMBER 24, 2020
The European Union has agreed a deal with Moderna for up to 160 million doses of its mRNA COVID-19 vaccine candidate.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Pharma Times
NOVEMBER 24, 2020
Less-frequent, fixed-dose use approved for two cancer indications
BioPharma Reporter
NOVEMBER 24, 2020
GlaxoSmithKline has started dosing patients in a Phase 3 clinical program investigating the safety and efficacy of its Respiratory Syncytial Virus (RSV) candidate vaccine for maternal immunisation.

Scienmag
NOVEMBER 24, 2020
News from the Journal of Marketing Researchers from Boston University and University of Virginia published a new paper in the Journal of Marketing that examines how consumers respond to AI recommenders when focused on the functional and practical aspects of a product (its utilitarian value) versus the experiential and sensory aspects of a product (its […].

BioSpace
NOVEMBER 24, 2020
Days after Purdue Pharma reached a combined $8.4 billion criminal and civil settlement with the U.S. Department of Justice, the OxyContin drugmaker pleaded guilty to three criminal charges to resolve its role in the ongoing opioid crisis in the United States.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content