Moderna gets $1.5B from US for coronavirus vaccine supply
Bio Pharma Dive
AUGUST 11, 2020
The U.S. has already invested heavily in Moderna's vaccine, having previously committed $1 billion to fund clinical trials and manufacturing scale-up.

Bio Pharma Dive
AUGUST 11, 2020
The U.S. has already invested heavily in Moderna's vaccine, having previously committed $1 billion to fund clinical trials and manufacturing scale-up.

Pharma Mirror
AUGUST 11, 2020
CPhI South East Asia – organised by Informa Markets – has been rescheduled to May 18-20, 2021 to give the ASEAN pharma community more time to recover and to mitigate any global disruptions caused by COVID-19. The decision to reschedule the event to 2021 reflects changing attendee needs. By moving to spring next year, we. The post CPhI South East Asia rescheduled for 2021 appeared first on Pharma Mirror Magazine.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Bio Pharma Dive
AUGUST 11, 2020
The shot, developed by the state-backed Gamaleya Research Institute, hasn't yet been tested in the large, placebo-controlled tests scientists emphasize are needed to prove a vaccine is protective.
BioSpace
AUGUST 11, 2020
Although the world is eagerly awaiting news of an effective and safe vaccine against COVID-19, several companies are moving ahead testing therapeutic antibodies against the disease.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Bio Pharma Dive
AUGUST 11, 2020
The unusual startup already has partial rights to multiple drugs developed by MyoKardia and affiliates of BridgeBio, and plans to acquire many more.
Outsourcing Pharma
AUGUST 11, 2020
American Gene Technologies has received the agencyâs approval to launch the first human trial for its lead HIV program, starting in Baltimore and DC.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

BioSpace
AUGUST 11, 2020
Moderna signed a contract worth up to $1.525 billion with the U.S. government to supply 100 million doses of mRNA-1273, the company’s experimental COVID-19 vaccine.

pharmaphorum
AUGUST 11, 2020
In our latest UK Leaders article Pinder Sahota, UK general manager at Novo Nordisk, tells us how he hopes to bring the NHS and pharma together to improve outcomes in diabetes and other long-term conditions. Though Novo Nordisk is perhaps not as much of a household name as other pharma companies in the UK, it has a huge presence in diabetes, with a bigger market cap than the likes of GSK and AstraZeneca – and UK GM Pinder Sahota is keen to shout about the organisation that he describes as a “slee

BioSpace
AUGUST 11, 2020
Celleron Therapeutics signed a licensing agreement with Roche for the worldwide rights to cancer drug emactuzumab, a monoclonal antibody directed against colony-stimulating factor 1 (CSF-1R) expressed on macrophages.
pharmaphorum
AUGUST 11, 2020
Russia has become the first country in the world to approve a coronavirus vaccine, amid scepticism from international experts who have been unable to review or verify clinical data from the jab called ‘Sputnik V’ According to press reports the vaccine has been tested on one of President Vladimir Putin’s daughters. But World Health Organization leaders have said that they want to review the vaccine, called Sputnik V after Russia’s pioneering space probe, as the vaccine took just

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

BioSpace
AUGUST 11, 2020
Olorofim is in a Phase IIb study that focuses on rare and resistant life-threatening invasive fungal infections, such as invasive aspergillosis (including azole-resistant strains), scedosporiosis, lomentosporiosis, fusariosis, scopulariopsosis and coccidioidomycosis.

Scienmag
AUGUST 11, 2020
Demonstrate the production of 3D structure in 620nm pixels with over 50 times higher resolution than that of 8K QLED TV for the first time in the world. Application available on wearable devices and the result published in ACS Nano, the world’s most prest Credit: Korea Electrotechnology Research Institute (KERI) Companies across the world are […].

pharmaphorum
AUGUST 11, 2020
Bayer is to buy UK-based KaNDy Therapeutics in a deal worth more than $800 million, adding the biotech’s menopause drug to its women’s health pipeline. Bayer will pay $425 million up front and potential milestone payments of up to $450 million until launch, followed by potential undisclosed “triple digit million” milestone payments once the drug is on the market.
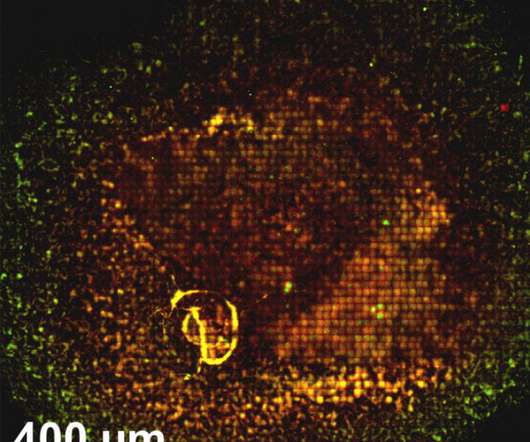
Scienmag
AUGUST 11, 2020
Fundamental research in bacterial physiology Credit: Salim Timo Islam Research by INRS (Institut National de la Recherche Scientifique) Professor Salim Timo Islam and his PhD student Fares Saïdi has recently revealed that multicellular physiology in the social bacterium Myxococcus xanthus–a bacterium that can actively reorganize its community according to the environment in which it is […].

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
BioSpace
AUGUST 11, 2020
A study published in the Journal of the American Medical Association on July 29 now shows that a new blood test may be helpful for detecting Alzheimer’s disease as early as 20 years before the onset of cognitive impairment.
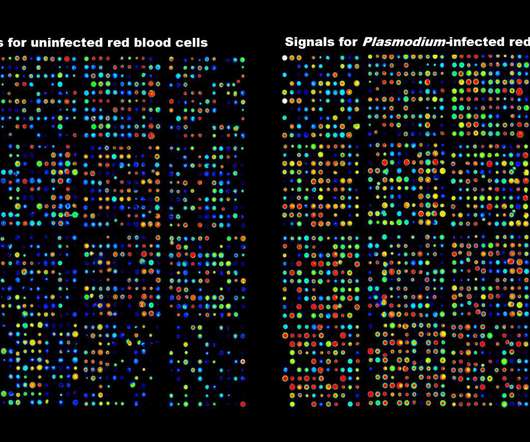
Scienmag
AUGUST 11, 2020
New research into malaria suggests targeting enzymes from the human host, rather than from the pathogen itself, could offer effective treatment for a range of infectious diseases, including COVID-19. Credit: RMIT University The study, conducted by an international team and led by RMIT University’s Professor Christian Doerig, outlines a strategy that could save years of […].
BioPharma Reporter
AUGUST 11, 2020
Regeneron Pharmaceuticals aims to post initial virology and biomarker data from clinical trials of its COVID-19 antibody cocktail next month.
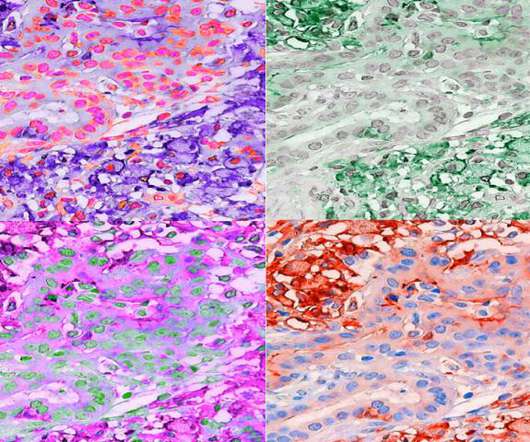
Scienmag
AUGUST 11, 2020
Antibody that blocks TREM2 protein frees immune cells to reject cancer Credit: William Vermi/Martina Molgora Immunotherapy has revolutionized cancer treatment by stimulating the patient’s own immune system to attack cancer cells, yielding remarkably quick and complete remission in some cases. But such drugs work for less than a quarter of patients because tumors are notoriously […].

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

BioSpace
AUGUST 11, 2020
Please check out the biopharma industry coronavirus (COVID-19) stories that are trending for August 12, 2020.

XTalks
AUGUST 11, 2020
It’s hard enough to get older adults to get routine vaccines but during a global pandemic, it’s nearly impossible. That’s why GlaxoSmithKline (GSK) has launched a disease awareness campaign — called “ Brought to You By Vaccines ” — to help those ages 50 and older identify gaps in their own immunity against vaccine-preventable diseases. Despite the high rates of vaccination among children in the US, with nearly 93 percent of kids ages 1.5 to three having received multiple doses of the polio vacci

BioSpace
AUGUST 11, 2020
A recent report in the New England Journal of Medicine reveals that the racial and ethnic minority groups most negatively-impacted by COVID-19 are also the least represented in these clinical studies.

BioPharma Reporter
AUGUST 11, 2020
The Bill & Melinda Gates Foundation provides $150 million for production of $3 doses of COVID-19 vaccines.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
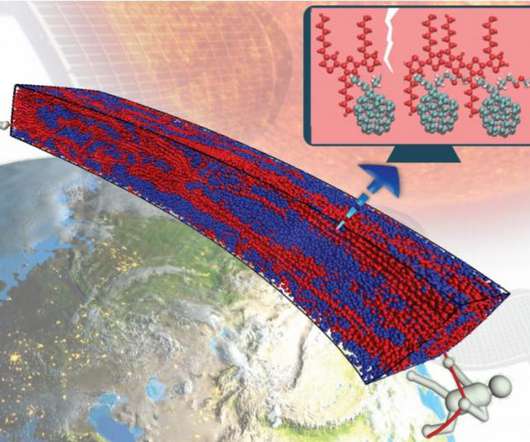
Scienmag
AUGUST 11, 2020
Computational experiments on semiconducting polymers show under harsh loading conditions — stretching and compression – the addition of small molecules enhances performance and stability; points to promising new direction for solar cell research Credit: Professor Ganesh Balasubramanian’s laboratory (Group for Interfacial and Nanoengineering), Department of Mechanical Engineering and Mechanics, Lehigh University, Bethlehem, USA.

Pharma Times
AUGUST 11, 2020
Independent Data Monitoring Committee recommended continuing Phase IIb study

Scienmag
AUGUST 11, 2020
Research conducted at an EPA facility on the UNC-Chapel Hill campus shows that expired and sterilized used N95 masks are still very good at protecting wearers. Credit: UNC School of Medicine CHAPEL HILL, NC – Aug. 11, 2020 – This spring, due to limited national supplies of N95 face masks, hospitals across the country asked […].

Pharma Times
AUGUST 11, 2020
Up to 750,000 unused testing kits to be recalled

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

BioSpace
AUGUST 11, 2020
Whether you’re an experienced professional in the field or just embarking on your new career, it’s time to look at your qualifications and career plans from an entirely different perspective.

pharmaphorum
AUGUST 11, 2020
Intouch Group has announced the hire of Susan Perlbachs as its chief creative officer (CCO). As CCO, Perlbachs will be responsible for creative leadership across all Intouch Group affiliates, locations and clients. She will be based in Intouch’s New York office. “I’m looking forward to being part of this award-winning organisation that I’ve admired for quite some time,” said Perlbachs. “I’m thrilled to work with the incredibly talented executive creat
BioSpace
AUGUST 11, 2020
Just-published research in Cell outlines a way to make even immunotherapy-resistant cancers treatable.

Pharma Times
AUGUST 11, 2020
Drug cleared for use in SMA patients aged two months or older

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content