AstraZeneca to buy Alexion for $39B
Bio Pharma Dive
DECEMBER 12, 2020
The deal launches AstraZeneca, a company that has been growing mostly because of newer drugs for cancer and diabetes, into the market for rare disease treatments.

Bio Pharma Dive
DECEMBER 12, 2020
The deal launches AstraZeneca, a company that has been growing mostly because of newer drugs for cancer and diabetes, into the market for rare disease treatments.
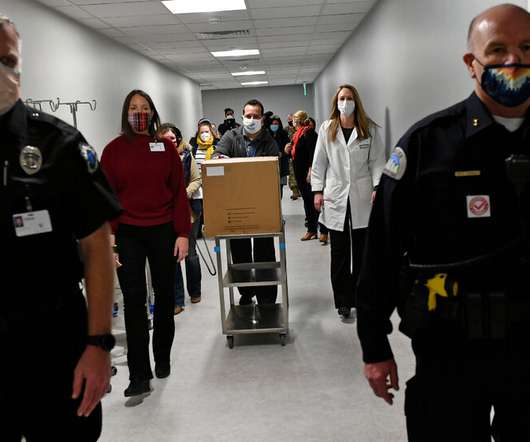
NY Times
DECEMBER 12, 2020
Distributing supplies is a daunting logistical challenge, involving sophisticated tracking equipment, military contingencies and tight security.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

The Pharma Data
DECEMBER 12, 2020
Greater scientific presence in immunology by adding Alexion ‘s innovative complement-technology platforms and strong pipeline . Dedicated rare disease unit to be headquartered in Boston. Geographical presence to be enhanced with broad coverage across primary, speciality and highly specialised care. Double-digit revenue growth through 2025; acquisition strengthens AstraZeneca’s broad-based revenue and the company will further globalise Alexion’s portfolio .
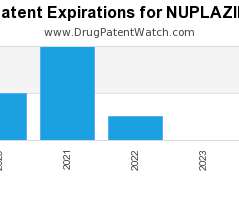
Drug Patent Watch
DECEMBER 12, 2020
Annual Drug Patent Expirations for NUPLAZID Nuplazid is a drug marketed by Acadia Pharms Inc and is included in two NDAs. It is available from one supplier. There are eighteen…. The post New patent expiration for Acadia Pharms drug NUPLAZID appeared first on DrugPatentWatch - Make Better Decisions.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
The Pharma Data
DECEMBER 12, 2020
SATURDAY, Dec. 12, 2020 – The U.S. Food and Drug Administration approved Pfizer’s coronavirus vaccine for emergency use on Friday, clearing the way for the launch of a national campaign to inoculate enough Americans to stem the spread of COVID-19. In what looks like a powerful turning point in the pandemic, the United States becomes the sixth country — in addition to Britain, Bahrain, Canada, Saudi Arabia and Mexico — to authorize the vaccine, The New York Times reported.

NY Times
DECEMBER 12, 2020
The deal for the biopharmaceutical company is the latest big takeover this year, even amid the pandemic.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Drug Patent Watch
DECEMBER 12, 2020
DEXILANT (dexlansoprazole) Takeda pharms usa Patent: 6,462,058 Expiration: Dec 15, 2020 See More … For more information on how DrugPatentWatch can help with your pharmaceutical business intelligence needs, contact admin@DrugPatentWatch.com…. The post Drug Patent Expirations for the Week of December 13, 2020 appeared first on DrugPatentWatch - Make Better Decisions.

BioTech 365
DECEMBER 12, 2020
Yiling Pharmaceutical, a Chinese Enterprise Going Global Under Intricate International Situations Yiling Pharmaceutical, a Chinese Enterprise Going Global Under Intricate International Situations SHIJIAZHUANG CITY, China–(BUSINESS WIRE)–In recent years, the advance of traditional Chinese medicine to the world has become a … Continue reading →
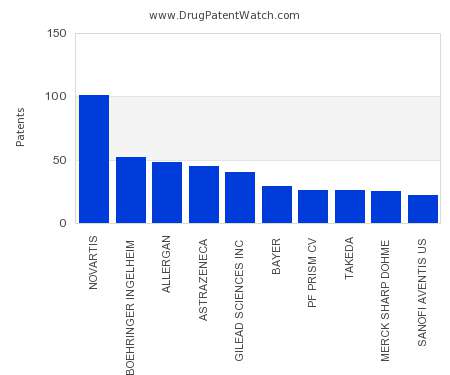
Drug Patent Watch
DECEMBER 12, 2020
This chart shows the pharmaceutical companies with the most patents in Brazil. Patents must be filed in each country (or, in some cases regional patent office) where patent protection is…. The post Which pharmaceutical companies have the most drug patents in Brazil? appeared first on DrugPatentWatch - Make Better Decisions.

BioTech 365
DECEMBER 12, 2020
Amarin Reports Encouraging Efficacy and Safety Results from Pilot Study Treating COVID-19 Infected Outpatients with VASCEPA® (Icosapent Ethyl) in Late Breaker Presentation at National Lipid Association (NLA) Scientific Sessions 2020 Amarin Reports Encouraging Efficacy and Safety Results from Pilot Study … Continue reading →

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

The Pharma Data
DECEMBER 12, 2020
Sanofi and GlaxoSmithKline (GSK) have decided to push back the phase 2b trial of their adjuvanted COVID-19 vaccine after finding it produced only a weak immune response in older patients, delaying the potential delivery of a vaccine that could contribute to the mass vaccination effort in the U.S. The companies announced the delay Friday, disclosing that although interim results from its 441-participant phase 1/2 study showed the vaccine elicited an immune response comparable to recovered patient

BioTech 365
DECEMBER 12, 2020
National Lipid Association Releases Official Scientific Statement on Coronary Artery Calcium Scoring for ASCVD Risk Reduction National Lipid Association Releases Official Scientific Statement on Coronary Artery Calcium Scoring for ASCVD Risk Reduction JACKSONVILLE, Fla., Dec.

XTalks
DECEMBER 12, 2020
Advancements in the logistics industry are often imagined to be self-driving cars or drones that can deliver packages, but these goals are currently more aspirational than they are tangible. Still, there is a great deal of transformation happening in logistics firms today. There are many new technological advancements that firms are incorporating to enhance their performance and reduce the costs of their retail delivery strategies for the food and beverage industry.

BioTech 365
DECEMBER 12, 2020
Intellia Therapeutics Achieves Normal Human Alpha-1 Antitrypsin Protein Levels in Non-Human Primates Through Targeted Gene Insertion for the Treatment of AAT Deficiency Intellia Therapeutics Achieves Normal Human Alpha-1 Antitrypsin Protein Levels in Non-Human Primates Through Targeted Gene Insertion for the … Continue reading →

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

XTalks
DECEMBER 12, 2020
Advancements in the logistics industry are often imagined to be self-driving cars or drones that can deliver packages, but these goals are currently more aspirational than they are tangible. Still, there is a great deal of transformation happening in logistics firms today. There are many new technological advancements that firms are incorporating to enhance their performance and reduce the costs of their retail delivery strategies for the food and beverage industry.

BioTech 365
DECEMBER 12, 2020
AstraZeneca to Acquire Alexion, Accelerating the Company’s Strategic and Financial Development AstraZeneca to Acquire Alexion, Accelerating the Company’s Strategic and Financial Development Greater scientific presence in immunology by adding Alexion’s innovative complement-technology platforms and strong pipeline Dedicated rare disease unit … Continue reading →

BioTech 365
DECEMBER 12, 2020
U.S. CDC Committee of Independent Health Experts Recommends Vaccination with Pfizer and BioNTech COVID-19 Vaccine for Persons Ages 16 Years and Older U.S.
Let's personalize your content