5 trends in biotech dealmaking to watch in 2021
Bio Pharma Dive
JANUARY 13, 2021
Brain drugs, private equity players and blank-check companies are just some of the things deal experts say they'll be keeping an eye on this year.

Bio Pharma Dive
JANUARY 13, 2021
Brain drugs, private equity players and blank-check companies are just some of the things deal experts say they'll be keeping an eye on this year.
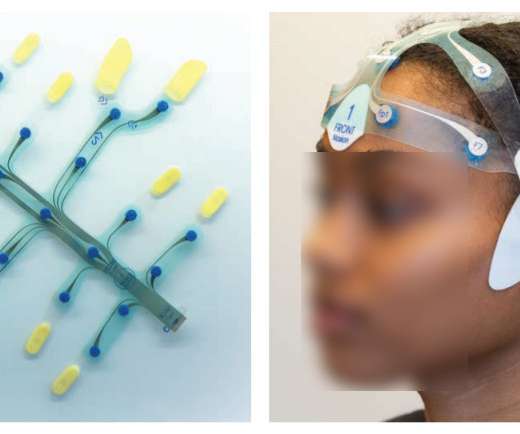
Pharma Mirror
JANUARY 10, 2021
By Boris Goldstein, Co-Founder & Executive Chairman at Brain Scientific COVID-19 is known for its impacts on a patient’s respiratory system, but it has also been found to affect individuals in unique ways and with varying severity levels depending on their age, risk factors, and comorbidities. Several studies have recently shown that the development of neurological conditions and symptoms are prevalent in COVID patients.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

World of DTC Marketing
JANUARY 15, 2021
HEY NOW: If you want a great example of why pharma needs to build expertise in-house and rely less on agencies, read the newest white paper from Intouch Group. I read the whitepaper, and it was full of vague obscurities that are, frankly, embarrassing. Want a great example of agency babble? Read this. “Pharma needs to turn what was a very smart, fast patchwork of change into something that becomes sustainable, durable and scalable, and start supporting HCPs in a complete shift in paradigm,

NY Times
JANUARY 12, 2021
In their first abortion case since Justice Amy Coney Barrett joined the court, the justices reinstated a requirement that women seeking medication abortions pick up a pill in person.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Bio Pharma Dive
JANUARY 11, 2021
Biogen faces a crucial FDA decision on its Alzheimer’s drug aducanumab, while Vertex gets used to investor pressure to do a deal. Sarepta, meanwhile, is picking up the pieces after a disappointing gene therapy readout.
pharmaphorum
JANUARY 15, 2021
J&J has announced early trial results that suggest its single-shot coronavirus vaccine provides a sustained response against the virus ahead of a phase 3 trial readout due later this month. The UK has caused controversy in recent weeks by tinkering with the dosing regimens for coronavirus vaccines from AstraZeneca and Pfizer/BioNTech, which are being rolled as part of the country’s mass vaccination programme.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.
NY Times
JANUARY 11, 2021
Participants with the disease still declined, but much more slowly than those receiving a placebo, investigators say.

Bio Pharma Dive
JANUARY 11, 2021
Study results could come soon for several coronavirus vaccines, as well as experimental medicines from Amgen, Vertex and Sage Therapeutics.

BioSpace
JANUARY 15, 2021
Janet Lambert, CEO of the Alliance for Regenerative Medicine (ARM), painted a rosy picture despite a few challenges at the 2021 Cell & Gene State of the Industry Briefing during this year’s Biotech Showcase, held virtually January 11-15.

World of DTC Marketing
JANUARY 11, 2021
KEY POINTS: The social media shutdown of far-right hate groups and speech is once again spotlighting freedom of speech issues. Whose responsibility is it to ensure that health information, for example, online is accurate and doesn’t lead to bad patient decisions? Pharma has an opportunity to help clarify misinformation via a content and personalization strategy.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

pharmaphorum
JANUARY 15, 2021
Pharma’s reputation is currently riding high as the industry comes together to tackle COVID-19, but GSK’s UK general manager Neale Belson says there is always more companies can do to build their culture and make sure employees and customers trust they are doing the right thing. When we spoke with Belson about the GSK’s response to COVID-19 , he pointed out how the pandemic has allowed him to get to know his colleagues even better than he anticipated.

Bio Pharma Dive
JANUARY 11, 2021
Full one-year results from a Phase 3 study show the therapy, known as Roctavian, sharply reduced bleeds and use of blood-clotting treatments.
BioSpace
JANUARY 11, 2021
Evidence continues to build for long-lasting immunity to COVID-19 following disease infection, with the publication of a new study showing a durable immune response as far as eight months after the initial onset of symptoms.

World of DTC Marketing
JANUARY 12, 2021
SUMMARY: TV doesn’t make sense for smaller and older patient populations. DTC marketers should be thinking digital and aligning execution with their needs. BMS has been advertising Opdivo via DTC TV ads but does TV really make sense? It’s estimated that 228,820 adults (116,300 men and 112,520 women) in the United States will be diagnosed with lung cancer.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
XTalks
JANUARY 15, 2021
Ortho Clinical Diagnostics received emergency use authorization (EUA) from the US Food and Drug Administration (FDA) for its VITROS SARS-CoV-2 antigen test for detection of active COVID-19 infection. The rapid antigen test can run up to 130 tests in an hour with and can deliver same-day results, making it useful for mass-scale COVID-19 testing. The antigen test provides precise, clinically reliable results generated on the company’s high-volume VITROS systems, which are installed in more than 5,

Bio Pharma Dive
JANUARY 12, 2021
Moderna and Gilead reported billions in sales for their coronavirus treatments, but Regeneron's antibody drug cocktail hasn't gained traction. BioNTech, meanwhile, could report critical new vaccine results in days.

BioSpace
JANUARY 14, 2021
Like many biopharma companies, the global COVID-19 pandemic has caused delays while also offering opportunities for NeuroRx.

pharmaphorum
JANUARY 15, 2021
Women with newly-diagnosed advanced ovarian cancer have a new treatment option in England after NICE recommend interim funding for GlaxoSmithKline’s Zejula (niraparib) in final guidance. Zejula competes with AstraZeneca/Merck & Co’s PARP inhibitor class rival Lynparza (olaparib) but in this case it has an advantage in this maintenance therapy use as it can be used regardless of whether the BRCA mutation is present.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.
BioPharma Reporter
JANUARY 11, 2021
Codagenixâs intranasal COVID-19 vaccine has entered Phase 1 clinical trials: with the first volunteers dosed in the UK.
Bio Pharma Dive
JANUARY 15, 2021
Binney Street Capital plans to invest in eight to 10 companies over the next three years. Developing new drugs will be a core focus, though investments could also go toward health services, digital health or diagnostics.

BioSpace
JANUARY 13, 2021
The COVID-19 pandemic that has upended life across the globe has also ignited a reawakening to the value of vaccines, and Hu believes the momentum will carry forward into the new year.

Scienmag
JANUARY 13, 2021
International Ukraine Genetic Diversity Project finds a quarter of the genetic variation in Europe, dramatically increasing information on population diversity and medical genetic variation Credit: Oakland University Today, the largest study of genetic diversity in Ukraine was published in the open science journal GigaScience. The project was an international effort, bringing together researchers in Ukraine, […].

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

XTalks
JANUARY 12, 2021
International Delight has partnered with Post Holdings to combine coffee creamers with breakfast-favorite cereals, Fruity Pebbles and Cocoa Pebbles. The products will be available nationwide for a limited time. The leading coffee creamer is bringing a unique, exciting and nostalgic experience to coffee lovers who crave stimulating flavors. Pebbles celebrate their 50 th birthday this year and they decided to celebrate this milestone in 2021 by partnering with International Delight. “At Inte
Bio Pharma Dive
JANUARY 11, 2021
Analysts cautioned that results for Lilly's drug, donanemab, weren't a total home run. Even so, investors sent the company's market value higher by roughly $18 billion.

Druggist
JANUARY 9, 2021
Migraine is a common condition affecting an estimated 6 million people in the UK. With approximate 190,000 migraine attacks experienced by people in the UK (NICE, 2012), pharmacies are among the first points of call to find over the counter migraine medication. What is the best painkiller for migraine? Today I will review common over the counter migraine medication.
pharmaphorum
JANUARY 15, 2021
South Korea’s Celltrion has announced encouraging top-line results for its potential antibody therapy for COVID-19, with the first part of a phase 2/3 trial showing it could cut recovery times and chances of progression from moderate to more severe disease. Several other companies are also working on antibody therapies and Eli Lilly’s bamlanivimab gained an FDA Emergency Use Authorization in November last year, shortly after Regeneron’s cocktail of casirivimab and imdevimab.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

XTalks
JANUARY 14, 2021
Mealworms, the larval form of the mealworm beetle, have become the first insect in Europe deemed safe for human consumption. After receiving an application from Micronsutris , a French insect farmer, the European Food Safety Authority (EFSA) found no safety concerns for dried yellow mealworms. The EFSA’s decision is the first step in heralding in EU-wide approval of snacks and other foods containing the insect as an ingredient.

Bio Pharma Dive
JANUARY 13, 2021
The deal expands an earlier agreement between Regeneron and the U.S., though many of the issues limiting the drug's use haven't been solved.

Pharma Times
JANUARY 11, 2021
Partnership will aim to research and implement quantum computing in pharma R&D
BioSpace
JANUARY 15, 2021
Mesenchymal stem cells derived from umbilical cord tissue could help treat ARDS in COVID-19 patients, according to results of a Phase I/IIa clinical trial published by University of Miami researchers.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.
Let's personalize your content