8 key clinical trials to watch for the rest of 2021
Bio Pharma Dive
JUNE 21, 2021
The next six months could feature clinical milestones for CRISPR gene editing, the treatment of COVID-19, microbiome drugs and gene-targeted cancer therapy.
Bio Pharma Dive
JUNE 21, 2021
The next six months could feature clinical milestones for CRISPR gene editing, the treatment of COVID-19, microbiome drugs and gene-targeted cancer therapy.

World of DTC Marketing
JUNE 23, 2021
SKIMMERS SUMMARY: Jefferies analysts found doctors have shown strong interest in prescribing Aduhelm for about 35% of early-stage Alzheimer’s patients with mild cognitive impairment after surveying 50 U.S. neurologists or psychiatrists currently treating about 12,000 Alzheimer’s patients. Source: Fierce Pharma If that enthusiasm pans out in the long run, it could mean at least 300,000 patients taking the drug, or $10 billion to $15 billion in peak Aduhelm from U.S. sales alone, which is above co
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Camargo
JUNE 23, 2021
While the COVID-19 pandemic has accelerated innovation in many areas, including clinical trial design, many recent advancements were already in motion before 2020 as sponsors and CROs sought to improve the patient journey and encourage trial participation. Traditional trials, especially those for pediatric populations, often place enormous burdens on patients and their families.
pharmaphorum
JUNE 21, 2021
It would be easy to forget that back in 2019, BioNTech was an early-stage biotech firmly focused on cancer vaccines, before being catapulted onto the world-stage with its COVID-19 shot. Now, the German company has issued a reminder of its original R&D mission with the start of the first phase 2 trial of BNT111 – the first of several cancer vaccine candidates – in patients with skin cancer.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Bio Pharma Dive
JUNE 23, 2021
A group of statisticians who had argued for rejection were overruled, internal memos show, as high-ranking agency officials got behind an accelerated clearance for Biogen's drug.

World of DTC Marketing
JUNE 24, 2021
SUMMARY: Consumers don’t real pay attention to fair balance in TV ads. Pharma TV ads drive people online to search for more information. Safety pages on Pharma product websites continue to have high utility. Only requirement in TV ads should be for “boxed” products. As we have seen over the past few years, Pharma companies are a business, a regulated business, but still a business.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharma Times
JUNE 22, 2021
An extra £40m will go toward enhancing mental health services for children and young people

Bio Pharma Dive
JUNE 22, 2021
New results from a key study show the treatment's benefit is holding up well over time. But the FDA wants to see more data, delaying when UniQure and partner CSL expect to file for approval.

JAMA Internal Medicine
JUNE 21, 2021
This cross-sectional study uses administrative health care data on 2.9 million households from the first 45 weeks of 2020 to assess the association between social gatherings and SARS-CoV-2 transmission by studying whether COVID-19 rates increase after birthdays in a household.
BioSpace
JUNE 21, 2021
Use of the anti-parasitic drug ivermectin could reduce COVID-19 related deaths, data from a peer-reviewed study shows.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
pharmaphorum
JUNE 22, 2021
Sanofi and Translate Bio have started a first clinical trial of an mRNA-based seasonal flu vaccine, adding to the COVID-19 shot already in testing as part of their three-year old collaboration. Enthusiasm for mRNA vaccines is riding high amid the successful rollout of COVID-19 shots from Pfizer/BioNTech and Moderna, and attention is now turning toe the possibility of using the approach to tackle other infectious diseases.
Bio Pharma Dive
JUNE 23, 2021
Led by CEO Samantha Singer, Abata Therapeutics has $95 million to advance its plans to engineer regulatory T cells for treating autoimmune diseases.

XTalks
JUNE 21, 2021
In a new study, Amazon’s Halo app has been shown to predict body fat percentage with the same level of accuracy as standard lab testing. The app uses just four photos taken from a smartphone — of the front, back and each side of the body — to build a 3D model and produce body fat percentage results using the app’s built-in machine learning algorithms.

Pharma Times
JUNE 21, 2021
Aubagio has been authorised for the treatment of paediatric patients aged ten to 17 years living with RRMS

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
BioSpace
JUNE 21, 2021
Recent research shows those who had been hospitalized due to COVID-19 might already have a reduced sense of smell and taste, not just for the short term, but possibly for life.

Bio Pharma Dive
JUNE 22, 2021
Results from a Phase 3 study could help Merck secure a full approval for use of Keytruda in the tumor type, potentially extending the therapy's edge over rival therapies.
Scienmag
JUNE 20, 2021
Researchers shed light on a potential intervention that could make tumors responsive to modern-day immunotherapies Credit: Mei Huang, PhD BOSTON – Researchers from Massachusetts General Hospital (MGH) have discovered a biological mechanism that transforms cells exposed to carcinogens from environmental factors like smoking and ultraviolet light into immunogenic cells that can be harnessed therapeutically to […].

Pharma Times
JUNE 21, 2021
Children treated with gene therapy prior to symptom presentation achieved motor milestones not seen in the natural history of SMA

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

pharmaphorum
JUNE 25, 2021
Former Cognoa executive Andy Molnar has been appointed chief executive of the Digital Therapeutics Alliance, a global trade association for companies developing evidence-based DTx products. Molnar, who served as vice president of market access and government affairs at Cognoa, joins the four-year-old trade association at a time when the potential of using DTx to deliver remote healthcare has been thrust into the spotlight by the coronavirus pandemic.
Bio Pharma Dive
JUNE 24, 2021
The lack of specific data frustrated analysts, but shares in Arcus rose by double digits Thursday. Partner Gilead is expected to make a decision on whether to license the drug later this year.
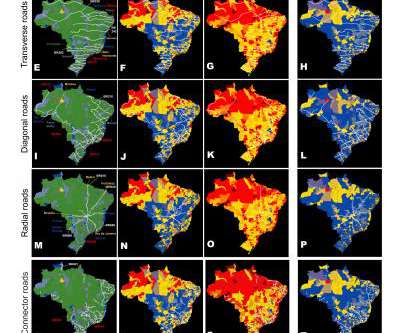
Scienmag
JUNE 21, 2021
The impact of super-spreader cities, highways, and intensive care availability in the early stages of the COVID-19 epidemic in Brazil Credit: Nicolelis, M.A.L. et al. 2021. Scientific Reports, doi.org/10.1038/s41598-021-92263-3 São Paulo, Brazil – A multidisciplinary analysis by a group of Brazilian scientists, published online in the journal Scientific Reports on June 21, 2021, revealed that […].

XTalks
JUNE 23, 2021
AstraZeneca scored a major win last week in a lawsuit launched by European Union (EU) officials that accused the drugmaker of failing to deliver on promised supplies of its COVID-19 vaccine. A Brussels court found that the European Commission has no exclusivity or right of priority over contracting parties, and also dismissed other measures being sought by the Commission.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
pharmaphorum
JUNE 22, 2021
AstraZeneca’s SGLT2 inhibitor Farxiga has been growing fast thanks to a first-in-class approval in heart failure, but will now have to share the market with Boehringer Ingelheim and Eli Lilly’s rival therapy Jardiance. . The European Commission has become the first regulator to approved Jardiance (empagliflozin) as a treatment for adults with symptomatic chronic heart failure with reduced ejection fraction (HFrEF), also known as systolic heart failure.

Bio Pharma Dive
JUNE 25, 2021
A key committee of the EMA gave a positive review of Astellas and Fibrogen's roxadustat, as well as Bristol Myers Squibb's cell therapy Abecma and UCB's psoriasis drug Bimzelx.

Scienmag
JUNE 20, 2021
Credit: Kuanish Reymbaev The first global standards to embed health and wellbeing into the education system have been created amid a rise in mental health problems during the COVID-19 pandemic. Researchers at the Centre for Adolescent Health at the Murdoch Children’s Research Institute (MCRI) led the two-year project at the invitation of the World Health […].
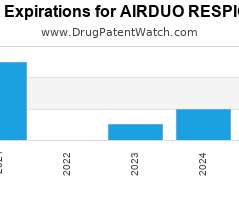
Drug Patent Watch
JUNE 22, 2021
Annual Drug Patent Expirations for AIRDUO+RESPICLICK Airduo Respiclick is a drug marketed by Teva Pharm and is included in one NDA. It is available from three suppliers. There are twenty-one…. The post New patent expiration for Teva Pharm drug AIRDUO RESPICLICK appeared first on DrugPatentWatch - Make Better Decisions.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
pharmaphorum
JUNE 21, 2021
The NHS App has soared in popularity since it was updated in May to allow people to record their COVID-19 vaccination status, with more than 2 million new signups taking the total number of users to 6 million. . The app – which should not be confused with the NHS COVID-19 track and trace app – was used by five million people in the last month, according to the Department of Health and Social Care.
Bio Pharma Dive
JUNE 21, 2021
Final clinical trial results confirmed treatment dramatically reduced the risk of hospitalization and death among adults with mild-to-moderate COVID-19.
Scienmag
JUNE 21, 2021
Scientists find a way to efficiently use visible light from the sun to break down CO2, open doors to novel means of alleviating global warming Credit: Image courtesy: Shinji Kawasaki and Yosuke Ishii from Nagoya Institute of Technology Carbon dioxide (CO2) emissions from human activities have risen drastically over the last century and a half […].
Drug Patent Watch
JUNE 22, 2021
Annual Drug Patent Expirations for ARMONAIR+RESPICLICK Armonair Respiclick is a drug marketed by Teva Pharm and is included in one NDA. It is available from one supplier. There are nineteen…. The post New patent expiration for Teva Pharm drug ARMONAIR RESPICLICK appeared first on DrugPatentWatch - Make Better Decisions.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.
Let's personalize your content