Clearmind licenses psychedelic compounds for mental disorders
Pharmaceutical Technology
APRIL 18, 2024
Clearmind Medicine has entered into a licensing agreement with Yissum to develop Generation 3.0 psychedelic compounds for mental disorders.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
APRIL 18, 2024
Clearmind Medicine has entered into a licensing agreement with Yissum to develop Generation 3.0 psychedelic compounds for mental disorders.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Outsourcing Pharma
SEPTEMBER 14, 2023
Insilico Medicine is due to receive $80 million upfront plus potential milestone payments as Exelixis gains global rights to develop and commercialize the Hong Kong firmâs small molecule cancer treatment.

STAT News
JANUARY 5, 2023
Last fall, Novartis agreed to license a best-selling cancer drug so that generic companies could make copies for distribution to 44 low- and middle-income nations, marking the first time a voluntary license was arranged for a cancer medicine. ANNECY, FRANCE — At first blush, the deal was hailed as a breakthrough.

Bio Pharma Dive
APRIL 23, 2024
Shares of the biotech rose about 5% after a medicine licensed from Takeda hit the main and key secondary goals of a mid-stage study focused on major depressive disorder.
Pharmaceutical Technology
JUNE 20, 2023
The US Food and Drug Administration (FDA) has now granted fast-track and rare paediatric disease designations, and the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) has also granted promising innovative medicine status to vamorolone for DMD.

Pharmaceutical Technology
MARCH 24, 2023
Moderna has entered a strategic partnership with Generation Bio for the development of non-viral genetic medicines. Moderna’s biological and technical expertise will be combined with core technologies of the non-viral genetic medicine platform from Generation Bio.

Pharma Mirror
APRIL 18, 2023
Dudley, UK, April 18th 2023: Sterling Pharma Solutions, a global contract development and manufacturing organisation, today announced that it has been granted a Manufacturer’s Authorisation for Investigational Medicinal Products from the United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA).

Bio Pharma Dive
NOVEMBER 9, 2021
rights to rimegepant — a sizable licensing deal, though not the buyout that some of the biotech's investors appeared to be hoping for. The pharmaceutical giant will pay Biohaven $500 million for ex-U.S.
Pharmaceutical Technology
OCTOBER 4, 2022
Shionogi & Co and the United Nations (UN)-backed public health organisation Medicines Patent Pool (MPP) have entered a voluntary licence agreement for the former’s oral Covid-19 antiviral candidate ensitrelvir fumaric acid (S-217622).
Pharmaceutical Technology
AUGUST 11, 2022
Gemini Therapeutics has signed a definitive agreement to merge with Disc Medicine in an all-stock deal to create a clinical-stage biopharmaceutical company. The merged company is expected to be named Disc Medicine, which will have corporate headquarters in Watertown, Massachusetts, US. Last year, Disc in-licensed bitopertin from Roche.
Pharmaceutical Technology
JUNE 19, 2023
Bio-Thera Solutions and Biomm have entered a licensing and supply agreement for Bio-Thera’s BAT2206, a ustekinumab biosimilar. It plans to seek regulatory approval from the European Medicines Agency, the China National Medical Products Administration and the US Food and Drug Administration.
Pharmaceutical Technology
JANUARY 23, 2023
“We look forward to utilising our development and commercial capabilities to expand the potential of this innovative medicine to patients beyond China. “We We have a strong track record of working with companies that share our focus on bringing transformative medicines to patients around the globe who need them.”
Pharmaceutical Technology
APRIL 25, 2023
German biotechnology firm 3B Pharmaceuticals (3BP) has entered into a licensing agreement with Novartis Innovative Therapies for its fibroblast activation protein (FAP)-targeting peptide technology. 3BP receives an initial payment of $40m, and $425m as development, regulatory and commercial milestone payments.

Bio Pharma Dive
OCTOBER 27, 2021
The deal with the Medicines Patent Pool is meant to increase access in lower-income countries to molnupiravir, an experimental drug that could become the first oral treatment for COVID-19.
Pharmaceutical Technology
JULY 28, 2022
“By leveraging our expansive commercial infrastructure, we will provide the resources needed to optimise the commercialisation of rezafungin and ensure patient access to this life-saving medicine in the US. “We The post Melinta and Cidara sign licensing deal for rezafungin appeared first on Pharmaceutical Technology.

Pharmaceutical Technology
MARCH 17, 2023
Pharmanovia and Aeterna Zentaris have entered an exclusive licensing agreement for the latter’s medicine Ghryvelin (macimorelin). Under the deal, Pharmanovia will acquire the exclusive rights and license to commercialise Ghryvelin in the EEA and the UK from Consilient Health.

Bio Pharma Dive
AUGUST 2, 2022
Founded by serial entrepreneur Alexis Borisy, the company comes equipped with two experimental medicines it licensed from Merck KGaA and Blueprint Medicines.

STAT News
OCTOBER 20, 2022
Amid a push to widen access to medicines to poor countries, Novartis has reached an agreement to license one of its best-selling cancer treatments so that generic manufacturers can produce copies for distribution to 44 low- and middle-income nations. Continue to STAT+ to read the full story…
STAT News
JULY 28, 2022
In a much ballyhooed move, ViiV Healthcare finalized a deal to license its long-acting injectable HIV prevention shot to the Medicines Patent Pool so that generic companies can make and distribute lower-cost versions to low- and middle-income countries.

BioSpace
DECEMBER 11, 2023
Backed by ARCH Venture Partners and Fujifilm, as well as technology licensed from MIT, Tome is looking to create curative cell and integrative gene therapies.

BioTech 365
NOVEMBER 18, 2020
Foundation Medicine Enters into Patent Licensing and Technology Agreement with TwinStrand Biosciences Foundation Medicine Enters into Patent Licensing and Technology Agreement with TwinStrand Biosciences CAMBRIDGE, Mass.–(BUSINESS
Bio Pharma Dive
DECEMBER 14, 2021
Fitusiran, which Sanofi licensed from Alnylam, could finally get to market after trial delays and safety concerns slowed its progress. But gene therapies and other new medicines might provide competition.
BioPharma Reporter
DECEMBER 7, 2023
Symbiosis Pharmaceutical Services can now test small molecule, biological and advanced therapeutical medicinal products (ATMP) following a Â1 million investment.

Fossil Remedies
OCTOBER 16, 2022
To launch the best PCD pharma franchise company, you will have to acquire the necessary licenses from authorities. The next important thing is the license. Every pharma company needs a drug license number to begin distribution, marketing, and manufacturing activities. License required to start a Pharmaceutical Business in India.

BioTech 365
AUGUST 17, 2021
Revive Therapeutics Signs Exclusive Worldwide License Agreement For Medicinal Mushroom Ganoderma Lucidum To Treat Cancer Revive Therapeutics Signs Exclusive Worldwide License Agreement For Medicinal Mushroom Ganoderma Lucidum To Treat Cancer Novel discovery and research originated from Universidad Central del Caribe … Continue reading → (..)

Bio Pharma Dive
JANUARY 31, 2023
For $10 million up front, UniQure has licensed rights to its second experimental treatment for the condition, a medicine from startup Apic Bio that should enter human testing later this year.

STAT News
DECEMBER 21, 2022
California authorities are seeking to revoke a license held by a CVS Health mail-order pharmacy unit for violating several state laws that govern shipments of various controlled substances — including prescription painkillers and ADHD medicines — directly to patients.

Scienmag
JANUARY 12, 2022
Nicholas Hopkinson at Imperial College London welcomes the move, saying this will give doctors another means to help smokers quit. E-cigarettes are currently regulated as consumer products so cannot be promoted as smoking cessation aids, he explains.

Medical Xpress
MARCH 30, 2023
In a recent interview posted on JMIR TV, JMIR Publications' CEO Dr. Gunther Eysenbach speaks with Dr. Andrew Taylor from Yale University School of Medicine about their paper titled "How Does ChatGPT Perform on the United States Medical Licensing Examination?

BioTech 365
OCTOBER 27, 2021
The Medicines Patent Pool (MPP) and Merck Enter Into License Agreement for Molnupiravir, an Investigational Oral Antiviral COVID-19 Medicine, to Increase Broad Access in Low- and Middle-Income Countries The Medicines Patent Pool (MPP) and Merck Enter Into License Agreement for … Continue reading →

pharmaphorum
OCTOBER 21, 2022
The Medicines Patent Pool (MPP) and Novartis have unveiled a new voluntary licensing agreement to allow generic drugmakers in seven middle income nations to develop, manufacture, and supply generic versions of the leukaemia treatment nilotinib. Nilotinib is a twice-daily oral medication used to treat chronic myeloid leukaemia (CML).

BioTech 365
JUNE 25, 2021
Aurinia Announces Licensing Partner Otsuka Filed Initial Marketing Authorization Application (MAA) for Voclosporin with the European Medicines Agency (EMA) Aurinia Announces Licensing Partner Otsuka Filed Initial Marketing Authorization Application (MAA) for Voclosporin with the European Medicines Agency (EMA) VICTORIA, British … Continue reading (..)
STAT News
OCTOBER 20, 2022
Three dozen institutional investors are urging the boards at several of the world’s largest pharmaceutical companies to establish concrete metrics for linking executive compensation with policies that widen access to medicines to low- and middle-income countries.

Bio Pharma Dive
MAY 30, 2023
The biotech, which went public in 2021 to develop a cancer drug licensed from Daiichi Sankyo, may try to acquire a different tumor-targeting medicine to grow its pipeline.

pharmaphorum
NOVEMBER 16, 2022
The 2022 edition of the Access to Medicine Index (ATMI) – which places the top 20 pharma companies for their efforts to improve delivery of medicines to lower income countries – sees GSK retain the number one position, newly joined by Johnson & Johnson in second and AstraZeneca in third. Jayasree Iyer.

Bio Pharma Dive
MARCH 30, 2023
The licensing deal for the FDA-approved medicine, Brexafemme, includes milestone payments that could add up to $503 million.
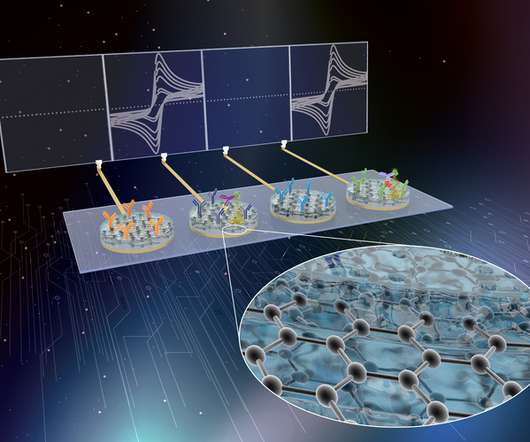
Scienmag
MARCH 7, 2022
announced that the Institute’s electrochemical eRapid technology has been licensed to Antisoma Therapeutics Pty. The licensing agreement was coordinated by Harvard University’s Office of Technology Development and grants the company exclusive, worldwide access to the Wyss Institute’s […]. a subsidiary of The iQ Group Global.

Drug Discovery World
FEBRUARY 16, 2023
Challenges remain with current delivery approaches, and genetic medicines applications have been generally restricted to the liver and ex vivo settings. Aera’s technology also includes a licensed therapeutic enzyme platform based on the discovery of novel, compact, and programmable gene editing enzymes.
Bio Pharma Dive
OCTOBER 27, 2020
The startup, which aims to develop lower-cost alternatives to branded medicines, has licensed two drugs from CStone Pharmaceuticals in a deal that could signal a coming price war in cancer immunotherapy.

pharmaphorum
MARCH 24, 2022
There’s a rich history of finding useful medicines from fungi, from the antibiotic penicillin to immune suppressant cyclosporine and cholesterol drug lovastatin. The post GSK partners LifeMine on fungi-derived medicines appeared first on. Image by jggrz from Pixabay .
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?



Let's personalize your content