UK MHRA strengthens regulations on fluoroquinolone antibiotics
Pharmaceutical Technology
JANUARY 23, 2024
The UK MHRA has introduced further restrictions on the usage of fluoroquinolone antibiotics, strengthening the previous regulations.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
 Regulation Related Topics
Regulation Related Topics 
Pharmaceutical Technology
JANUARY 23, 2024
The UK MHRA has introduced further restrictions on the usage of fluoroquinolone antibiotics, strengthening the previous regulations.
Pharmaceutical Technology
OCTOBER 9, 2023
Diabetes researchers oppose the regulation of islet transplantation as a biologic, following the approval of CellTrans’ Lantidra.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JANUARY 16, 2024
As the deadline nears for the government to reveal its proposed prices, pharmaceutical companies are continuing to fight regulation.

Pharmaceutical Technology
APRIL 23, 2024
At the Swiss Biotech Day Conference, key opinion leaders discussed ways to reduce disparities caused by reduced regulatory capacities.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

AuroBlog - Aurous Healthcare Clinical Trials blog
DECEMBER 13, 2022
The National Commission for Homoeopathy has prepared a regulation defining a structure to the Bachelor of Homoeopathic Medicine and Surgery (BHMS) course with changes including incorporation of a methodology to supplement modern advancement, research and technology in Homoeopathy (SMART-Homoeopathy).

BioPharma Reporter
MARCH 19, 2024
With the pharma and healthcare landscape increasingly being shaped by artificial intelligence (AI), the US Food and Drug Administration (FDA) has taken proactive steps to outline a comprehensive strategy for regulating AI in medical products.
pharmaphorum
MARCH 1, 2024
There is a pressing need for companies to transition their EU clinical trials that are ongoing under the “old” regulatory framework of the Clinical Trials Directive to the Clinical Trials Regulation. The risk for those that don’t meet the 30th January 2025 deadline for transition is that they will lose their legal basis.

Pharmaceutical Technology
DECEMBER 7, 2023
GlobalData’s report assesses the drugs in the Apoptosis Regulator BAX pipeline by therapy areas, indications, stages, MoA, RoA, molecule type and the key players in the development pipeline.

Pharmaceutical Technology
DECEMBER 7, 2023
GlobalData’s report assesses the drugs in the Apoptosis Regulator BAX pipeline by therapy areas, indications, stages, MoA, RoA, molecule type and the key players in the development pipeline.
Pharmaceutical Technology
NOVEMBER 8, 2023
The executive order highlights the US sentiment towards AI regulation, but presents a fragmented approach with possibility of discord.

Bio Pharma Dive
APRIL 12, 2024
The decision clears a concern hanging over GLP-1 medicines like Ozempic since reports of suicidal ideation and self-harm among people taking the drugs surfaced in July.

Pharmaceutical Technology
MARCH 23, 2023
This week, the Medicines and Healthcare products Regulatory Agency (MHRA) is introducing major changes to clinical trial regulation in the UK with expedited timelines at several stages. The MHRA hopes this new framework “will remove obstacles to innovation” and “streamline the regulation of clinical trials” amongst other things.

Bio Pharma Dive
JULY 14, 2021
The go-ahead from British regulators was the last hurdle standing in the way of the two companies wrapping up their $39 billion deal announced last year.

Pharmaceutical Technology
APRIL 23, 2024
Discover Novartis AG's groundbreaking patent for the innovative KIR-CAR, a chimeric antigen receptor revolutionizing T cell therapy. Explore the unique components and potential applications of this game-changing technology.

Bio Pharma Dive
DECEMBER 16, 2022
The EMA’s drug review committee recommended five new drugs be approved in Europe during its December meeting, including CSL’s Hemgenix, AstraZeneca’s Imjudo and Amicus’ Pombiliti.
Bio Pharma Dive
APRIL 20, 2021
Regulators in the U.S. The EMA reiterated the vaccine's benefits outweigh its risks but recommended a warning be added to highlight the rare, but serious side effect. are expected to soon make a similar decision.
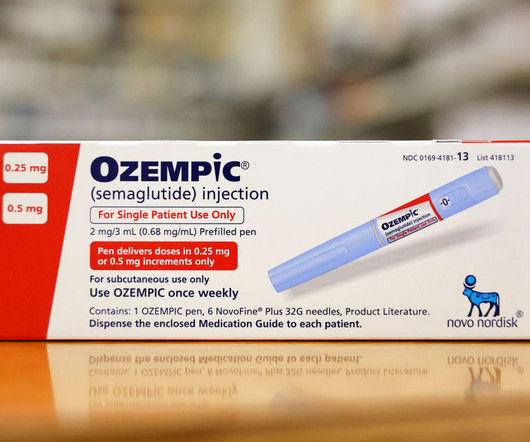
Fierce Pharma
APRIL 12, 2024
Eight months after the European Medicines Agency (EMA) opened an investigation into a potential link between popular GLP-1 receptor agonists and suicidal thoughts or actions, the agency has determi | The regulators began investigating meds in the GLP-1 drug class for a potential association in July after two reported cases of suicidal thoughts and (..)

ProRelix Research
NOVEMBER 27, 2023
The United States Food and Drug Administration (FDA) has strict regulations regarding pharmacovigilance activities that are a part of post-marketing studies for drugs and medical devices that are required as […] The post FDA Pharmacovigilance Regulations Update appeared first on ProRelix Research.

Bio Pharma Dive
JULY 22, 2022
The positive decision comes as governments aim to expand supply and quicken distribution of the vaccine to combat a widening global outbreak. Separately, the EMA backed approvals of 11 other medicines.

Fierce Pharma
OCTOBER 13, 2023
Europe's drug regulator wasn't swayed by Amylyx's phase 2 data. Even as Amylyx’s ALS drug gains steam in the U.S. after a long-awaited FDA approval, the company is struggling with regulatory hurdles overseas. The European Commission will make the final call on the drug's approval in the bloc by the end of the year.

Bio Pharma Dive
JANUARY 19, 2022
The news sparked headlines about an attempt by regulators to target big tech, but it could have serious implications for healthcare, too.

XTalks
FEBRUARY 26, 2024
Key developments in food regulations in 2024 are expected, focusing on safety, transparency and public health. For stakeholders in the food industry, understanding and adapting to these regulations is not just a matter of compliance but a strategic imperative for business success.

BioSpace
SEPTEMBER 20, 2023
With 88% of life sciences organizations using or planning to use AI in recruitment and/or hiring, AI regulation is a priority for the industry.

World of DTC Marketing
FEBRUARY 21, 2022
The post The drug industry continues to dare regulation appeared first on World of DTC Marketing.com. With over 80% of voters aligned that drug prices are too high big pharma continues to act recklessly as if to say, “we’ll do what’s best for investors and us regardless of the effects on society.”

Bio Pharma Dive
JANUARY 26, 2022
The decision is a further sign of regulators' scrutiny of accelerated approvals for cancer therapies, an initiative that's led to several market withdrawals.

Bio Pharma Dive
DECEMBER 15, 2023
While a positive recommendation for the gene editing treatment Casgevy was expected, a clearance for Biogen’s Skyclaris, acquired through a recent acquisition, was no sure bet.
Pharmaceutical Technology
APRIL 18, 2023
Three regulators in different jurisdictions have accepted Bristol Myers Squibb’s applications for Abecma (idecabtagene vicleucel) for earlier use to treat triple-class exposed relapsed and/or refractory multiple myeloma in adult patients.

ProRelix Research
DECEMBER 14, 2023
The European Medicines Agency (EMA) coordinates the European […] The post EMA Pharmacovigilance Regulations Updates appeared first on ProRelix Research. Pharmacovigilance is the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other medicine-related problem.

Bio Pharma Dive
DECEMBER 1, 2023
The EMA's safety committee has more questions for makers of the in-demand therapies as it reviews whether the drugs are linked to the risk of suicidal thoughts.

Bio Pharma Dive
FEBRUARY 4, 2022
In a regulatory filing, Biogen said the FTC and the SEC are looking for information about Aduhelm's approval and marketing, meaning the drug is now the focus of four government probes.

ProRelix Research
FEBRUARY 12, 2023
Historical events such as the sulfanilamide elixir tragedy that resulted in the mass poisoning and the deaths of hundreds of patients in the 1930s and the thalidomide scandal which caused […] The post Clinical Trial Regulations in the United States appeared first on ProRelix Research.

NPR Health - Shots
MARCH 12, 2023
Neighbors were inundated with smoke and left wondering why the site wasn't regulated in the first place. An unregulated landfill that accepts vegetative waste has burned underground for months. Image credit: Zoe McDonald/WBHM)

pharmaphorum
AUGUST 25, 2023
Mapping the landscape of clinical trial regulation Mike.Smith Fri, 25/08/2023 - 10:00 Bookmark this

FDA Law Blog
JANUARY 28, 2024
Gonzalez — In a new publication in the Washington Legal Foundation’s (WLF) Legal Backgrounder , we argue that FDA’s recent proposed rule regulating Laboratory Developed Tests (LDTs) is vulnerable to legal challenge, once finalized. By Jeffrey N. Gibbs & Steven J.

Bio Pharma Dive
MAY 20, 2022
The brain-infused treatment for a rare neurological disease would be one of only three gene therapies commercially available in the EU. PTC plans to ask for FDA approval later this year.
Bio Pharma Dive
JULY 10, 2023
Three reports from Iceland spurred the EMA to evaluate whether certain Novo Nordisk drugs, including Ozempic, might cause thoughts of self harm.

FDA Law Blog
MARCH 31, 2024
Gibbs — On March 21, 2024, the House Energy and Commerce held a subcommittee hearing titled “Evaluating Approaches to Diagnostic Test Regulation and the Impact of the FDA’s Proposed Rule.” By Ana Loloei & Jeffrey N. FDA, which was not invited to participate, would surely have concurred.

Pharma Marketing Network
OCTOBER 13, 2023
In the ever-evolving digital landscape, pharma marketers must stay up to date with the latest ad regulations. Navigating the complex web of rules and regulations requires careful consideration and preparation. This includes understanding the various laws and regulations that govern digital advertising.

Bio Pharma Dive
NOVEMBER 17, 2021
A European Medicines Agency panel tasked with reviewing drugs voted against the biotech's controversial Alzheimer's medicine, signaling an approval is unlikely next month

NPR Health - Shots
JULY 30, 2022
And a lack of federal regulation or guidance has states stepping in. Delta-8 is a hemp product designed to get users high, but misleading labels or added chemicals have caused people to get sick.

FDA Law Blog
MARCH 10, 2024
Hyman, Phelps & McNamara Director Larry Houck will participate as a panelist focusing on this timely topic at the Food and Drug Law Institute’s (“FDLI’s”) Legal and Practical Issues in Cannabis Regulation Conference next month. The conference, held in Washington, D.C., April 4th and 5th, is an in-person and virtual event.

Bio Pharma Dive
OCTOBER 30, 2023
In addition to Sanofi’s investment, the biotech signaled it’s weighing deals with “multiple” other companies that have expressed interest in its work.

pharmaphorum
OCTOBER 11, 2022
An explosion of digital mental health tools being offered for sale has presented regulators with a problem – how should they be classified and assessed for safety or efficacy? ” The post Wellcome funds work on regulating digital mental health tools appeared first on. . The healthcare charity is providing £1.8
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content