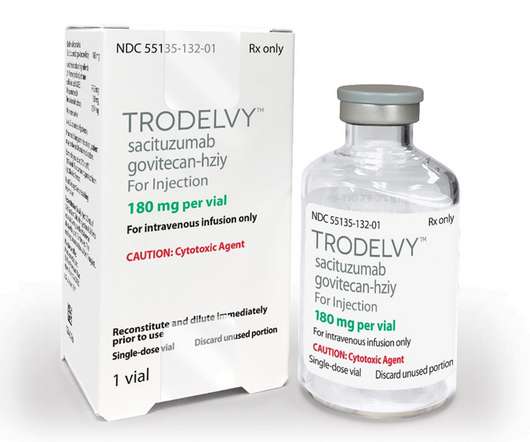
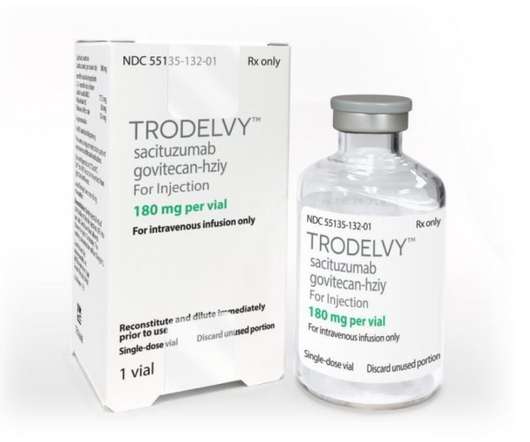
Trodelvy has been Approved for ER+/HER2- Patients with Advanced or Metastatic Breast Cancers
XTalks
FEBRUARY 7, 2023
HR+/HER2- breast cancer is the most common type of breast cancer, with the National Cancer Institute (NCI) estimating 287,850 new cases of female breast cancer in 2022 alone. Despite decades of advances, people living with pre-treated HR+/HER2- metastatic breast cancer need new treatment options. months vs. 11.2




















Let's personalize your content