FDA generic drug approvals rose in 2023 in bid for improved access
Pharmaceutical Technology
FEBRUARY 23, 2024
The FDA Office of Generic Drugs reported a rise in generic drug approvals, as several first-time generics entered the market.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
FEBRUARY 23, 2024
The FDA Office of Generic Drugs reported a rise in generic drug approvals, as several first-time generics entered the market.

Pharmaceutical Technology
OCTOBER 25, 2023
Experts discuss the key trends in quality improvements and API reshoring for the generic drugs market at CpHI Europe.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Patent Watch
OCTOBER 2, 2023
The FDA conducted a study to identify factors that may predict the likelihood of generic drug marketing applications.

Drug Patent Watch
OCTOBER 6, 2020
Here is a copy of the talk I gave at the recent Marcusevans 13th Portfolio Management and Pipeline Optimization for Generics. I cover: How to find and evaluate generic entry…. The post Finding and Evaluating Generic Drug Market Entry Opportunities appeared first on DrugPatentWatch - Make Better Decisions.

STAT News
MARCH 30, 2023
Supreme Court to review a controversy over so-called skinny labels for medicines, arguing that an appeals court finding threatens the availability of lower-cost generic drugs. For instance, a generic drug could be marketed to treat one type of heart problem, but not another.

BioSpace
JANUARY 14, 2021
Billionaire entrepreneur Mark Cuban, best known as the owner of the Dallas Mavericks and an investor on the ABC business reality series “Shark Tank,” is diving into generic drugs with a new startup, dubbed Mark Cuban Cost Plus Drug Company.
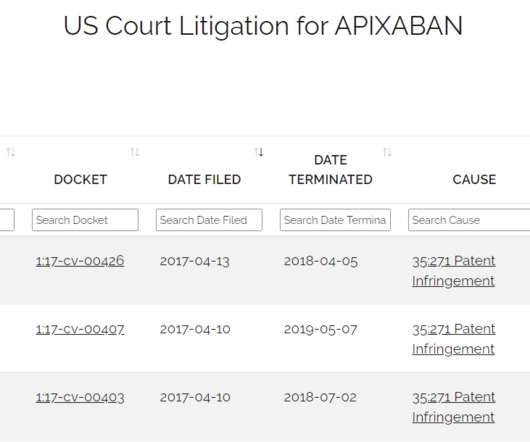
Drug Patent Watch
AUGUST 12, 2020
Just because a drug has received FDA approval does not mean that it is available in the marketplace. The post Generic Drugs Approved but not Launched – How to Tell When Generic Drugs Will hit the Market appeared first on DrugPatentWatch - Make Better Decisions.

BioTech 365
JUNE 25, 2021
GCC Generic Drug Market Trends, Share, Size, Opportunity and Forecasts 2021-2026 – ResearchAndMarkets.com GCC Generic Drug Market Trends, Share, Size, Opportunity and Forecasts 2021-2026 – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “GCC Generic Drug Market: Industry Trends, Share, Size, Growth, Opportunity and Forecast (..)

BioTech 365
OCTOBER 4, 2021
Canada Generic Drug Market Industry Trends, Share, Size, Growth, Opportunity and Forecasts 2021-2026 – ResearchAndMarkets.com Canada Generic Drug Market Industry Trends, Share, Size, Growth, Opportunity and Forecasts 2021-2026 – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “Canada Generic Drug Market: Industry Trends, (..)

BioTech 365
MAY 28, 2021
Global Generic Drugs Market Report 2021: The New Generics Era – The Patent Cliff, Types of Generic Drugs, Simple Generics, Super Generics, Biosimilars, ANDA Approvals – ResearchAndMarkets.com Global Generic Drugs Market Report 2021: The New Generics Era – The Patent … Continue reading →

BioTech 365
NOVEMBER 18, 2021
Brazil Generic Drug Market (2021 to 2026) – Industry Trends, Share, Size, Growth, Forecast and Opportunities – ResearchAndMarkets.com Brazil Generic Drug Market (2021 to 2026) – Industry Trends, Share, Size, Growth, Forecast and Opportunities – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “Brazil Generic (..)

NY Times
SEPTEMBER 18, 2021
Competition for market share at rock-bottom prices has led to shortages, price-spikes, allegations of price-fixing, and substandard and even dangerous practices.

FDA Law Blog
SEPTEMBER 6, 2023
Farquhar — A drug manufacturer’s bad post-inspection grade from the U.S. Food and Drug Administration – labeled an “Official Action Indicated” classification – is generally devastating for the facility, not least because it can stall FDA approval of applications to market drugs manufactured at the facility.

BioTech 365
APRIL 15, 2021
Global Generic Drugs (Small Molecule Generics vs Biosimilars) Market Forecast & Opportunities, 2021-2026 by Application, Drug Delivery, Form, Source, Distribution Channel – ResearchAndMarkets.com Global Generic Drugs (Small Molecule Generics vs Biosimilars) Market Forecast & Opportunities, 2021-2026 by Application, Drug Delivery, … (..)
Pharmaceutical Commerce
FEBRUARY 9, 2024
Technavio analysis notes that an increase drug patent expirations could be a contributing factor.

BioTech 365
APRIL 26, 2021
United States Generic Drugs Market Report 2020-2026: Focus on CNS, Cardiovascular, Dermatology, Genitourinary/Hormonal, Respiratory, Anti-infective & Oncology – ResearchAndMarkets.com United States Generic Drugs Market Report 2020-2026: Focus on CNS, Cardiovascular, Dermatology, Genitourinary/Hormonal, Respiratory, Anti-infective & (..)

STAT News
MARCH 30, 2023
What happened to the market for monkeys? Are some drugs too cheap? We also discuss what leads to generic drug shortages, whether every major pharmaceutical firm needs a weight-loss drug, and what it means when drug company cancels a conference appearance. And why are biotech stocks still in the tank?

Pharmaceutical Technology
NOVEMBER 21, 2022
Japan is currently the fourth largest market in the world. Based on GlobalData estimates, the Japanese pharmaceutical market generated JPY9.392 trillion ($67.32 Other contributing factors include the decreasing number of post-market studies and difficulties in primary research and manufacturing.
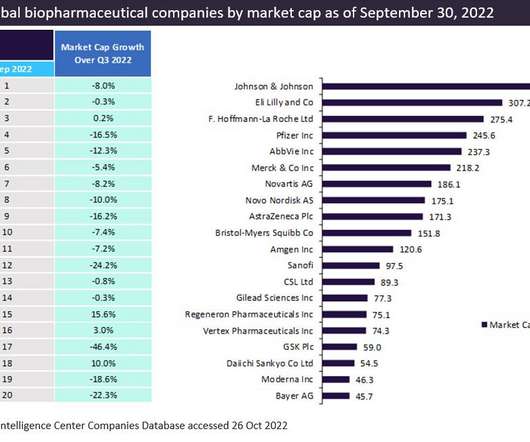
Pharmaceutical Technology
DECEMBER 20, 2022
The top 20 global biopharmaceutical companies exhibited a downward trend in aggregate market capitalisation by 9.1% This downturn in market cap was attributed to a decline in the demand for Covid-19 vaccines and therapies. trillion in Q2 2022 to $3.14 Eli Lilly was in second place, followed by Roche and Pfizer. and 10%, respectively.
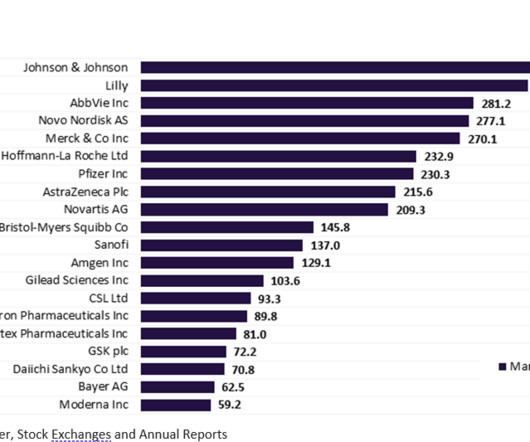
Pharmaceutical Technology
MAY 16, 2023
More than half of the top 20 global biopharmaceutical companies saw a fall in market capitalisation over Q1 2023. decline in total aggregate market capitalisation from $3.61 Bayer reported the highest market capitalisation growth of 23.1% Sanofi and Regeneron’s market capitalisation grew by 12.4% This resulted in a 3.4%

Pharmaceutical Technology
SEPTEMBER 15, 2022
Various factors have contributed to the need and growth of API chemical suppliers such as rising healthcare expenditure, increasing disposable incomes, growing geriatric population, increasing incidence of chronic diseases, patent expiration of blockbuster drugs, increased consumption of generic drugs, and intervention of the new generation APIs.

Pharma in Brief
FEBRUARY 8, 2024
On January 26, 2024, Health Canada published a Notice concerning its application of the definition of Canadian Reference Product ( CRP ) in the Food and Drug Regulations ( Regulations ). For instance, the generic drug must be “the pharmaceutical equivalent of the [CRP]” and be “bioequivalent with the [CRP]”.

FDA Law Blog
APRIL 2, 2024
Perhaps unsurprisingly given the extraordinary focus on drug pricing in the last decade, generic competition—FDA’s only real way to have an effect on drug pricing—tops this year’s list. FDA believes this change would effectuate timelier and more cost-efficient generic drug development.”

Pharmaceutical Technology
NOVEMBER 21, 2022
Japan is currently the fourth largest market in the world. Based on GlobalData estimates, the Japanese pharmaceutical market generated JPY9.392 trillion ($67.32 Other contributing factors include the decreasing number of post-market studies and difficulties in primary research and manufacturing.

Fierce Pharma
NOVEMBER 22, 2023
Looking to protect its lucrative immunology drug Rinvoq, AbbVie is suing a clutch of generic drug makers that are attempting to market copycat versions of the blockbuster. |
Outsourcing Pharma
OCTOBER 12, 2020
Technology impacting generic drugs include artificial intelligence, telemedicine, fraud-preventing digital solutions and more, according to a report.
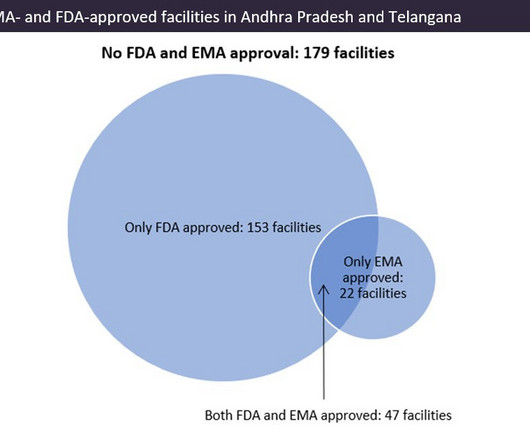
Pharmaceutical Technology
APRIL 4, 2023
Indian pharma manufacturing continues to be the backbone of drug supplies worldwide, and GlobalData analysis suggests US overreliance on the country for generic drug supply. Source: GlobalData, Pharma Intelligence Center Drug Database (Accessed March 20, 2023). © GlobalData. ©GlobalData.

FDA Law Blog
APRIL 8, 2024
Department of Health and Human Services (HHS) recently published a White Paper on Policy Considerations to Prevent Drug Shortages and Mitigate Supply Chain Vulnerabilities in the United States —with input from several HHS stakeholders, including FDA, CMS, and the Administration for Strategic Preparedness and Response. By Kalie E.

Drug Channels
MAY 2, 2023
In my most recent video webinar , I explored how the rapid expansion of patient-paid prescriptions—via cash-pay pharmacies and discount card vendors—is transforming the prescription market. Below, I follow the dollar when a patient uses a discount card to pay for a generic drug prescription. d/b/a Drug Channels Institute.

BioPharma Reporter
DECEMBER 22, 2022
Expanding generic drug competition and a decline in the demand for COVID-19 vaccines and therapies have brought down the aggregate market capitalization of the global top 20 biopharmaceutical companies by 9.1%.

World of DTC Marketing
APRIL 12, 2021
The goal of getting a drug to market as fast as possible so they can recoup drug development costs has the potential for mistakes that could cost lives. Coupled with all this is the continued outsourcing of raw materials and other steps in drug development. Up to 90% of drugs now sold in the US and UK are generic.

Drug Channels
JUNE 22, 2023
In my March video webinar , I explored how the rapid expansion of patient-paid prescriptions—via cash-pay pharmacies and discount card vendors—is transforming the prescription market. Below, I follow the dollar when a patient uses a discount card to pay for a generic drug prescription. d/b/a Drug Channels Institute.

Pharmaceutical Technology
MAY 31, 2023
According to GlobalData, Phase I drugs for Solid Tumor have a 54% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. GlobalData’s report assesses how Sym-021’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks. Buy the report here.

Pharma in Brief
NOVEMBER 5, 2023
On October 19, 2023, the Ontario government proposed regulatory amendments that may eliminate the need to submit certain clinical studies and evidence when requesting that a “well-established drug” be (i) listed or (ii) designated as interchangeable on Ontario’s publicly-funded formulary. Requirements for obtaining a listing are set out in O.

pharmaphorum
JULY 29, 2021
The UK has levied another big fine for anticompetitive activity in the pharma market in a fortnight, with Advanz Pharma and former owners on the hook for more than £100 million ($140 million) after increasing the price of a thyroid disease drug by 1,110% over an eight-year period. ” Advanz itself was directly fined £40.9

pharmaphorum
JUNE 6, 2022
American entrepreneur and investor Mark Cuban tells us why he and his partners launched the Mark Cuban Cost Plus Drug Company (MCCPDC), a public-benefit corporation and online pharmacy that provides patients access to medications at a lower cost. . “Manufacturers get a bad rap as being the source of high pricing for patient meds.

Pharmaceutical Technology
JULY 15, 2022
Nothing epitomises market failures more than the cost of insulin,” Newsom said in a video posted on Twitter. CivicaRx is a non-profit generic drug company producing affordable insulin biosimilars in collaboration with the Juvenile Diabetes Research Foundation (JDRF). In-house manufacturing the norm.

The Pharma Data
MARCH 15, 2022
Agency Supports Development of Complex Generic Drug-Device Combination Product to Improve Competition and Access to More Affordable Medicines. This complex generic drug-device combination product, which is a metered-dose inhaler, should not be used to treat acute asthma attacks. Today, the U.S.

pharmaphorum
JULY 29, 2021
Mylan’s Semglee has been approved for over a year as a regular biosimilar to Lantus (insulin glargine), meaning that it could be used in place of Sanofi’s drug, but only if specifically prescribed for a patient. Biosimilars marketed in the US typically have list prices 15% to 35% lower than their reference products at launch.

Pharmaceutical Technology
FEBRUARY 24, 2023
The overhaul will address drug marketing exclusivity length, pricing, patient access, innovation incentives, antimicrobial resistance, clinical trials, supply chain security and shortages, and environmental impact. The EU is planning a sweeping revision of its pharma legislation in March, the largest change in 20 years.

World of DTC Marketing
JUNE 22, 2022
A new feature—a sensor that monitors changes in the heart rate for irregularities that can lead to strokes and heart failure—has just been approved by America’s Food and Drug Administration (FDA). Then there is the cost of generic drugs. billion in 2020 if it had purchased 77 of 89 drugs from Cuban’s firm.

Drug Channels
NOVEMBER 28, 2023
In this issue: A reality check on Express Scripts’ purported “cost-based” pharmacy network Mark Cuban explains how and why the PBM market will change Why two drug prices are better than one Pharmacists sing the retail pharmacy shakeout blues Plus, two Philly legends meet at a cardiology conference! d/b/a Drug Channels Institute.

Drug Discovery World
APRIL 10, 2023
Could India become an important centre of drug discovery innovation? India is traditionally known as ‘the pharmacy of the world’ due to its huge pharmaceuticals manufacturing capacity and high quantity of exports, particularly generic medicines and vaccines. Diana Spencer explores the evolution of the Hyderabad life sciences hub.

FDA Law Blog
MAY 25, 2022
That effort, which has most recently emerged in the forms of the “ Bringing Low-cost Options and Competition while Keeping Incentives for New Generics Act of 2021 ” ( H.R. 2853 , or “BLOCKING Act”), the “ Expanding Access to Low-Cost Generics Act of 2021 ” ( S. And now there’s a report and analysis to back up that claim!
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content