Scribe and Sanofi expand genetic therapy development deal
Pharmaceutical Technology
JULY 18, 2023
Scribe Therapeutics and Sanofi have expanded partnership to progress the development of in vivo genetic therapies to treat genomic diseases.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
JULY 18, 2023
Scribe Therapeutics and Sanofi have expanded partnership to progress the development of in vivo genetic therapies to treat genomic diseases.

Drug Discovery World
NOVEMBER 14, 2023
Since the mutation only affects a single gene, it makes it a prime candidate for genome editing treatments. Verve-101 is an investigational, in vivo base editing medicine designed to be a single-course treatment that inactivates the PCSK9 gene in the liver to durably lower blood LDL-C. “Of
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Drug Discovery World
MARCH 9, 2023
The post FDA clears in vivo gene editing therapy for trials in US appeared first on Drug Discovery World (DDW). The US Food and Drug Administration (FDA) has cleared Intellia Therapeutics’ Investigational New Drug (IND) application for NTLA-2002 for the treatment of hereditary angioedema (HAE).

XTalks
OCTOBER 20, 2023
Clinical-stage genome editing company Intellia Therapeutics has received clearance from the US Food and Drug Administration (FDA) for its Investigational New Drug (IND) application to start a pivotal phase III trial of NTLA-2001 for the treatment of transthyretin (ATTR) amyloidosis with cardiomyopathy.

pharmaphorum
JUNE 28, 2021
The first ever clinical data with a CRISPR/Cas9 drug used to edit the genomes of cells within the body has yielded impressive results in patients with ATTR amyloidosis, a life-threatening rare disease. . — Eric Topol (@EricTopol) June 26, 2021. — Eric Topol (@EricTopol) June 26, 2021.
Drug Discovery World
DECEMBER 12, 2022
The meeting will focus on the application of genome engineering in identification of novel drug targets through large-scale CRISPR based functional genomics studies, developing advanced cellular and in vivo disease models for target validation, and its applications in therapeutic genome editing. Register here.

Roots Analysis
FEBRUARY 27, 2024
The global genome editing market is anticipated to grow at a CAGR of 12.6% The global genome editing market is anticipated to grow at a CAGR of 12.6% Recently, in July 2019, a first in vivo clinical trial for a CRISPR-based therapy was initiated. during the forecast period 2023-2035. during the forecast period 2023-2035.

BioTech 365
MAY 11, 2021
New Preclinical Data Presented at the 2021 American Society of Genetic & Cell Therapy Annual Meeting Highlights Precision BioSciences’ ARCUS In Vivo Genome Editing New Preclinical Data Presented at the 2021 American Society of Genetic & Cell Therapy Annual Meeting … Continue reading →

BioSpace
MARCH 10, 2021
Intellia Therapeutics presented preclinical data of its non-viral genome editing platform at the Keystone eSymposium: Precision Engineering of the Genome, Epigenome and Transcriptome.

Pharmaceutical Technology
MAY 17, 2023
Scribe Therapeutics has entered a strategic collaboration with Eli Lilly and Company subsidiary Prevail Therapeutics for accelerating in vivo CRISPR-based therapies to target the causes of serious neurological and neuromuscular diseases. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Drug Discovery World
APRIL 10, 2023
Genome Valley Another driving factor has been the creation and investment in Genome Valley in Hyderabad, India’s first structured hub for life sciences R&D, which has established Hyderabad as the country’s major life sciences cluster, ranking above Chennai and Bengaluru. Hyderabad is recognised as the vaccine hub of India.
Pharmaceutical Technology
FEBRUARY 15, 2023
Innovation S-curve for the pharmaceutical industry CRISPR nuclease is a key innovation area in pharmaceutical development CRISPR, which refers to clustered regularly interspaced short palindromic repeats, are bacteriophage-derived DNA sequences that had previously infected the prokaryote and are found in the genomes of bacteria and archaea.

BioSpace
AUGUST 31, 2021
On June 26, Intellia announced the first-ever clinical data supporting the safety and efficacy of in vivo CRISPR genome editing in human patients.

Pharmaceutical Technology
FEBRUARY 23, 2023
Moderna has entered a strategic research and development partnership with ElevateBio’s Life Edit Therapeutics to discover and develop new in-vivo mRNA gene editing therapies. The company’s nuclease collection includes several Protospacer Adjacent Motifs (PAMs), short sequences that help determine the genome’s DNA segments.

pharmaphorum
JUNE 23, 2022
Novartis has shouldered its way into the in vivo gene editing category via a deal with US biotech Precision BioSciences, focused on a therapy for sickle cell disease (SCD). Precision Bio aims to address those challenges using its proprietary ARCUS nuclease platform, which is designed to insert a transgene with high accuracy into the genome.
Drug Discovery World
DECEMBER 15, 2022
Inducing double strand breaks at unintended locations in the genome can lead to off-target genetic modifications that are difficult to predict and potentially deleterious. Complicating matters even more, researchers currently lack effective and reliable methods for detecting and measuring off-target genomic events.
Pharmaceutical Technology
SEPTEMBER 29, 2022
The alliance will leverage the CRISPR genome editing technologies of Scribe to facilitate in genetic modification of new natural killer (NK) cell therapeutics for cancer. Under the agreement, Sanofi will receive non-exclusive rights to Scribe’s CRISPR by Design platform of wholly-owned enzymes for developing ex vivo NK cell therapies.

pharmaphorum
JANUARY 29, 2021
Genome editing is an exciting but still nascent field, and companies in the area face as many obstacles as they do opportunities. Sangamo CEO Sandy Macrae told us how his company is being cautious about the hype and finding ways to be financially viable in an emerging space. Zinc fingers. billion in funding.
Drug Discovery World
APRIL 24, 2024
The strategic partnership will combine Variant Bio’s genomic discovery capabilities and VB-Inference platform with Evotec’s knowledge of antifibrotic drug discovery. The strategic partnership will combine Variant Bio’s genomic discovery capabilities and VB-Inference platform with Evotec’s knowledge of antifibrotic drug discovery.

XTalks
MAY 22, 2024
John Finn, PhD Chief Scientific Officer Tome Biosciences Dr. Finn has over 20 years of experience in the gene therapy space with a focus on genome editing and delivery technologies. PGI is a cutting-edge gene editing technology that allows for the insertion of large sequences of DNA with site-specific precision.

Drug Discovery World
AUGUST 15, 2023
Recombinant lentiviral vectors are used for ex vivo transgene delivery in several gene-modified cell therapies, such as chimeric antigen receptor (CAR) T cell therapy.

Pharmaceutical Technology
FEBRUARY 15, 2023
They are engineered to cut specific genomic targets in order to modify the expression of single genes and proteins. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Gene splicing using nucleases.
Drug Discovery World
AUGUST 15, 2023
Reliable analytical methods such as digital PCR to characterise lentiviral vectors and the cells transduced with them are critical to helping ensure the safety and efficacy of these biologics. The post New whitepaper explores digital PCR for lentiviral vector characterisation appeared first on Drug Discovery World (DDW).
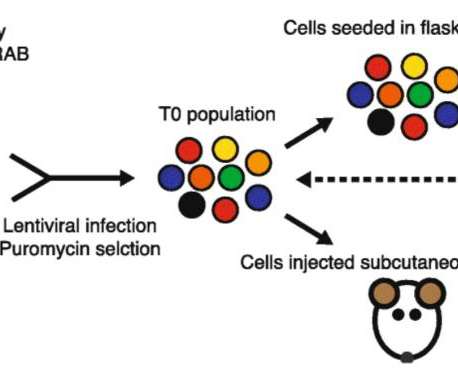
Scienmag
NOVEMBER 16, 2020
Wnt-regulated lncRNA discovery enhanced by in vivo identification and CRISPRi functional validation. Genome Med 12, 89 (2020). Singapore scientists uncover potential role of long non-coding RNAs in pancreatic cancer Credit: From Figure 4 in Liu, S., Harmston, N., Glaser, T.L.

Drug Discovery World
APRIL 8, 2024
Aviv Regev, Head and Executive Vice President of Research and Early Development at Genentech, opened the session with a discussion of how advances in genomic sequencing are fostering the development of data-rich cell atlases and what that data could mean for new insights into cancer biology and novel therapeutics.

Pharmaceutical Technology
FEBRUARY 23, 2023
DNA binding site prediction, peptide structure optimisation, and AI-assisted genome analysis are some of the accelerating innovation areas, where adoption has been steadily increasing. However, not all innovations are equal nor do they follow a constant upward trend.

The Pharma Data
DECEMBER 13, 2020
Nasdaq: DTIL) a clinical stage biotechnology company dedicated to improving life with its novel and proprietary ARCUS® genome editing platform, today announced that Abid Ansari, Chief Financial Officer, notified the Company that he will be leaving the organization after nearly five years to pursue a new career opportunity. “On DURHAM, N.C.,

Drug Discovery World
DECEMBER 6, 2023
“We are impressed by the unique delivery and editing technologies that Eligo has developed, and we believe this will set the stage for Eligo to forge new paths in the application of in vivo gene-editing technologies.” The investment will fuel pre-IND and IND activities to achieve early human data readouts in a PhIb/IIa clinical trial.
Pharmaceutical Technology
FEBRUARY 14, 2023
These systems include human and mouse cell lines, and even in vivo in live animals. These systems include human and mouse cell lines, and even in vivo in live animals. However, not all innovations are equal and nor do they follow a constant upward trend.

Drug Discovery World
JULY 18, 2023
Rolf Turk , Senior Manager, Genomics Medicine at Integrated DNA Technologies, examines how CRISPR is being used to enhance cancer therapies. Years of development and fast-paced research have continued to unlock its potential, expanding how CRISPR can be used to treat, detect, and prevent common diseases such as cancer and Covid-19.

Drug Discovery World
MARCH 26, 2024
More importantly, it also highlighted that significant challenges remain in selecting the right therapies for the right patients despite the progress made through genomic testing and implementation of circulating tumour (ct) DNA analyses. We were there to present the latest findings from our research.

The Pharma Data
DECEMBER 2, 2020
” VANCOUVER, BC, December 02, 2020 /24-7PressRelease/ — Eyam Vaccines and Immunotherapeutics (EYAM) today announced that former President and CEO of Genome Prairie is joining EYAM. Dr. Pontarollo’s primary areas of research and technical expertise include Genomics, Molecular Biology, Vaccine development and Immunology.
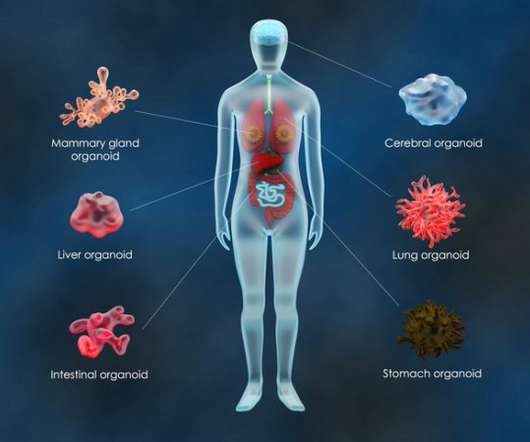
Drug Discovery World
JANUARY 12, 2023
The genome is the blueprint for life, consisting of genes made up of thousands of pairs of DNA bases. CRISPR allows researchers to target and edit specific genes in order to better understand the genome, human diseases and the effects of specific mutations. What are organoids? .

Pharmaceutical Technology
MARCH 9, 2023
Innovation S-curve for the pharmaceutical industry Transgenic murine models is a key innovation area in pharmaceutical s Transgenic murine models refer to mice that have been genetically altered for the purposes of understanding the in vivo functions of genes.

pharmaphorum
JULY 13, 2022
Verve Therapeutics has started dosing patients in a phase 1b trial of its in vivo gene-editing drug for high cholesterol, designed to permanently switch off the PCSK9 gene with a one-shot treatment. dosing of *first patient* with VERVE-101, an in vivo CRISPR base editing medicine. Today: we are announcing.

The Pharma Data
DECEMBER 13, 2020
which develops genome editing technologies to accelerate drug discovery and develop novel therapeutics for a broad range of diseases, today announced the appointment of Bo Zhang, Ph.D., 14, 2020 10:00 UTC. BEIJING & CAMBRIDGE, Mass.–( –( BUSINESS WIRE )– EdiGene, Inc. as Head of the US Subsidiary, and Kehua Fan, M.D.,
Drug Discovery World
APRIL 25, 2024
For drug discovery and development, cell lines allow researchers to test the efficacy of therapeutics before moving into in vivo studies. It is a crucial for the development of biopharmaceuticals since they use a living host to produce the therapeutic in question.

The Pharma Data
JUNE 7, 2023
Acuitas’ LNP technology will support Bayer’s in vivo gene editing and protein replacement programs by specifically delivering RNA payloads to the desired target organ, the liver. a biotechnology company specializing in the development of lipid nanoparticle (LNP) delivery systems for molecular therapeutics. Source link: [link]

Drug Discovery World
APRIL 4, 2023
The Food and Drug Administration (FDA) in the US has made several key drug decisions over the last few weeks. Here’s a summary. Orserdu (elacestrant), Stemline Therapeutics The first and only treatment specifically indicated for patients with ESR1 mutations in ER+, HER2- advanced or metastatic breast cancer was approved in the US in February.

Drug Discovery World
JANUARY 17, 2024
CTCs reflect high levels of tumour heterogeneity, represent clonal evolution, and are suitable for treatment selection, real-time longitudinal disease monitoring and in vitro/in vivo culture. Brett Swansiger, Chief Commercial Officer at ANGLE discusses the importance of liquid biopsies in precision medicine trials for cancer.
Drug Discovery World
JANUARY 23, 2023
There are more than 2,000 microRNAs coded for in the human genome, and different tissues express them at different levels. There are more than 2,000 microRNAs coded for in the human genome, and different tissues express them at different levels. DS: How will your work ultimately impact disease treatment?

XTalks
DECEMBER 20, 2023
As we step into 2024, the life sciences continue to evolve at an unprecedented pace, driven by technological innovation, a deeper understanding of human biology and the application of new technologies in areas like drug development and health wearables. Regulatory bodies are also taking note of the applications of AI in drug development.
Drug Discovery World
FEBRUARY 6, 2023
The company is co-founded by former team leaders and multi-disciplinary tool builders from 10x Genomics. We isolate antibodies from single human B cells using single cell genomics and assess, engineer, and advance these antibodies with other technologies. This has consequences for safety and efficacy.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content