Nearing FDA approval anniversary, Phathom gets more bad news on Voquezna impurities
Fierce Pharma
FEBRUARY 10, 2023
Nearing FDA approval anniversary, Phathom gets more bad news on Voquezna impurities fkansteiner Fri, 02/10/2023 - 08:45
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 news fda-approvals
news fda-approvals 
Fierce Pharma
FEBRUARY 10, 2023
Nearing FDA approval anniversary, Phathom gets more bad news on Voquezna impurities fkansteiner Fri, 02/10/2023 - 08:45
BioSpace
DECEMBER 9, 2020
All eyes on the FDA as they review a COVID-19 vaccine, FDA approval of an Emergency Use Authorization to LabCorp’s home test kit for COVID-19, the first testing device that does not require a prescription and more news.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioTech 365
JUNE 9, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: FDA grants approval for Alzheimer’s disease treatment. from European Biotechnology – first and foremost in European biotech [link].

BioTech 365
JUNE 9, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: AdComm member quits over FDA approval of Biogen’s Aduhelm.AdComm member quits over FDA approval of Biogen’s Aduhelm ntaylor Wed, 06/09/2021 – 06:08 from FierceBiotech: Biotech [link].

BioTech 365
APRIL 9, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Provention warns FDA requests could delay diabetes approval.Provention warns FDA requests could delay diabetes approval ntaylor Fri, 04/09/2021 – 05:51 from FierceBiotech: Biotech [link].

BioTech 365
OCTOBER 18, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Biogen’s ALS drug has failed phase 3. Will FDA approve anyway? Will FDA approve anyway? Biogen’s ALS drug has failed phase 3.

BioTech 365
AUGUST 27, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Agenus, with FDA approval in sight, showcases anti-PD-1 data.Agenus, with FDA approval in sight, showcases anti-PD-1 data ntaylor Fri, 08/27/2021 – 05:40 from FierceBiotech: Biotech [link].

BioTech 365
AUGUST 24, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: The FDA wants more data. Stealth is filing for approval anyway.The FDA wants more data.

BioPharma Reporter
JULY 7, 2023
The first and only approved anti-amyloid Alzheimer's Disease (AD) treatment shown to reduce the rate of disease progression and slow cognitive impairment has been granted approval by the FDA.

BioTech 365
AUGUST 26, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Sesen, still reeling from FDA rejection, pulls filing for EU approval of cancer drug.Sesen, still reeling from FDA rejection, pulls filing for EU approval of cancer drug … Continue reading →

BioTech 365
JANUARY 26, 2022
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Incyte pulls FDA filing for cancer drug approval over confirmatory study, following Gilead out of the exit.Incyte pulls FDA filing for cancer drug approval over confirmatory study, … Continue reading →

BioTech 365
FEBRUARY 17, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Amgen KRAS drug gets FDA priority review, teeing up August approval decision.Amgen KRAS drug gets FDA priority review, teeing up August approval decision ntaylor Wed, 02/17/2021 – … Continue reading →

Camargo
AUGUST 5, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. Approval of the Month: Repurposed Transplant Drug Approved Based Only on Real-World Evidence. FDA Inspections Ramping Up?

BioTech 365
OCTOBER 11, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Merck makes beeline to FDA, seeking approval for COVID-19 antiviral 10 days after reporting data.Merck makes beeline to FDA, seeking approval for COVID-19 antiviral 10 days after … Continue reading →

BioTech 365
JANUARY 14, 2022
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: FDA approval, global partnership on the agenda at Mithra as phase 3 menopause trials hit goals—with one blemish .FDA

BioTech 365
JUNE 24, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Lilly to file for accelerated FDA approval of Alzheimer’s drug after Aduhelm OK opens the floodgates.Lilly to file for accelerated FDA approval of Alzheimer’s drug after Aduhelm … Continue reading →

BioTech 365
APRIL 5, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Pfizer’s Vizimpro is among 3 FDA-approved drugs that combat COVID-19 in lung cells: report.Pfizer’s Vizimpro is among 3 FDA-approved drugs that combat COVID-19 in lung cells: report … Continue reading → (..)

BioTech 365
SEPTEMBER 23, 2020
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: FDA to set high bar for emergency COVID-19 vaccine approvals.FDA to set high bar for emergency COVID-19 vaccine approvals ntaylor Wed, 09/23/2020 – 08:50 from FierceBiotech: Biotech … Continue reading →

Camargo
DECEMBER 10, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. Voxzogo Approved as a Growth-Improvement Therapy for Children with Dwarfism. Most (119) of the participants were enrolled in an open-label extension.

Camargo
MAY 10, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA’s Accelerated Approval Program Comes Under Fire. It will be interesting to see if the FDA follows the ODAC recommendations.

Camargo
SEPTEMBER 8, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. Genus contested the FDA’s classification of its barium sulfate contrast imaging agent as a drug rather than a device.

Drug Discovery World
MARCH 1, 2024
News round-up for 26 Feb – 1 March by DDW Digital Content Editor Diana Spencer. The news highlights this week all focus on regulatory decisions, both positive and negative. The post This week in drug discovery (26 Feb – 1 March) appeared first on Drug Discovery World (DDW).

Camargo
JANUARY 14, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA Issues Advice on Combination Products. The guidance could also serve as a good regulatory primer for those new to combination products.

XTalks
FEBRUARY 26, 2024
The US Food and Drug Administration (FDA) has granted expanded approval to Genentech’s (part of Roche) Xolair (omalizumab) to help reduce allergic reactions to various foods after accidental exposure. The FDA said patients who take Xolair must continue to avoid foods they are allergic to. million children and 13.6

BioSpace
MARCH 22, 2021
The life sciences industry is a bustling beehive of activity. Here’s a quick compilation of some of today’s newsworthy announcements.

Camargo
MARCH 9, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA: Sponsors Seldom Disclose RTF Letters’ Existence or Contents and Often Ignore Agency Advice. The FDA found that fully 84.5%
Pharmaceutical Technology
DECEMBER 23, 2022
The US Food and Drug Administration (FDA) has granted approval for Gilead Sciences ’ Sunlenca (lenacapavir) plus other antiretroviral(s) (ARV) to treat human immunodeficiency virus type 1 (HIV-1) infection. Sunlenca is also said to be the first-ever capsid inhibitor-based HIV therapy to receive such approval.
STAT News
MARCH 8, 2023
The news came just days after the test’s maker, Lucira, filed for bankruptcy, blaming the FDA’s “protracted” approval process for its financial problems. Now the FDA has released a rare comment clarifying what happened during its authorization process. Read the rest…
Pharmaceutical Technology
MAY 26, 2023
After earning multimillion dollar revenues while being an authorised preferred treatment for Covid-19, the US Food and Drug Administration (FDA) has granted a full approval to Pfizer’s oral antiviral Paxlovid (nirmatrelvir + ritonavir). In response to the approval news, some pointed concerns about long-term safety.

World of DTC Marketing
JULY 13, 2021
SUMMARY: The FDA has many puzzled as to why they approve some drugs with questionable data and ask for more data on other drugs. The FDA approves drugs on the potential to save a life, the cost of the drugs is never considered in the process. Our healthcare system cannot continue to support marginally effective drugs.

Camargo
OCTOBER 14, 2020
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA Establishes Digital Health Center of Excellence. FDA Commissioner Stephen M. FDA Issues Guidances Dealing with ANDAs.

Camargo
NOVEMBER 15, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA Guidance Addresses Real-World Evidence Data Standards. FDA Publishes ICH Guidelines on Continuous Manufacturing. Emphasis in the original).

Camargo
APRIL 7, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. To be successful, such trials require out-of-the-box thinking and experience with the FDA. The FDA then rejected the NDA later in 2013.

Camargo
JUNE 4, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. Approval of the Month: Non-Opioid for Post-Surgical Pain Gets FDA Nod. Since two-thirds of the more than 50 million U.S.

World of DTC Marketing
NOVEMBER 10, 2020
SUMMARY : Pfizer’s 90 percent effectiveness is very impressive, but the rate is based on a relatively small number of people and the data has not been peer-reviewed but today headlines rule the news, not content. Pfizer’s results are promising and are coming at a time when the world is anxiously awaiting positive news.

Camargo
FEBRUARY 4, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. Approval of the Month: Previously Approved Oncology Drug Cleared for Rare Pediatric Indication.

Camargo
OCTOBER 14, 2021
Each month, Camargo’s “In the News” series highlights important changes and advancements in the regulatory and development space and explores how those changes could impact your program. FDA Discusses Focus Areas for PDUFA VII During Public Meeting. Improved predictability in Human Factors and User-Related Risk reviews.
BioPharma Reporter
NOVEMBER 7, 2022
The news comes two years after the drug received US Food and Drug Administration (FDA) accelerated approval. GSKâs Blenrep has proved unsuccessful in a Phase III study in multiple myeloma.

World of DTC Marketing
JULY 15, 2021
SUMMARY: The Committee on Oversight and Reform and the Committee on Energy and Commerce is investigating the approval process for Biogen’s new Alzheimer’s drug. Janet Woodcock acknowledged on Wednesday her agency may have misstepped in the handling of its polarizing approval of a new Alzheimer’s drug.

XTalks
JULY 10, 2023
Euronext Brussels (UCB), a biopharma company headquartered in Brussels, Belgium, recently announced that the US Food and Drug Administration (FDA) has granted approval for its drug, Rystiggo (rozanolixizumab), for the treatment of generalized myasthenia gravis (gMG). What Is Generalized Myasthenia Gravis (gMG)?
XTalks
JUNE 30, 2023
Bayer, a renowned German pharmaceutical company, has recently announced that its Ultravist 300 and 370 (iopromide) injection has won approval from the US Food and Drug Administration (FDA) for use in contrast-enhanced mammography (CEM). The FDA has not approved Iomeron.
Pharmaceutical Technology
JANUARY 30, 2023
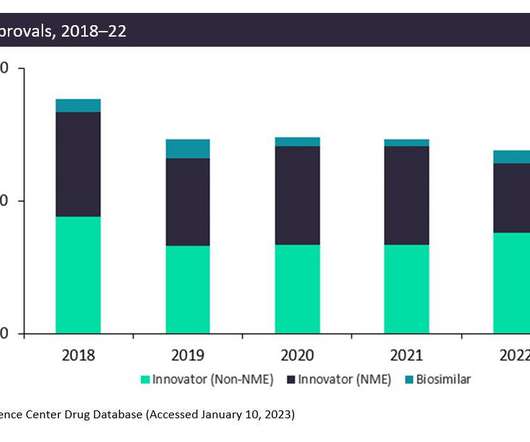
The FDA approved fewer innovative drugs, New Molecular Entities (NMEs), in 2022 than it did in 2021: only 42 drugs compared to 59 drugs. This is due to generally more stringent criteria on approvals in the wake of the Aduhelm scandal. However, non-NME and biosimilar approvals increased in 2022.

Pharmaceutical Technology
JANUARY 12, 2023
The US Food and Drug Administration (FDA) has approved AstraZeneca and Avillion’s Airsupra (albuterol/budesonide), to treat asthma. Airsupra has been approved for the as-needed treatment or prevention of bronchoconstriction and for reducing the exacerbations risk in asthma patients aged 18 years and above.

Drug Discovery World
MARCH 22, 2024
News round-up for 18-22 March by DDW Digital Content Editor Diana Spencer. This has been an interesting week for cell and gene therapies, with two landmark FDA approvals, two significant fundraising efforts and potentially ground-breaking study results in glioblastoma.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content