Food Allergy Awareness Week 2024: How can new treatments target an unmet need?
Pharmaceutical Technology
MAY 16, 2024
IgGenix is planning to initiate a clinical trial later this year investigating its peanut allergy monoclonal antibody IGXN001.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
MAY 16, 2024
IgGenix is planning to initiate a clinical trial later this year investigating its peanut allergy monoclonal antibody IGXN001.
AuroBlog - Aurous Healthcare Clinical Trials blog
APRIL 13, 2023
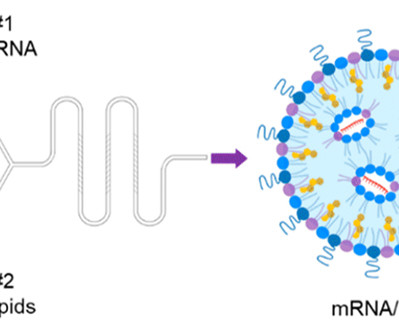
More treatments – and faster-acting treatments – for peanut allergies are urgently needed, and a new study outlines a promising drug technology based around a tiny nanoparticle just a few billionths of a meter in size. In tests on mice, the nanoparticle reversed peanut allergies and prevented them from developing in the first place.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
STAT News
JANUARY 18, 2023
The National Institute of Allergy and Infectious Diseases reported Wednesday that a Phase 3 clinical trial of a vaccine was stopped because the vaccine was ineffective at preventing HIV infection. Yet another experimental HIV vaccine has failed. Read the rest…

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Pharma Mirror
DECEMBER 1, 2021
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, sponsored a randomized, double-blind, placebo-controlled clinical trial that included 969. The post Emmes Announces Completion of Third ACTT Clinical Trial for COVID-19 appeared first on Pharma Mirror Magazine.

BioTech 365
OCTOBER 29, 2021
Global Peanut Allergy Clinical Trials Review, H2 2021 Report – ResearchAndMarkets.com Global Peanut Allergy Clinical Trials Review, H2 2021 Report – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “Peanut Allergy – Global Clinical Trials Review, H2, 2021” clinical trials has been added to ResearchAndMarkets.com’s (..)
Antidote
SEPTEMBER 1, 2023
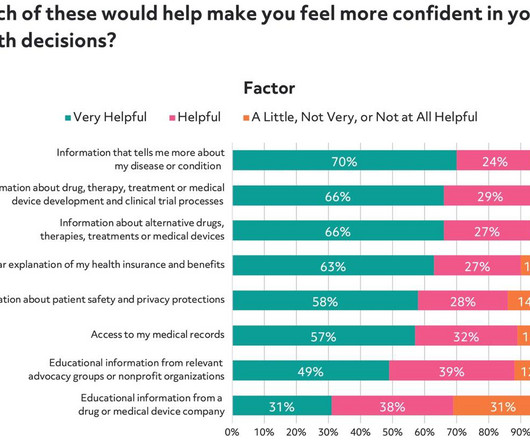
As part of our efforts to better understand patients and their motivations to participate in clinical trials, Antidote and SCORR Marketing partnered on a survey to gain a deeper understanding of how patients think about the medical research process.

Trialfacts
JANUARY 23, 2024
More Study Details About the Study A peanut allergy can be severe and life-threatening if not treated promptly. Peanut allergies are one of the most common food allergies in children in the USA, with more than 2% of children being affected. Participants will receive a new potential treatment for their peanut allergy at no cost.

pharmaphorum
DECEMBER 23, 2021
Children in England with peanut allergies could be among the first in Europe to get access to Aimmune’s oral immunotherapy Palforzia, after the drugmaker agreed a supply deal with NHS England. In a second phase, doses are then taken at home and raised gradually over at least 22 weeks, with a clinic visit required at each dose increase.

Medical Xpress
FEBRUARY 28, 2023
A four-year clinical trial led by Edwin Kim, MD, at the UNC School of Medicine, has found that an increased dosage of a unique type of peanut allergy immunotherapy continues to show promise for children.
NY Times
DECEMBER 11, 2020
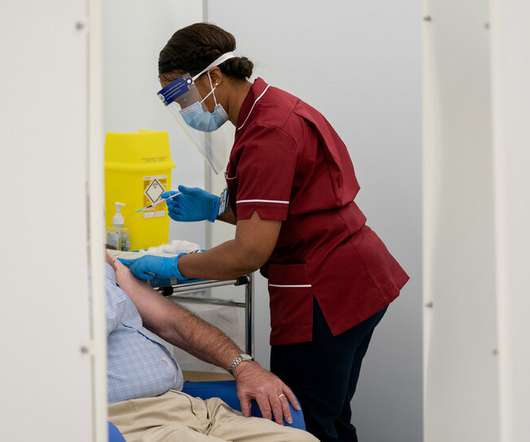
British health officials recommended that people with severe allergy reactions not be given the vaccine. Such reactions to vaccines are rare, even in people who have allergies to food or bee stings.

JAMA Internal Medicine
JULY 16, 2023
This randomized clinical trial aims to determine whether a direct oral penicillin challenge is noninferior to the standard of care of penicillin skin testing followed by an oral challenge in patients with a low-risk penicillin allergy.

Scienmag
JANUARY 20, 2022
A clinical trial funded by the National Institutes of Health has found that giving peanut oral immunotherapy to highly peanut-allergic children ages 1 to 3 years safely desensitized most of them to peanut and induced remission of peanut allergy in one-fifth. The immunotherapy consisted of a daily oral dose of peanut flour for 2.5
Scienmag
MAY 6, 2022
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has launched an early-stage clinical trial to evaluate an investigational preventative vaccine for Epstein-Barr virus (EBV).

Antidote
NOVEMBER 20, 2023
In both 2018 and 2023, Antidote partnered with SCORR marketing to conduct surveys intended to increase the understanding of patients’ perceptions of clinical trials. Our 2018 survey collected data from nearly 4,000 individuals to gain insight into what matters most when considering a clinical trial.

Clinical Trial Podcast
SEPTEMBER 26, 2022
To answer these two questions, regulatory authorities around the world require medical product manufacturers to conduct Phase I, also known as early phase clinical trials. The post Complexity in Early Phase Clinical Trials with Dr. The post Complexity in Early Phase Clinical Trials with Dr. Oren Cohen?

Drug Discovery World
OCTOBER 13, 2022
New research has shown that standard cat allergy treatment can be enhanced to make it more effective and faster acting, and the benefits last for a year after treatment ends. The findings of the study, supported by the National Institutes of Health, were published in the Journal of Allergy and Clinical Immunology. .
Scienmag
NOVEMBER 18, 2021
Louis and Massachusetts General Hospital/Harvard Medical School in Boston have received a seven-year, $22 million grant from the National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health (NIH) to help lead a multicenter clinical trial evaluating whether a novel immunosuppressant can […].

Drug Discovery World
JULY 19, 2022
An early-stage clinical trial has been launched in the US investigating a vaccine against Nipah virus infection. . The clinical trial has been launched by the National Institute of Allergy and Infectious Diseases (NIAID) to evaluate an investigational vaccine to prevent infection with Nipah virus.
Drug Discovery World
MAY 18, 2023
A clinical trial of an experimental universal influenza vaccine developed by researchers at the National Institute of Allergy and Infectious Diseases’ (NIAID) Vaccine Research Center (VRC) has begun enrolling volunteers at Duke University in Durham, North Carolina.

The Pharma Data
JANUARY 7, 2021
This clearance is a pivotal regulatory milestone that further supports Camallergy’s leadership and capabilities in the field of peanut allergy immunotherapy with the only cGMP-certified manufacturing facility in Europe producing peanut oral immunotherapy,” said Sherden Timmins, Chief Technology Officer. 8, 2021 07:00 UTC. About Camallergy.

Drug Discovery World
FEBRUARY 26, 2024
The US Food and Drug Administration (FDA) has approved monoclonal antibody Xolair (omalizumab) for immunoglobulin E-mediated food allergy in some adults and children one year or older for the reduction of allergic reactions (Type I).

pharmaphorum
AUGUST 5, 2020
DBV Technologies’ long and tortuous path to an FDA verdict on its Viaskin Peanut allergy shot has led to another dead end. DBV says it plans to request a meeting with the FDA to discuss its comments in the CRL as well, as that may be needed in another clinical trial to support another refiling. “We

XTalks
NOVEMBER 6, 2020
Canadian clinical-stage biotech company Symvivo Corporation has developed an oral COVID-19 vaccine that entered clinical trials this week. The first healthy volunteer was dosed with the vaccine in Australia as part of the bacTRL-Spike COVID-19 Phase I clinical trial. COVID-19 Clinical Trials.

Medical Xpress
DECEMBER 22, 2022
The study analyzed data from two phase 3 clinical trials and involved an international group of researchers, including those from Children's Hospital of Philadelphia (CHOP).
Scienmag
APRIL 21, 2021
Credit: NIAID A Phase 2/3 trial to evaluate a new fully-human polyclonal antibody therapeutic targeted to SARS-CoV-2, called SAB-185, has begun enrolling non-hospitalized people with mild or moderate cases of COVID-19. The therapeutic […].

Rethinking Clinical Trials
JANUARY 24, 2023
INSPIRE is supported within the NIH Pragmatic Trials Collaboratory by a grant from the National Institute of Allergy and Infectious Diseases (NIAID). The post January 24, 2023: INSPIRE Intervention Goes Live, Will Use Predictive Algorithm to Reduce Unnecessary Antibiotic Prescribing appeared first on Rethinking Clinical Trials.
BioPharma Reporter
MAY 9, 2022
Last week the US National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, launched a clinical trial to investigate a preventative vaccine for Epstein-Barr virus (EBV). Itâs part of growing efforts to create a vaccine against the virus.
Medical Xpress
JANUARY 30, 2023
A newly published paper in The Lancet shows that an experimental vaccine against Marburg virus (MARV) was safe and induced an immune response in a small, first-in-human clinical trial.

Rethinking Clinical Trials
NOVEMBER 29, 2022
The NIH Pragmatic Trials Collaboratory is excited to welcome the INSPIRE Demonstration Project to its portfolio of innovative, large-scale pragmatic clinical trials embedded in healthcare systems. Shruti Gohil. Dr. Susan Huang. Dr. Richard Platt. Huang and Platt are experienced investigators in the NIH Collaboratory.

Scienmag
MAY 25, 2021
May 25, 2021 — Critical Path Institute (C-Path) today announced its schedule for the Design of Clinical Trials in New-Onset Type 1 Diabetes: Regulatory Considerations for Drug Development Workshop, to be held virtually June 15-16. Together […].

Medical Xpress
MARCH 6, 2023
A clinical trial testing a freeze-dried, temperature-stable experimental tuberculosis (TB) vaccine in healthy adults found that it was safe and stimulated both antibodies and responses from the cellular arm of the immune system. A non-temperature stable form of the candidate previously had been tested in several clinical trials.
Scienmag
FEBRUARY 26, 2022
Peanut allergies are hard to treat and can cause fatal or near-fatal anaphylactic reactions. People usually don’t outgrow peanut allergy, requiring lifelong scrutiny of food labels to avoid even small traces of peanut.

Scienmag
MAY 21, 2021
The findings of a clinical trial by Trinity College Dublin researchers of treatment for atopic dermatitis have been published today in The Lancet journal (Friday, 21st May, 2021). Results of the clinical trial at […].
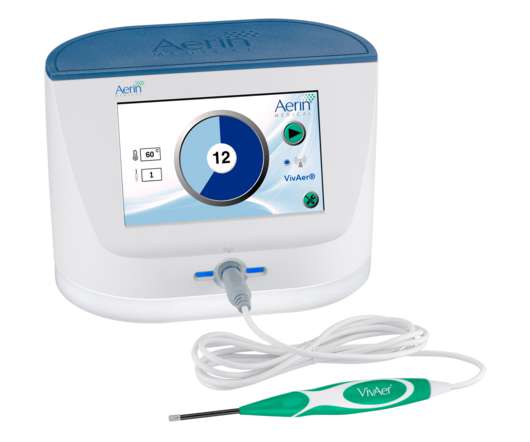
XTalks
JULY 6, 2022
Their 48-month clinical study has revealed that VivAer can substantially ease the symptoms of NVC and improve nasal breathing. The four-year results from the VivAer clinical trial were published in the International Forum of Allergy & Rhinology.
Pharmaceutical Technology
OCTOBER 6, 2022
from the US National Institutes of Health (NIH) unit National Institute of Allergy and Infectious Diseases (NIAID) to develop a prophylactic intranasal vaccine against Neisseria gonorrhoeae (NG). This trial will evaluate the safety of the vaccine and generate efficacy data. Intravacc has received a contract worth $14.6m

Rethinking Clinical Trials
JUNE 14, 2023
The project is supported within the NIH Collaboratory under an award from the National Institute of Allergy and Infectious Diseases. The post June 14, 2023: IMPACt-LBP and INSPIRE Have Updated Study Snapshots, Ethics and Regulatory Documentation appeared first on Rethinking Clinical Trials.
Pharmaceutical Technology
SEPTEMBER 16, 2022
The latest conditional recommendation is based on the final data from the SOLIDARITY clinical trial, which was sponsored by the WHO. These results are in line with those obtained from the placebo-controlled, double-blind ACTT-1 trial of the National Institute of Allergy and Infectious Diseases.
Pharmaceutical Technology
JUNE 16, 2023
The Phase IIa trial (NCT05734040) in Australia will see a potential 500 volunteers given OVX836 in combination with quadrivalent influenza vaccines (QIVs). The developer has tested OVX836 in four completed clinical trials.

The Pharma Data
MARCH 31, 2022
A Phase 2 clinical trial evaluating various additional COVID-19 booster shots has begun enrolling adult participants in the United States. Vaccine manufacturers have previously studied some variant vaccine candidates and are currently conducting clinical trials of Omicron-specific vaccines.
Drug Discovery World
MAY 1, 2024
A single injection of an experimental monoclonal antibody called L9LS was 77% effective at preventing malaria infection in children in Mali, according to the results of a mid-stage clinical trial. It was developed by scientists at the National Institutes of Health (NIH). “A
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content