Avrobio halts gene therapy research and considers a sale
Bio Pharma Dive
JULY 12, 2023
The decision to explore “strategic alternatives” comes about six weeks after the company sold its most advanced treatment to Novartis for nearly $90 million.

Bio Pharma Dive
JULY 12, 2023
The decision to explore “strategic alternatives” comes about six weeks after the company sold its most advanced treatment to Novartis for nearly $90 million.
AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 12, 2023
Researchers in Japan found a connection between tooth loss, gum disease, and shrinkage in a region of the brain involved in memory and Alzheimer’s disease called the hippocampus.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
JULY 12, 2023
The European drug regulator says it now has 150 reports of possible cases involving self-injury and suicidal thoughts among people taking GLP-1 medicines like Ozempic.

Pharmaceutical Technology
JULY 12, 2023
Innovate UK’s New Cancer Therapeutics programme awarded the grant to the Imperial College spinout to support pre-clinical development.

Bio Pharma Dive
JULY 12, 2023
Backed by F-Prime Capital and Sofinnova Partners, the company claims to have technology that could allow it replace eye cells destroyed in age-related and inherited ocular conditions.

Pharmaceutical Technology
JULY 12, 2023
The EMA is reviewing data on the potential effects of high-profile GLP-1RAs in causing suicidal thoughts and thoughts of self-harm.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharmaceutical Technology
JULY 12, 2023
To that end, the company presented positive top-line Phase III results for its once-daily stimulant for ADHD.

Rethinking Clinical Trials
JULY 12, 2023
Complete materials are now available from the NIH Pragmatic Trials Collaboratory’s recent workshop, “Driving Tomorrow’s Outcomes Through Clinical Research in Real-World Settings. “ The 2-day workshop, held in June at the 2023 AcademyHealth Annual Research Meeting in Seattle, introduced concepts in the design, conduct, and implementation of pragmatic clinical trials embedded in healthcare systems, with a focus on methods relevant to health services researchers.

Bio Pharma Dive
JULY 12, 2023
The European Commission called Illumina’s closing of the deal without its approval an “unprecedented” move that undermines its system for regulating the competitive landscape.

Rethinking Clinical Trials
JULY 12, 2023
Dr. Christopher Granger and Dr. Neha Pagidipati In this Friday’s PCT Grand Rounds, Christopher Granger and Neha Pagidipati of Duke University will present “Lessons From the COORDINATE-Diabetes Trial.” The Grand Rounds session will be held on Friday, July 14, 2023, at 1:00 pm eastern. The Coordinating Cardiology Clinics Randomized Trial of Interventions to Improve Outcomes (COORDINATE)–Diabetes was a cluster randomized trial assessing the effect of a multifaceted intervention of

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Pharmaceutical Technology
JULY 12, 2023
The US FDA has awarded fast track designation to clinical-stage biotechnology firm Synlogic’s labafenogene marselecobac for phenylketonuria.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 12, 2023
Indian pharmaceutical & nutraceutical companies are increasingly recognizing the advantages of outsourcing various aspects of their operations, intensifying their focus on this strategic approach to offer a competitive cost structure, which makes it an attractive destination for outsourcing, said Vivek Srivastava, senior vice president, innovation, business strategy and scientific affairs & Yuvraj Datta, director, manufacturing […]

Pharmaceutical Technology
JULY 12, 2023
Takeda has withdrawn the biologics licence application (BLA) submitted to the US FDA for TAK-003, a tetravalent dengue vaccine candidate.

BioSpace
JULY 12, 2023
Following the regulator’s request for more data beyond the scope of its Phase III TIDES trial, Takeda decided to voluntarily withdraw the Biologics License Application for its TAK-003 dengue vaccine.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharmaceutical Technology
JULY 12, 2023
Canada's Solve FSHD has awarded four grants to augment the understanding of facioscapulohumeral muscular dystrophy (FSHD).

BioPharma Reporter
JULY 12, 2023
Lisa Moneymaker is full of energy, enthusiasm, and wisdom. She is well-known on the pharma circuit and itâs easy to see why. She has been chief technology officer and chief product officer at Saama since October last year and feels passionately about women in science and women in general â and although her journey was without many challenges, she appreciates that is not the case for everyone.
Pharmaceutical Technology
JULY 12, 2023
Septerna has raised $150m in a Series B funding round to develop its pipeline comprising new therapies that act on GPCRs.
Fierce Pharma
JULY 12, 2023
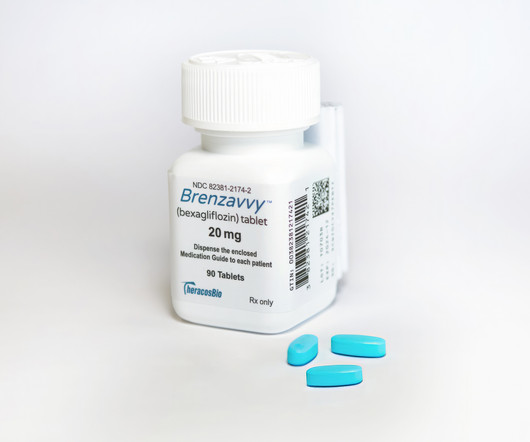
As another SGLT2 diabetes drug hits the market, the obvious question is: How will it find a way to compete against formidable blockbusters Farxiga and Jardiance? | TheracosBio and Mark Cuban's online distributor Cost Plus Drug Company have partnered to provide newly approved SGLT2 inhibitor Brenzavvy at a major discount to competitors in the class, including AstraZeneca's Farxiga and Eli Lilly and Boehringer Ingelheim's Jardiance.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Pharmaceutical Technology
JULY 12, 2023
Crossbow Therapeutics has raised Series A funds worth $80m to develop a new class of antibody treatments for cancer.

BioSpace
JULY 12, 2023
For closing its $8 billion acquisition of Grail without regulatory approval, the European Union has slapped Illumina with a record $476 million fine—the maximum sanction allowed under the EU’s rules.

Pharmaceutical Technology
JULY 12, 2023
The NHS is set to expand a project to tackle antimicrobial resistance, following the success of a pilot.

BioSpace
JULY 12, 2023
As the market recovers from the effects of the COVID-19 pandemic, employers are beginning to regain some of the power they lost in recruiting, forcing candidates to adjust their asks.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Pharmaceutical Technology
JULY 12, 2023
The SkyePay system allows patients to receive payments for participating in clinical trials.
Outsourcing Pharma
JULY 12, 2023
Acasti Pharma, a late-stage biopharma company with drug candidates addressing rare and orphan diseases, has selected WuXi Clinical Development to conduct its STRIVE-ON phase 3 safety trial for GTX-104, a novel injectable nimodipine formulation for intravenous infusion (IV) that addresses high, unmet medical needs for aneurysmal subarachnoid haemorrhage (aSAH).

Pharma Marketing Network
JULY 12, 2023
The internet is a vast resource of information on health and wellness. However, not all of this information is created equal. Some of it is accurate and reliable, while other information may be misleading or even harmful. This is especially true when it comes to pharmaceutical-sponsored health information. Pharmaceutical companies have a vested interest in promoting their products, so it’s important to be critical of any information they put out.
Outsourcing Pharma
JULY 12, 2023
Ora, a clinical research organization (CRO) for ophthalmology pharmaceutical and device development, has partnered the North West London Clinical Trials Alliance, to improve the quality and access to clinical research in the region.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
BioSpace
JULY 12, 2023
The FDA recently approved the first cellular therapy for Type 1 diabetes and others may not be far behind. But experts say challenges still exist to the widespread application of these treatments.

Fierce Pharma
JULY 12, 2023
Merck is bringing more heat in its battle against drug price negotiation provisions in the Inflation Reduction Act (IRA), filing Tuesday in federal court in Washington, D.C. | Merck is bringing more heat in its battle against drug price negotiation provisions in the Inflation Reduction Act (IRA), filing Tuesday for a decision without a trial. In its request for a summary judgment, Merck reiterated its claim that the drug price negotiation program violates the First and Fifth Amendments of the Co

Pharma Marketing Network
JULY 12, 2023
The pharmaceutical industry is facing a number of challenges, including increasing competition, rising costs, and changing regulations. In order to succeed in this challenging environment, pharmaceutical companies need to adopt a patient-centric approach to marketing. Patient-centric pharmaceutical marketing is based on the understanding that patients are more than just consumers of drugs.

Outsourcing Pharma
JULY 12, 2023
2San, a supplier of self-diagnostic tests, has partnered with Proov to distribute their proprietary at-home fertility tests in select Hy-Vee stores across eight states in the Midwest.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content