European Clinical Supply Planning: Balancing Cost, Flexibility and Time

Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.

When a CRO is bidding on a project where clinical supplies will be one of the aspects to manage on behalf of the client via a partner, leveraging the expertise of a chosen clinical supply partner can be a valuable resource in demonstrating the CRO’s understanding of and ability to deliver upon critical drug-supply related aspects of the project, and ability to hit key milestones such as FPI for their client.

This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

The sourcing of commercial drugs for use in clinical trials either as a comparator, rescue or co-medication can be a complex process. Sponsors can face a number of challenges when trying to obtain these critical supplies for their studies, including addressing product lead times, availability, expiry limitations, safety and cost, among others. Choosing the right partner with global sourcing, regulatory, quality and clinical packaging expertise can provide the necessary guidance to help sponsors
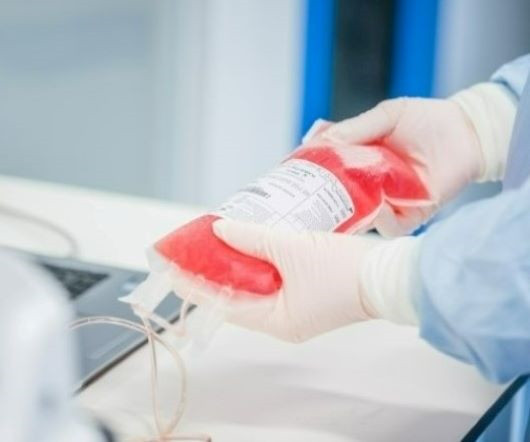
As demand for advanced therapies increases, so does the need for more specialized supply chain support, as these products have strict transportation and handling requirements. Deviations from the intended supply chain can delay timelines and affect these high value products, which can be detrimental to budgets, but more importantly can have dire effects on the patients whose lives depend on receiving these critical doses safely and on time.

Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.

Clinical development organizations face a wide array of challenges when it comes to data, many of which can impact the operational effectiveness of their clinical trials. In this whitepaper, experts from Revvity Signals explore how solutions like TIBCO® Spotfire® enable better, more streamlined studies. The whitepaper also features a success story from Ambrx, a leading biopharmaceutical company, detailing how it has leveraged Spotfire to tackle data quality and collaboration challenges in clinic

There’s been a rapid shift towards decentralization in clinical trials & it’s clear why. The potential for reaching a larger pool of recruits is possible when sponsors can bring more trial activities to the patient. Tele visits, digital consent, new monitoring sensors, & direct-to-patient supply are virtual tools that existed before the pandemic, but now there’s swift adoption of these methods because they’ve been proven to help launch & complete trials more effectively.

White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

FourKites
A research study conducted by The Journal of Commerce and FourKites surveyed hundreds of international shippers, exploring how their usage of global supply chain visibility technology has evolved since the onset of global disruptions caused by COVID-19. For international shippers, ocean freight visibility has evolved from optional to essential and satisfaction with visibility varies greatly depending on how it is obtained and delivered.

Aggregage
New vaccine mandates and testing policies will affect employers with more than 100 workers. Get Paycor’s free, customizable vaccination policy template to communicate critical details and new requirements to your employees. Get Paycor’s Template today!

Let's personalize your content