Researchers advance blood tests’ ability to detect cancer earlier
Pharma Times
JANUARY 23, 2024
The advancement could allow earlier detection of cancer in the blood and better treatment
Pharma Times
JANUARY 23, 2024
The advancement could allow earlier detection of cancer in the blood and better treatment

Bio Pharma Dive
JANUARY 23, 2024
A black box warning for secondary T cell malignancies should be added to the labeling of all six approved CAR-T therapies, the agency said.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JANUARY 23, 2024
The UK MHRA has introduced further restrictions on the usage of fluoroquinolone antibiotics, strengthening the previous regulations.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 23, 2024
The National Institutes of Pharmaceutical Education and Research (NIPERs) can consider their own in-house scientists for the new faculty positions or recruit pharmaceutical industry professionals for the Professor of Practice positions in the absence of suitable applications for faculty positions, recommends a study by the Department of Pharmaceuticals (DoP).
Pharmaceutical Technology
JANUARY 23, 2024
Genentech will utilise GenEdit’s non-viral polymer nanoparticles for gene therapy delivery in autoimmune diseases.
AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 23, 2024
Wegovy, Ozempic and Mounjaro are weight loss and diabetes drugs that have made quite a splash in health news. They target regulatory pathways involved in both obesity and diabetes and are widely considered breakthroughs for weight loss and blood sugar control. [link] But do these drugs point toward a root cause of metabolic disease?
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Rethinking Clinical Trials
JANUARY 23, 2024
Ethics and regulatory onboarding documentation is now available for RAMP, one of the NIH Pragmatic Trials Collaboratory's newest pragmatic clinical trials. The documents include meeting minutes and supplementary materials summarizing recent discussions of ethics and regulatory issues associated with the study. The consultation took place by video conference and included representation from the study's principal investigator and study team, members of the NIH Collaboratory's Ethics and Regulatory
Bio Pharma Dive
JANUARY 23, 2024
J&J and Bristol Myers joined the latest funding round for the startup, which was led by the newly launched Mirae Asset Capital Life Science.

Pharmaceutical Technology
JANUARY 23, 2024
The company ceased activity for its CKD program to sharpen its clinical focus in the Phase II/III program for notalgia paresthetica.

pharmaphorum
JANUARY 23, 2024
Bringing clinical trials to patients has become easier with the advent of digital technologies and decentralised clinical trials (DCTs). Learn how the hybrid approach combining traditional and digital methods can benefit patients and advance medical research.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Pharmaceutical Technology
JANUARY 23, 2024
Inhalation Sciences has entered into a collaboration with Germany-based Actarmo Medical for respiratory drug development services.
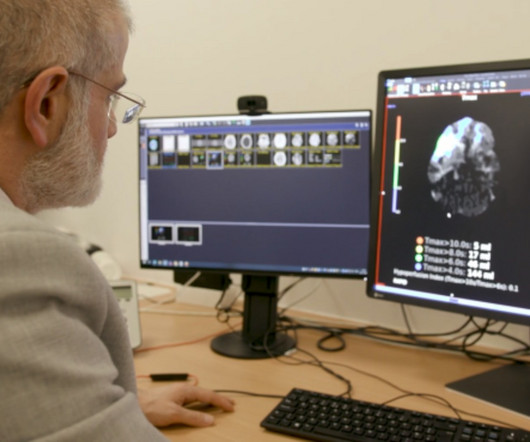
pharmaphorum
JANUARY 23, 2024
Two artificial intelligence-powered software tools – e-Stroke and RapidAI – have been recommended for use in the NHS to help detect stroke from CT brain scans by NICE. The health technology assessment (HTA) agency said there is some evidence that people admitted to hospital with a suspected had faster or better access to treatment after using the software, although for now it is unclear whether that was a direct consequence of the digital technology.

Pharmaceutical Technology
JANUARY 23, 2024
Sanofi has signed a definitive agreement for the acquisition of all outstanding shares of Inhibrx for an equity value of $1.7bn.
Fierce Pharma
JANUARY 23, 2024
While sales for Johnson & Johnson’s Darzalex continue to boom—as the company reported in its quarterly earnings presentation on Tuesday—the pharma major got more good news about its blockbuster | An appeal arbitrator has rejected Genmab’s claim that it is due additional milestone and royalty payments from Johnson & Johnson for Darzalex. The Danish biotech was hoping to overturn a 2-1 ruling in a second arbitration between the companies over Darzalex royalties.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
Pharmaceutical Technology
JANUARY 23, 2024
Calluna has four monoclonal antibody candidates in the pipeline under development for inflammatory and fibrotic diseases.

Fierce Pharma
JANUARY 23, 2024
Early evidence showing Carvykti’s potential to prolong patients’ lives in an earlier multiple myeloma setting didn’t save Johnson & Johnson and Legend Biotech’s CAR-T medicine from a public int | Early evidence showing the drug's potential to prolong patients' lives didn't save Johnson & Johnson and Legend Biotech's Carvykti from a public interrogation by the FDA.

pharmaphorum
JANUARY 23, 2024
Sanofi agrees $2.2bn deal to buy California biotech Inhibrx and its long-acting therapy for rare disease AATD, which causes life-threatening emphysema.

Fierce Pharma
JANUARY 23, 2024
After failing to resolve a mountain of talcum-powder lawsuits through two unsuccessful bankruptcy attempts, Johnson & Johnson has agreed to a $700 million settlement that would free the company | After failing to resolve a mountain of talcum-powder lawsuits through two unsuccessful bankruptcy attempts, Johnson & Johnson has agreed to a $700 million settlement that would free the company from some consumer protection claims—and a small part of the talc litigation that it faces.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
pharmaphorum
JANUARY 23, 2024
Study finds evidence that an AlZpath immunoassay for phosphorylated tau could deliver an effective blood test for Alzheimer's, paving the way for screening and early treatment
Drug Discovery World
JANUARY 23, 2024
The world’s most comprehensive database for understanding microbial distribution and function in the ocean has been released, opening up new opportunities for drug discovery. Scientists at the King Abdullah University of Science and Technology (KAUST) in Thuwal, Saudi Arabia used the KAUST Metagenomic Analysis Platform (KMAP) to analyse massive amounts of sequencing data to release Global Ocean Gene Catalog 1.0.

Outsourcing Pharma
JANUARY 23, 2024
Yesterday (January 22), Saama announced the release of several âgroundbreakingâ features within its AI-driven platform, including generative AI (GenAI) chat and interactive review listings (IRL), that it says create a workflow-driven environment for cross-functional collaboration.

pharmaphorum
JANUARY 23, 2024
Eli Lilly’s gene therapy for otoferlin-related deafness has restored the hearing of the first child enrolled in its ongoing phase 1/2 trial

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Outsourcing Pharma
JANUARY 23, 2024
Manufacturer for the pharma and life sciences industry, Gerresheimer, is investing more than $88 million to expand its operations in Peachtree City, Georgia.

pharmaphorum
JANUARY 23, 2024
In this new episode of the pharmaphorum podcast, web editor Nicole Raleigh welcomes guest Cem Zorluhar, CEO of Er-Kim Pharmaceuticals, for a conversation on antimicrobial resistance (AMR) and what the biotech industry should be focussing on in order to address this critical issue outside of the West.

Pharmaceutical Commerce
JANUARY 23, 2024
Study describes how a game theoretic model can successfully analyze how adoption of blockchain technology can reveal quality information.

JAMA Internal Medicine
JANUARY 23, 2024
The US Supreme Court’s decision in the Dobbs v Jackson Women’s Health Organization case allows individual states to determine access to abortion. Since the decision, 14 states have prohibited abortion at any gestational age.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Outsourcing Pharma
JANUARY 23, 2024
Sano Genetics says it is developing software that will enable âthe precision medicine revolutionâ and has raised $11.4 million in new funding.

pharmaphorum
JANUARY 23, 2024
Ukraine-based digital pharma marketing specialist Viseven gets private equity investment from Horizon Capital
Pharma Times
JANUARY 23, 2024
Autosomal dominant polycystic kidney disease affects more than 12 million people globally

JAMA Internal Medicine
JANUARY 23, 2024
This cross-sectional study estimates the incidence of rape-related pregnancies in US states with abortion bans.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content