Curavit launches HEOR services for digital therapeutic trials
Pharmaceutical Technology
NOVEMBER 1, 2023
Curavit will incorporate HEOR services into trials to elucidate the real-world financial impact of a new product.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 heor
heor 
Pharmaceutical Technology
NOVEMBER 1, 2023
Curavit will incorporate HEOR services into trials to elucidate the real-world financial impact of a new product.

Outsourcing Pharma
NOVEMBER 6, 2023
With rising healthcare costs and âless understood digital therapeuticsâ Curavit has launched a health economics and outcomes research (HEOR) practice.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

XTalks
OCTOBER 4, 2023
Health economics and outcomes research (HEOR) and market access strategies play a critical role in ensuring that newly approved therapies are made available to patients. By harmonizing HEOR findings with targeted market access strategies, the path is paved for innovative treatments to achieve broader availability, benefiting patients. “We

Drug Channels
APRIL 29, 2022
Chris and Carolyn discuss the insights that health economics and outcomes research (HEOR) professionals bring to commercial teams. For more on how MMIT and Panalgo help pharma manufacturers leverage HEOR and real-world data for commercial strategies, click here to learn about the Patient Access Analytics tool.

pharmaphorum
MARCH 3, 2022
March 3, 2022, London, UK — Fishawack Health (FH), a leading commercialization partner for biopharmaceutical, medical technology, and wellness companies, today welcomes the US-based health economics and outcomes research (HEOR) consultancy Policy Analysis Inc. PAI) as part of its expanding Value, Evidence, and Access (VEA) capability.

BioSpace
MARCH 26, 2024
Health economics and outcomes research (HEOR) is a critical but sometimes overlooked part of drug development. Decentralized trials now make it easy.

Outsourcing Pharma
MARCH 13, 2024
In the dynamic world of clinical trials, where science intersects with technology and innovation, companies like Curavit are spearheading important advancements.

pharmaphorum
FEBRUARY 19, 2021
Ashfield Advisory , part of UDG Healthcare plc, today announced the acquisition of PHMR Limited, a Market Access Consultancy and specialist in Health Economics and Outcomes Research (HEOR). The post UDG Healthcare acquires market access, HEOR specialist, PHMR Limited as Ashfield Advisory appeared first on.

pharmaphorum
APRIL 12, 2021
UK comms agency Fishawack Health has acquired the health economics outcomes research (HEOR) consultancy PRMA Consulting. The company said that the acquisition will allow it to offer a HEOR consultancy service that spans the globe.
Cloudbyz
JUNE 7, 2023
Health Economics and Outcomes Research (HEOR) RWE is instrumental in Health Economics and Outcomes Research (HEOR), offering insights into the economic impact, including cost-effectiveness, and patient-reported outcomes of medical devices.

pharmaphorum
NOVEMBER 22, 2020
Formed in 2011 by David Rowley and chief operating officer Sandy Royden, OPEN Health has pursued several M&A deals over the years, with the most recent including a merger with med comms provider Peloton in 2018 – following OPEN’s acquisition with Amulet Capital Partners – and one last year with HEOR and market access firm Pharmerit.

pharmaphorum
DECEMBER 6, 2022
Work should also be undertaken on making product studies generalisable between national jurisdictions, the design of real-world evidence (RWE), healthcare evaluation outcomes research (HEOR) and clinical workflow pilot studies, and how DTx can be adapted for different languages and cultures.

Drug Channels
AUGUST 22, 2022
Reimbursement Strategies and Contracting Value-Based Contracting Frameworks and Considerations HEOR, RWE and Their Roles in Reimbursement Rebates and Reimbursement—Breaking Down the Pricing Strategy And more! Exclusive Offer: View the agenda and register today.

pharmaphorum
AUGUST 17, 2021
AXON is expecting to make several appointments in the US in the coming months to support existing clients and potential new opportunities, as we expand our work in medical affairs, clinical trial services, HEOR and real-world evidence, and public relations.

Clinical Trial Podcast
FEBRUARY 23, 2021
Health Economics and Outcomes Research (HEOR). This is where HEOR comes into play. This is where HEOR comes into play. A HEOR team helps create a body of evidence that the new drug or device has health benefits such as improved quality of life. HEOR has an in-depth understanding of country-specific requirements.

pharmaphorum
MARCH 30, 2021
So HEOR and market access must be involved up front, but she advocates a wider collaborative approach which also includes external perspectives from payers, policy makers and influential groups such as ISPOR. We had a lot of different perspectives from people in HEOR, market access, regulatory and marketing.

pharmaphorum
JUNE 8, 2022
Combining our skills and experiences across HEOR, market access, and policy will allow us to partner with clients to provide a deeper set of integrated capabilities not offered by other firms,” says David Sykes, President of Policy, Access, Value, and Evidence at FH. . We are delighted to welcome the Avalere team to FH.

XTalks
NOVEMBER 22, 2021
AppliedVR says it will continue testing the VR system to demonstrate both its clinical efficacy and cost-effectiveness to treat pain — for the latter, it plans to complete multiple health economics and outcomes (HEOR) studies with commercial payers.
pharmaphorum
DECEMBER 15, 2022
Thousands more UK patients with idiopathic pulmonary fibrosis (IPF) will now be eligible for treatment with Boehringer Ingelheim’s Ofev, following new guidance from health technology assessment agency NICE.

pharmaphorum
NOVEMBER 16, 2022
NICE has recommended that three COVID-19 therapies can continue to be used to treat patients – Pfizer’s Paxlovid, Roche’s RoActemra and Eli Lilly’s Olumiant – but turned down another five in draft guidance published this week.

pharmaphorum
SEPTEMBER 6, 2022
NICE has said it is unable to recommend NHS use of AstraZeneca’s Lynparza for some patients with prostate cancer, after failing to reach an agreement on price with the drugmaker.

pharmaphorum
JUNE 20, 2022
Thousands more people with breast cancer in England look set to be eligible for routine treatment with Eli Lilly’s Verzenios, after new guidance from NICE backed use of the drug after surgery for early-stage tumours.

pharmaphorum
SEPTEMBER 22, 2022
BeiGene’s BTK inhibitor Brukinsa (zanubrutinib) has become the first drug in the class to be recommended for routine NHS use in England and Wales to treat Waldenström macroglobulinaemia (WM) – a rare form of non-Hodgkin lymphoma (NHL).
pharmaphorum
JULY 14, 2022
Cost-effectiveness watchdog NICE has recommended routine NHS use of two new therapies for women with advanced breast cancer – Gilead’s Trodelvy and Novartis’ Piqray – in England. .

pharmaphorum
NOVEMBER 25, 2022
The European Commission has provided around €7 million ($7.2 million) in funding to a new project that aims to boost the ability of hospitals within the EU to collect real-world data (RWD) to gauge the effectiveness of novel cancer therapies.

pharmaphorum
AUGUST 19, 2021
Novartis’ Rydapt has become the first and only licensed treatment for rare and life-threatening blood disorder systemic mastocytosis (SM) to be cleared for routine NHS use, after getting a green light from NICE.
pharmaphorum
JULY 15, 2022
Health technology assessment (HTA) agency NICE has finalised its guidance on Amarin’s Vazkepa, clearing the path for GPs to start prescribing the drug in up to 425,000 NHS patients at high cardiovascular risk because of raised triglyceride levels. mmol/litre or above) in their blood who are already taking statins.
pharmaphorum
FEBRUARY 8, 2022
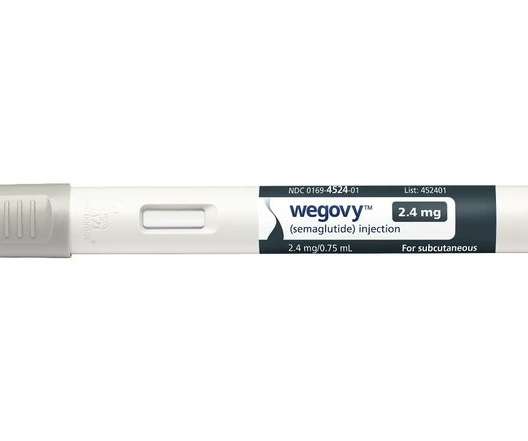
Novo Nordisk’s new obesity therapy Wegovy has been recommended for routine NHS use by UK cost-effectiveness watchdog NICE, but in a narrower group of patients than is covered by its MHRA-approved label.
pharmaphorum
DECEMBER 22, 2022
One of the most highly-anticipated developments in pharma next year will be the FDA’s verdict on Eisai’s accelerated application for lecanemab in Alzheimer’s disease, due early in January.
pharmaphorum
DECEMBER 17, 2021
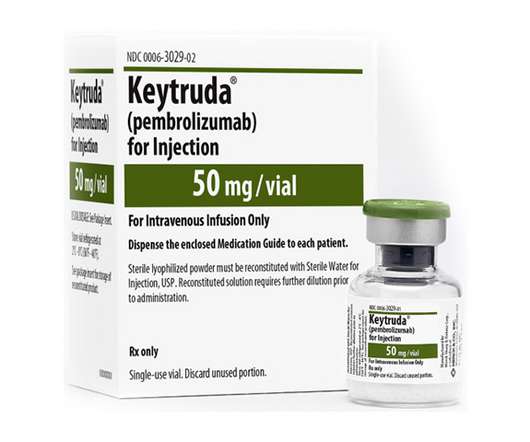
After three years of availability via the Cancer Drugs Fund (CDF), MSD’s checkpoint inhibitor Keytruda has now been approved for routine NHS use after surgery for people with melanoma to prevent the cancer returning.
pharmaphorum
NOVEMBER 16, 2021
NHS patients in England and Wales with drug-resistant epilepsy are in line for access to another treatment option, after NICE recommended routine use of Angelini group company Arvelle Pharma’s Ontozry.
pharmaphorum
APRIL 20, 2021
Novartis has secured backing from NICE for its relapsing multiple sclerosis (RMS) therapy Kesimpta in the UK, just two weeks after the drug was approved by the national drugs regulator. .

pharmaphorum
OCTOBER 21, 2021
UK cost-effectiveness watchdog NICE is set to recommend NHS use of Alnylam’s gene-silencing therapy Givlaari in England and Wales for the rare disease acute hepatic porphyria (AHP), after its advisors issued a positive verdict on the drug.

pharmaphorum
JULY 9, 2021
Orchard Therapeutics’ gene therapy for rare childhood disease metachromatic leukodystrophy (MLD) Libmeldy has been rejected for NHS use by NICE in draft guidance.
pharmaphorum
SEPTEMBER 1, 2021
Novartis’ cholesterol-lowering drug Leqvio will be made available on the NHS in England and Wales, and could help to prevent up to 30,000 deaths, according to NICE.

pharmaphorum
NOVEMBER 5, 2021
Tens of thousands of people with chronic kidney disease in England could be eligible for treatment with AstraZeneca’s SGLT2 inhibitor Forxiga, after NICE backed NHS use of the drug in draft recommendations.

pharmaphorum
JUNE 30, 2021
bluebird bio’s sickle cell disease (CD) gene therapy LentiGlobin is the latest recipient of an ‘innovation passport’ introduced in the UK earlier this year to speed up NHS access to promising new medicines.
pharmaphorum
DECEMBER 21, 2022
AstraZeneca and Daiichi Sankyo’s Enhertu will become an option for hundreds more people with breast cancer in England and Wales, thanks to new guidance from NICE that will make it available via the Cancer Drugs Fund.
pharmaphorum
MARCH 8, 2021
NHS patients in England will not be able to get access to AstraZeneca’s PARP inhibitor Lynparza if they have BRCA-positive advance prostate cancer, according to draft guidance from NICE.

pharmaphorum
NOVEMBER 7, 2022
Draft guidance in the UK has provisionally recommended that digital cognitive behavioural therapy (CBT) apps can be used within the NHS to treat children and young people with mild to moderate anxiety.

pharmaphorum
JANUARY 19, 2021
The Scottish Medicines Consortium (SMC) has given a green light to Roche’s Rozlytrek for a rare form of lung cancer, almost seven months after NICE backed the drug in England.

pharmaphorum
MARCH 9, 2022
There isn’t enough evidence to support the use of Merck & Co/MSD’s Keytruda in combination with chemotherapy as a first-line treatment for advanced triple negative breast cancer, according to the UK cost effectiveness watchdog NICE.

pharmaphorum
JANUARY 19, 2023
Alnylam’s gene-silencing drug Amvuttra has been recommended as a treatment for hereditary transthyretin-related (ATTR) amyloidosis by NICE, paving the way for the drug to be made available on the NHS in England and Wales.

pharmaphorum
JANUARY 13, 2023
Cholesterol-lowering statin drugs could be available to millions more patients in the UK, if new recommendations from health technology assessment (HTA) organisation NICE are implemented.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content