Islet transplantation for type 1 diabetes: Do regulators have it right?
Pharmaceutical Technology
OCTOBER 9, 2023
Diabetes researchers oppose the regulation of islet transplantation as a biologic, following the approval of CellTrans’ Lantidra.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
OCTOBER 9, 2023
Diabetes researchers oppose the regulation of islet transplantation as a biologic, following the approval of CellTrans’ Lantidra.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 2, 2024
With a focus on equipping regulators with advanced skills, the CDSCO conducted 23 residential training programmes tailored to address critical aspects such as […]
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
DECEMBER 21, 2022
But as the field grows in leaps and bounds, many authorities have prioritised the release of new guidelines, frameworks, and regulations to keep pace with these advances. This could spell high-stake consequences for consumers whose privacy and safety could be at risk if AI models are not regulated. Major AI deals in 2022.

AuroBlog - Aurous Healthcare Clinical Trials blog
DECEMBER 13, 2022
The National Commission for Homoeopathy has prepared a regulation defining a structure to the Bachelor of Homoeopathic Medicine and Surgery (BHMS) course with changes including incorporation of a methodology to supplement modern advancement, research and technology in Homoeopathy (SMART-Homoeopathy).

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Outsourcing Pharma
APRIL 29, 2024
OSP spoke to Berkeley Research Group healthcare managing director, Wendy Cheng for a discussion around developments in AI regulation for pharmaceuticals, clinical trials, and drug development at a global scale.

Pharmaceutical Technology
MARCH 23, 2023
This week, the Medicines and Healthcare products Regulatory Agency (MHRA) is introducing major changes to clinical trial regulation in the UK with expedited timelines at several stages. The MHRA hopes this new framework “will remove obstacles to innovation” and “streamline the regulation of clinical trials” amongst other things.

Advarra
JULY 26, 2022
As such, we are seeing an uptick in research examining the therapeutic effects of cannabis and its constituents. While there isn’t a significant amount of research on cannabidiol (CBD), it’s easily accessible in the U.S. This can make research challenging, but not insurmountable. Funding Research on Cannabis.

WCG Clinical
MARCH 27, 2024
To view it please enter your password below: Password: The post Pay to Participate in Research: Regulations and WCG’s Position appeared first on WCG. This content is password protected.

ProRelix Research
NOVEMBER 27, 2023
The United States Food and Drug Administration (FDA) has strict regulations regarding pharmacovigilance activities that are a part of post-marketing studies for drugs and medical devices that are required as […] The post FDA Pharmacovigilance Regulations Update appeared first on ProRelix Research.

Bio Pharma Dive
DECEMBER 12, 2023
Just before clearing Pfizer’s buyout of Seagen, the regulator threatened to block Sanofi’s licensing deal with Maze Therapeutics, a challenge the biotech called ‘‘unprecedented.”

ProRelix Research
FEBRUARY 12, 2023
Historical events such as the sulfanilamide elixir tragedy that resulted in the mass poisoning and the deaths of hundreds of patients in the 1930s and the thalidomide scandal which caused […] The post Clinical Trial Regulations in the United States appeared first on ProRelix Research.

Medical Xpress
FEBRUARY 13, 2023
UT Southwestern Medical Center researchers have mapped gene control elements in specialized cardiac cells responsible for coordinating heartbeats.

BioSpace
JUNE 2, 2022
Top research stories, including gene regulation and longevity, a head-to-head comparison of COVID-19 vaccines, a newly discovered type of brain cell and more.

ProRelix Research
DECEMBER 14, 2023
The European Medicines Agency (EMA) coordinates the European […] The post EMA Pharmacovigilance Regulations Updates appeared first on ProRelix Research. Pharmacovigilance is the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other medicine-related problem.

Outsourcing Pharma
APRIL 29, 2024
OSP spoke to Berkeley Research Group healthcare managing director, Wendy Cheng for a discussion around developments in AI regulation for pharmaceuticals, clinical trials, and drug development at a global scale.

Scienmag
MAY 6, 2021
Jude Children’s Research Hospital scientists have developed a highly efficient method to address a major challenge in biology–identifying the genetic ‘switches’ that regulate gene expression. Jude Children’s Research Hospital St. Credit: St.

Medical Xpress
DECEMBER 13, 2022
Researchers from the University at Buffalo School of Pharmacy and Pharmaceutical Sciences recently published in Molecular & Cellular Proteomics, describing their work in identifying key metabolic regulators involved in cancer cell resistance to gemcitabine (Gem), a standard-of-care chemotherapy for pancreatic dual adenocarcinoma (PDAC), the most lethal (..)

Scienmag
MAY 7, 2022
A research led by Hong Kong University of Science and Technology (HKUST) has revealed a novel mechanism that regulates secretion of sonic hedgehog (Shh), a key signaling molecular that plays an important role in cancer progression, in mammals, opening the door to novel therapeutic strategies for cancer induced by the hedgehog signaling pathway.
Drug Discovery World
MARCH 23, 2023
These changes represent the biggest overhaul in UK clinical trials regulation in over 20 years and are designed to make the UK one of the best countries in the world to conduct clinical research. The reforms are a significant step forward for UK clinical trials and come at a crucial time for industry clinical research in the UK.”

pharmaphorum
MAY 28, 2024
Navigating the ethical landscape of clinical research with minors: exploring the HHS and FDA regulations around parental permission, child assent, and adolescent autonomy under CFR 46. Learn how to ethically and legally involve young participants in studies.
Drug Discovery World
MARCH 27, 2024
Their research, published in the journal Cancer Discovery , reveals the role of epigenetics in controlling how cancer cells can become dormant – and suggests a strategy to target it before the cells wake up. They also found that in people with low expression of these enzymes, their cancer had a lower risk of coming back years later.

FDA Law Blog
MARCH 10, 2024
Hyman, Phelps & McNamara Director Larry Houck will participate as a panelist focusing on this timely topic at the Food and Drug Law Institute’s (“FDLI’s”) Legal and Practical Issues in Cannabis Regulation Conference next month. The conference, held in Washington, D.C., April 4th and 5th, is an in-person and virtual event.

Medical Xpress
MAY 9, 2023
An international group of doctors and public health experts have joined the clamor for a moratorium on AI research until the development and use of the technology are properly regulated.

WCG Clinical
DECEMBER 13, 2023
Dietary Supplements in Research Studies When used as investigational products in a study, dietary supplements may be subject to varying regulations and requirements. Next, let’s look at considerations for study design: Study objectives are crucial in defining the specific aim of the research and the intended use of the product.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 11, 2023
Clinical Establishment (CE) Regulation should be adopted on priority basis because it stipulates eligibility and qualification of personnel running a lab, standards or guidelines on the infrastructure and testing facilities.

Medical Xpress
FEBRUARY 23, 2023
We do know that an overactive innate immune system is causing severe COVID-19 disease, but it is unclear how this is regulated. But why is this the case? Unfortunately, we do not know exactly.

Scienmag
APRIL 22, 2021
Notch proteins are key regulators of growth and differentiation of both normal and cancer cells. Researchers in Turku, Finland, have now demonstrated that the activities of distinct Notch family members are modified differently by phosphorylation.

Scienmag
APRIL 11, 2022
Yale scientists uncover key regulator of body weight. Yale scientists have discovered that a protein known as augmentor-alpha regulates body weight in mice, an insight that could lead to new treatments for metabolic disorders. New Haven, Conn.

FDA Law Blog
MARCH 31, 2024
Gibbs — On March 21, 2024, the House Energy and Commerce held a subcommittee hearing titled “Evaluating Approaches to Diagnostic Test Regulation and the Impact of the FDA’s Proposed Rule.” By Ana Loloei & Jeffrey N. FDA, which was not invited to participate, would surely have concurred.

Scienmag
APRIL 25, 2022
BOSTON — Proper function of the kidney is critical for concentrating urine, regulating blood pressure, and for the tight control of electrolyte levels in the blood. The kidney achieves these important functions through many microscopic functional units, called nephrons. How these segments form during development and […].

Rethinking Clinical Trials
APRIL 24, 2024
Matthew Semler In this Friday's PCT Grand Rounds, Lauren Milner of the US Food and Drug Administration (FDA) and Jonathan Casey and Matthew Semler of Vanderbilt University will present "Waiver or Alteration of Informed Consent for Minimal Risk Clinical Investigations – FDA Regulation Development and Research Landscape."
Scienmag
JULY 19, 2022
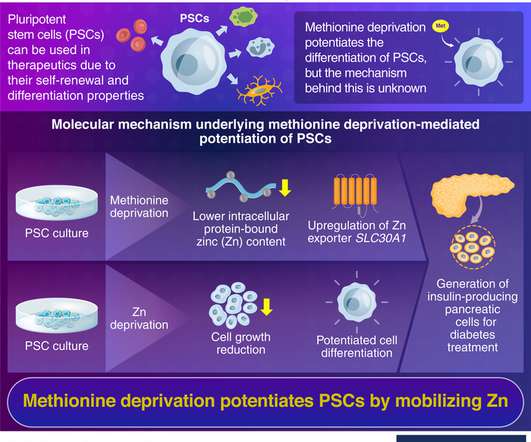
Differentiation of pluripotent stem cells (PSCs) is regulated through a methionine-mediated mechanism, which has now been pinpointed by Tokyo Tech researchers. They have revealed that zinc (Zn) plays a crucial role in PSC potentiation. They used these insights to design a protocol to convert PSCs into insulin-producing pancreatic ?

Bio Pharma Dive
JUNE 27, 2022
The biotech company revealed three new drug candidates for breast cancer, while laying out a path to regulators for its most advanced treatment.

Cloudbyz
APRIL 19, 2023
Introduction: The new European Union (EU) Clinical Trial Regulation (CTR), Regulation (EU) No 536/2014, came into force in January 2022, replacing the previous EU Clinical Trials Directive (2001/20/EC). This aims to increase transparency, reduce publication bias, and promote data sharing among researchers.

pharmaphorum
OCTOBER 11, 2022
An explosion of digital mental health tools being offered for sale has presented regulators with a problem – how should they be classified and assessed for safety or efficacy? Mental health problems are estimated to cost the UK economy at least £118 billion a year, according to research by the London School of Economics and Political Science.
Drug Discovery World
MAY 29, 2023
Scientists are calling for tighter regulation of stem cell treatments, claiming clinics worldwide are offering unproven ‘therapies’ for conditions as varied as hair loss and Parkinson’s. They are calling on regulators to halt these products until they can be proven safe and effective in clinical trials.

Worldwide Clinical Trials
MAY 1, 2024
For clinical research professionals, this journey often involves reconciling differing feedback from regulatory agencies, each with unique standards, priorities, and expectations. Maintaining open lines of communication helps address issues as they arise, allowing for real-time clarification and adjustments.

Pharma Tutor
APRIL 28, 2023
Expert Committee on Pharmacy Education, Law and Regulator admin Fri, 04/28/2023 - 15:45 ABOUT AUTHOR Dr. R. Thakur Chief Editor, Journal of Pharmaceutical Research Krupanidhi College of Pharmacy, Bengaluru, India. Email : drramsthakur@gmail.com

STAT News
OCTOBER 12, 2022
— Biotech companies developing cutting-edge cell and gene therapies often are tripped up by how much regulations around these drugs vary from country to country. On Wednesday, regulators from the United States, the European Union, and Japan came together to discuss the scale of the problem — and possible solutions.

STAT News
DECEMBER 15, 2022
and European regulators on Thursday to adopt new standards for approving antibody drugs against Covid, particularly for immunocompromised and other vulnerable patients. Biotech executives and a handful of academics pleaded with U.S.

pharmaphorum
JULY 19, 2022
Face-to-face discussion educates regulators on what matters most to patients, and guides future decision-making. Regulators are increasingly asking drug developers to include the patient voice in submissions, but the best way to go about this is less clear-cut. The rise of patient centricity. “It’s Personal approach.
Advarra
JANUARY 13, 2023
New privacy regulations seem to form every few months, especially with individual U.S. states adopting their own privacy regulations (e.g., Endpoint adjudication committees (EACs), also called clinical event committees (CECs), receive potentially identifiable research data from all over the world.

Cloudbyz
APRIL 20, 2023
The European Union (EU) Clinical Trial Regulation (CTR) No 536/2014, which came into effect in January 2022, has significantly changed the clinical trial landscape in the EU. This blog post outlines 10 key aspects of the new regulation that sponsors should be aware of when planning and conducting clinical trials in the EU.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content