Disc wins orphan drug tag for rare blood cancer
Pharmaceutical Technology
FEBRUARY 12, 2024
The humanised monoclonal antibody DISC-3405 is under investigation in a Phase I clinical trial, with data expected this year.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 tag
tag  Clinical Trials Related Topics
Clinical Trials Related Topics 
Pharmaceutical Technology
FEBRUARY 12, 2024
The humanised monoclonal antibody DISC-3405 is under investigation in a Phase I clinical trial, with data expected this year.

World of DTC Marketing
JUNE 1, 2021
The first clinic; trials were everything but encouraging but so much depend ended on this drug’s approval that Biogen returned and recited the data. If the drug is approved you can bet that it’s going to carry a huge price tag. Will they follow the science or will they put a price tag on hope?
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
FEBRUARY 22, 2024
Harmony is advancing pitolisant into a Phase III clinical trial that is expected to start in Q1 2024.

Rethinking Clinical Trials
DECEMBER 20, 2023
Yet enrollment of ethnic minorities in NIH clinical trials and for trials studying approved devices and drugs remains low. Further efforts are urgently needed to increase diversity in the cardiology workforce, which will improve clinical trial diversity and cardiovascular health for all.

Medical Xpress
MARCH 8, 2023
Before new therapies can reach patients, they must be tested in clinical trials in representative populations to show that they work and are safe. Failure to enroll enough participants in trials can delay the arrival of new therapies in the clinic and inflate their eventual price tags.

Rethinking Clinical Trials
FEBRUARY 28, 2024
The global decentralized clinical trial market is expected to grow at a compound annual growth rate of 30.1% There is agreement that trials need to meet the people, at home and covering clinical trial deserts. The skills may also vary between trials and getting participants the care they need.

Rethinking Clinical Trials
APRIL 18, 2023
Learn more Visit [link] Discussion Themes – What experts are being convened for the different trials? We have people with experience with post-viral symptoms, treating ME/CFS, general COVID knowledge, biomarker experience, clinical trials, patient-reported outcomes, and more. It’s all over the place.

Rethinking Clinical Trials
NOVEMBER 16, 2022
Ethics, Data Sharing, Pragmatic Clinical Trials. When you apply these rules to pragmatic clinical trials (PCTs), there are ethical challenges because PCTs often use waivers or alterations of informed consent and are embedded into ongoing clinical care using extant data. University of Michigan Medical School.

Rethinking Clinical Trials
MARCH 6, 2024
Schell Professor of Management and Applied Economics MIT Sloan School of Management Slides Keywords Food-as-Medicine, Randomized Clinical Trial, Diabetes Key Points Diabetes is common and costly. Speaker Joseph Doyle, PhD Erwin H. 9% of the U.S. From a researcher standpoint, we need more information.

Rethinking Clinical Trials
NOVEMBER 9, 2022
Interventional/Structural Fellow, Cleveland Clinic. Clinical Trial, Medical Education. The goal of the CardioNerds Clinical Trials Network is to pair equitable enrollment with trainee development. Among trial sites with affiliated programs, we invited program directors to nominate fellows to participate.

Fierce Pharma
SEPTEMBER 22, 2023
Merck and Eisai’s Keytruda-Lenvima tag team can’t seem to catch a break. Merck and Eisai’s Keytruda-Lenvima tag team can’t seem to catch a break.

Rethinking Clinical Trials
AUGUST 23, 2022
There is a national priority to increase diversity in clinical trials, from regulatory, funding agencies, industry, and others, so that the participant population reflects the U.S. The Duke Clinical Research Institute (DCRI) organized and sponsored a Think Tank to address diversity in clinical trial research.

Rethinking Clinical Trials
FEBRUARY 22, 2023
It will demonstrate that we can do trials at less cost than a normal trial (about 1% of a normal study). Tags #pctGR, @Collaboratory1 The post Grand Rounds February 17, 2023: The Heartline Trial: A New Paradigm in Conducting Virtual Clinical Trials (C. I think you’ll probably end up with a hybrid model.
Bio Pharma Dive
JUNE 30, 2021
The influential nonprofit maintains that the clinical trial evidence supporting Aduhelm is insufficient to prove a health benefit. But if some efficacy is assumed, ICER argues Biogen's $56,000 price tag is far too high.

Rethinking Clinical Trials
AUGUST 28, 2023
Food and Drug Administration (FDA) Slides Keywords Pragmatic trials; Research; Guidance, Regulatory, Data Governance Key Points The clinical trial enterprise needs modernization. Time, cost and failure to recruit trial participants are significant barriers that must be addressed. – What are the next steps?

Rethinking Clinical Trials
AUGUST 18, 2022
The Trial Innovation Network Recruitment Innovation Center aims to positively impact human health by improving participant enrollment and retention in multi-center clinical trials. Learn more: Improving Representation in Clinical Trials and Research. Wilkins, MD, MSCI) appeared first on Rethinking Clinical Trials.
XTalks
OCTOBER 12, 2022
Magnetic resonance imaging (MRI) has had a part in clinical trials for more than three decades now. However, the role of MRI in new drug clinical trials can involve diagnosis of lesions and determination of their severity, early identification of therapeutic responders to drug treatment and treatment monitoring and follow-up.

Rethinking Clinical Trials
AUGUST 28, 2023
Food and Drug Administration (FDA) Slides Keywords Pragmatic trials; Research; Guidance, Regulatory, Data Governance Key Points The clinical trial enterprise needs modernization. Time, cost and failure to recruit trial participants are significant barriers that must be addressed. – What are the next steps?

Rethinking Clinical Trials
MARCH 1, 2023
S1800A was a Phase II randomized study of ramucirumab plus pembrolizumab versus standard of care for NSCLC patients previously treated with immunotherapy performed within the Lung-MAP platform.

pharmaphorum
DECEMBER 23, 2021
Daiichi Sankyo has been granted breakthrough status by the FDA for patritumab deruxtecan, a HER3-targeted antibody-drug conjugate (ADC) in clinical trials for lung cancer. The post FDA gives Daiichi Sankyo’s HER3 drug a breakthrough tag appeared first on.

Rethinking Clinical Trials
JUNE 14, 2023
Tags #pctGR, @Collaboratory1 The post Grand Rounds June 9, 2023: Emulating Randomized Clinical Trials with Non-randomized Real-world Evidence Studies: Results From The RCT DUPLICATE Initiative (Shirley V. Wang, PhD) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
JUNE 7, 2023
Tracy Wang: Until there is more widespread experience in supporting decentralized trials, I do think there’s still value to the “local” clinical sites and certainly more patient familiarity and comfort.

Rethinking Clinical Trials
MARCH 28, 2023
It is an independent, nonprofit, research institute and leading funder of patient-centered comparative clinical effectiveness research (CER). It has grown to be a major force for supporting pragmatic research, both real-world evidence research and supporting observational research and pragmatic clinical trials. enabled research.

Rethinking Clinical Trials
OCTOBER 4, 2022
Senior Director Clinical Trial Operations. Neurological Clinical Research Institute. Clinical Trial Transformation Initiative (CTTI); Decentralized Clinical Trials (DCTs); Digital endpoints; Developing novel endpoints. Marianne Chase. Massachusetts General Hospital. Jörg Goldhahn, MD. ETH Zurich.

Rethinking Clinical Trials
OCTOBER 19, 2022
There are additional complexities with pragmatic clinical trials (PCTs), including overlapping roles and responsibilities of clinical and research staff; collection of broad sets of CDEs can make data monitoring more challenging; various and combined methods for data collection (e.g. What should be monitored and how?

STAT News
JANUARY 6, 2023
Such decisions are based on myriad factors, starting with the average $26,500 price tag. But there are other considerations, including the quality of the clinical trial data, side effect concerns, the patient population for which the medicine is approved, and budgetary constraints.

pharmaphorum
JUNE 8, 2021
Dementia specialist Prof Robert Howard of University College London (UCL) was also damning in his assessment of the decision, saying that the FDA has “sidestepped available clinical trial outcomes data that indicate the drug probably doesn’t work.”

Rethinking Clinical Trials
JULY 6, 2023
Over the past several years, a variety of factors have accelerated the need for decentralized trials, including the push to make trials more accessible, the increased speed of science, the possibility of environmentally conscious trials, and the need to be flexible in a rapidly changing world.

Rethinking Clinical Trials
OCTOBER 2, 2023
The PROTEUS Consortium’s objective is to ensure that patients, clinicians, and other decision-makers have high-quality PRO data from clinical trials and clinical practice to make the best decisions they can about treatment options.

XTalks
AUGUST 23, 2021
This is not a debate on approval,” said Stacie Weninger, PhD, president of the F-Prime Biomedical Research Initiative, in a focused topic session on the impact of drug approval on future clinical trials held on Tuesday July 27. Now that [aducanumab] is approved, how will this affect clinical trials going forward?”.

Rethinking Clinical Trials
JULY 22, 2022
There were four ACTIV fast-track focus areas: vaccines, preclinical, clinical trial capacity, and therapeutics – clinical. Candidate agents were triaged based on concurrent clinical trials, completion of a multiple ascending dose study, and availability of preclinical data before being scored based on predefined criteria.

Rethinking Clinical Trials
JULY 9, 2023
Clinical research can exacerbate disparities, because clinical trials typically are based in urban, academic medical centers, underrepresent diverse populations, and overlook community engagement strategies in trial planning and design.

Rethinking Clinical Trials
JANUARY 17, 2024
UPMC did not allow patients to receive experimental COVID-19 therapies outside of the context of a clinical trial and used the REMAP-CAP platform, a global pragmatic adaptive trial platform, in all clinic sites. McCreary, PharmD, BCIDP) appeared first on Rethinking Clinical Trials.

The Pharma Data
JANUARY 31, 2021
The phase 1 clinical trial was a randomized, double-blind and placebo-controlled study in 150 adult and elderly participants. These findings give confidence that Clover’s COVID-19 vaccine candidates are suitable for further clinical development.” About Trimer-Tag© Technology. . CHENGDU, China , Feb.

Rethinking Clinical Trials
MAY 19, 2023
Colwill Professor and Vice Chair Department of Family and Community Medicine University of Missouri Slides Keywords Electronic Health Record, Pragmatic Clinical Trial Key Points Patients bring patient-generated home blood pressure data into the clinical workflow. Koopman, MD, MS) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
JANUARY 26, 2023
In our study the staff could add the events, but for example in Sweden, where the data has been shown to be better detailed in the national registry, the folks in the Swedish trials is randomized and the follow up data is collected in the national registry.

Rethinking Clinical Trials
OCTOBER 31, 2023
Tags #pctGR, @Collaboratory1 The post Grand Rounds October 27, 2023: Digital, Decentralized and Democratized: Lessons From The Yale PaxLC Trial (Harlan M. Krumholz, MD, SM) appeared first on Rethinking Clinical Trials. A lot of participants have worked with their physician to stop a drug so they can participate.

Rethinking Clinical Trials
JANUARY 23, 2024
Tags #pctGR, @Collaboratory1 The post Grand Rounds January 19, 2024: Why Are Imaging RCTs Different? Lessons From Chest Pain Evaluation Trials (Pamela S. Douglas, MD, MACC, FASE, FAHA) appeared first on Rethinking Clinical Trials. We think about it as a screening tool to see if a patient needs an intervention.

Rethinking Clinical Trials
FEBRUARY 14, 2024
DCP learned that leveraging health systems for large-scale clinical trials is feasible. When people understood what we were trying to do, embed research into clinical care, people really engaged and a few regional directors made participation one of the drivers of research in their region and helped us get initial groundwork.

Rethinking Clinical Trials
MARCH 15, 2023
Tags #pctGR, @Collaboratory1 The post Grand Rounds March 10, 2023: Estimands in Cluster-Randomized Trials: Choosing Analyses that Answer the Right Question (Brennan Kahan, PhD) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
NOVEMBER 21, 2023
Discussion Themes -Do you think the results support a text message nudge in most clinical trials and what is the cost effectiveness of this? This is a low-cost strategy and would be a great option to use as one of multiple strategies to use in clinical trials. The study would then recalculate the gaps.

Rethinking Clinical Trials
OCTOBER 11, 2023
The results of the Technical Assistance Workgroup’s involvement in the Clinical Centers’ development and proposal process was a much stronger set of proposals for the UH3 phase of DECIPHeR. Discussion Themes -You previously mentioned that the practice of blinding is extremely common in clinical trials and less so in implementation studies.
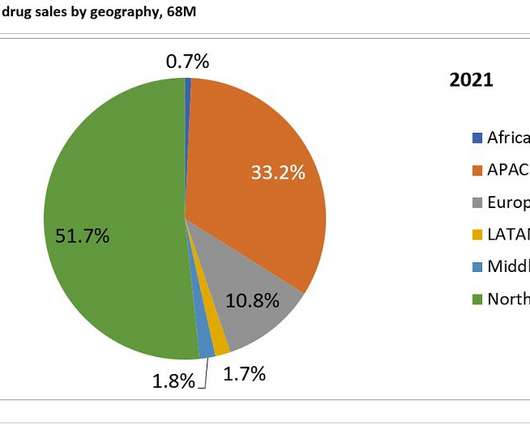
Pharmaceutical Technology
DECEMBER 15, 2022
Prior to the development of Camzyos and aficamten, obstructive HCM has not been the target of clinical trial development, so these two therapeutics would benefit a neglected patient population. of sales in 2021 and 2031 respectively. The second highest contributing market is APAC, generating 33.2%

Rethinking Clinical Trials
JANUARY 9, 2024
Tags #pctGR, @Collaboratory1 The post Grand Rounds Biostatistics Series January 5, 2024: Methods for Handling Missing Data in Cluster Randomized Trials (Rui Wang, PhD; Moderator: Fan Li, PhD) appeared first on Rethinking Clinical Trials.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content