Moderna now in the crosshairs of Sens. Warren, Welch over price tag for COVID vaccine
Fierce Pharma
JANUARY 25, 2023
Warren, Welch over price tag for COVID vaccine kdunleavy Wed, 01/25/2023 - 13:16 Moderna now in the crosshairs of Sens.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 tag covid
tag covid 
Fierce Pharma
JANUARY 25, 2023
Warren, Welch over price tag for COVID vaccine kdunleavy Wed, 01/25/2023 - 13:16 Moderna now in the crosshairs of Sens.

World of DTC Marketing
FEBRUARY 19, 2021
SUMMARY: More than 42,000 programmatic ads from 4,315 brands are running on websites spouting misinformation about the vaccine and COVID over the past year. Included in the programmatic ad errors is Pfizer, who developed a leading COVID vaccine. When will pharma stop using programmatic online ads? Restoring trust and accountability.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

World of DTC Marketing
JUNE 1, 2021
If the drug is approved you can bet that it’s going to carry a huge price tag. It could carry a price tag as high as $50,000-a-year per patient, according to drug analysts, adding billions of dollars to the nation’s health tab. . Will they follow the science or will they put a price tag on hope?
STAT News
DECEMBER 13, 2022
lawmakers wrote Pfizer chief executive officer Albert Bourla that he should “back off” from plans to charge Americans up to $130 for the company’s Covid-19 vaccine, a move they described as “pure and deadly greed.” argued the planned price tag is nearly four times the current $30 price paid by the U.S.

Rethinking Clinical Trials
APRIL 18, 2023
RECOVER has used cohort data to identify major symptom clusters and develop 5 platform protocols that will investigator priority symptom clusters and their causes; test known and novel interventions across domains; and evaluate treatments to improve Long COVID symptoms. It’s all over the place. No, there are no signs PASC is going away.

Rethinking Clinical Trials
JANUARY 17, 2024
UPMC did not allow patients to receive experimental COVID-19 therapies outside of the context of a clinical trial and used the REMAP-CAP platform, a global pragmatic adaptive trial platform, in all clinic sites. The COVID Therapeutics Committee created communication materials to be transparent and to get buy-in.

Rethinking Clinical Trials
NOVEMBER 1, 2022
HERO Registry; HERO TOGETHER; Hydroxychloroquine; COVID-19; PCORI; PCORnet. On March 21, 2020, in response to the COVID-19 pandemic, PCORI contacted leadership at Duke Clinical Research Institute and PCORnet and a decision was made to focus on the space of healthcare workers. Vanderbilt University Medical Center. Key Points.

The Pharma Data
DECEMBER 21, 2020
The Behavioural Insights Unit of the WHO released a meeting report of the Technical Advisory Group (TAG) on the special session on acceptance and uptake of COVID-19 vaccines, held on 15 October 2020. The discussion was structured around three key questions. Click here to download image in full size. . Source link.

Rethinking Clinical Trials
AUGUST 22, 2022
COVID-19, COVID-OUT. The COVID-OUT Trial is a remotely delivered, Phase 3, de-centralized clinical trial at 6 participating institutions to see if Ivermectin, Metformin or Fluvoxamine would prevent severe COVID-19. Learn more about COVID-OUT. University of Minnesota School of Public Health. Key Points.
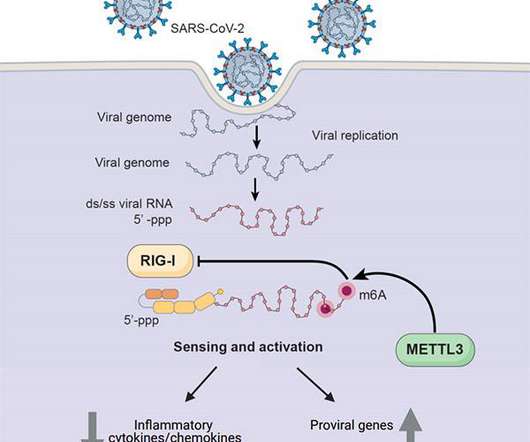
Scienmag
APRIL 29, 2021
Credit: UC San Diego Health Sciences Researchers at University of California San Diego School of Medicine have discovered one way in which SARS-CoV-2, the coronavirus that causes COVID-19, hijacks human cell machinery to blunt the immune response, allowing it to establish infection, replicate and cause disease.

Rethinking Clinical Trials
JULY 22, 2022
ACTIV, COVID-19, FNIH. ACTIV was stood up in about a month, launching on April 17, 2020, to develop a coordinated research response to speed COVID-19 treatment and vaccine options. In the therapeutics arm, the most promising therapeutic agents for COVID-19 were prioritized. Associate Vice President, Research Partnerships.

Rethinking Clinical Trials
JULY 27, 2022
ACTIV, ACTIV-6, COVID-19, Ivermectin, Inhaled Fluticasone. The key clinical questions of the ACTIV-6 study are: how to help someone feel better faster with newly diagnosed mild-moderate COVID-19 and how to prevent hospitalizations or death in someone with newly diagnosed mild-moderate COVID-19? Key Points.

World of DTC Marketing
SEPTEMBER 9, 2021
A study put a price tag on American’s bad eating habits: $50 billion a year in health care costs, attributable to cardiometabolic diseases such as heart disease, stroke, and type 2 diabetes. Then there is the cost of people who refuse to get vaccinated against COVID. Then there is the cost of unhealthy Americans.
World of DTC Marketing
JULY 20, 2022
Pfizer, for instance, hiked the cost of its leukemia medication Besponsa again this month, bringing its per-vial price tag to $21,056. “This is PPfizer’sfourth hike on the cancer drug during the Covid-19 pandemic—even as the company enjoys record-shattering profits from its vaccine,” “the analysis notes.

Pharma Tutor
NOVEMBER 19, 2021
We have also exported more than 65 million doses of COVID vaccines to nearly 100 countries this year. We have exported life-saving medicines and medical equipment to over 150 countries during the initial phase of the pandemic. Fri, 11/19/2021 - 16:19. Pharmapedia.
pharmaphorum
DECEMBER 17, 2020
Tik Tok is unlikely to spring to mind as a source of reliable information about complex issues, but scientists are using it to fly the flag for COVID-19 vaccines and other health topics. Additionally, we will soon introduce a new vaccine tag to detect and tag all videos with words and hashtags related to the COVID-19 vaccine,” says Morgan. “We

Scienmag
SEPTEMBER 9, 2020
Massive price tag of COVID-19 response in 73 low- and middle-income countries underscores benefits of investing in pandemic preparedness New modelling research, published in The Lancet Global Health journal, estimates that it could cost low- and middle-income countries (LMICs) around US$52 billion (equivalent to US$8.60

World of DTC Marketing
SEPTEMBER 7, 2022
In pharma, growth depends on new products with hefty price tags when over 80% of voters want lower costs for their prescription drugs. Although COVID-19 has slowed the growth of drug sales, the oncology market’s total growth is driven by the targeted drugs in oncology. ” The U.S.

The Pharma Data
NOVEMBER 29, 2021
1.1.529 a variant of concern, named Omicron, on the advice of WHO’s Technical Advisory Group on Virus Evolution (TAG-VE). All variants of COVID-19, including the Delta variant that is dominant worldwide, can cause severe disease or death, in particular for the most vulnerable people, and thus prevention is always key.

The Pharma Data
MAY 7, 2021
Johnson & Johnson COVID-19 Vaccinations Resume Following CDC and FDA Decision. Remarketing tags may not be associated with personally identifiable information or placed on pages related to sensitive categories. See more information and instructions on how to setup the tag on: [link] >. Skip to content. Learn More.

The Pharma Data
JANUARY 31, 2021
Adjuvanted S-Trimer COVID-19 vaccine candidates demonstrated favorable safety and tolerability profiles and strong neutralizing immune responses in a phase 1 trial. The study found that Clover’s adjuvanted S-Trimer COVID-19 vaccine candidates were well tolerated and safe. CHENGDU, China , Feb.

The Pharma Data
AUGUST 17, 2020
How We’re Mobilizing Our Resources to Help Find Solutions for COVID-19. Remarketing tags may not be associated with personally identifiable information or placed on pages related to sensitive categories. See more information and instructions on how to setup the tag on: [link] >. Learn More. Our Company.

The Pharma Data
AUGUST 14, 2020
How We’re Mobilizing Our Resources to Help Find Solutions for COVID-19. Remarketing tags may not be associated with personally identifiable information or placed on pages related to sensitive categories. See more information and instructions on how to setup the tag on: [link] >. Learn More. Our Company.

The Pharma Data
AUGUST 20, 2020
How We’re Mobilizing Our Resources to Help Find Solutions for COVID-19. Remarketing tags may not be associated with personally identifiable information or placed on pages related to sensitive categories. See more information and instructions on how to setup the tag on: [link] >. Learn More. Our Company.

Rethinking Clinical Trials
JULY 14, 2023
The study found that virtual parenting support coincided with the COVID-19 and helped families deal with the impact of the pandemic; GGC was delivered with high fidelity across cohorts, sessions, and sites; parents were satisfied with virtual GGC, with parents valuing the flexibility, connection and community.

The Pharma Data
DECEMBER 3, 2020
The Phase 1 trial was a randomized, observer-blind, placebo-controlled study to assess the safety, reactogenicity and immunogenicity of the adjuvanted COVID-19 S-Trimer vaccine candidates formulated with different antigen levels. The manuscript describing the detailed results will be accessible on an online preprint server shortly.

The Pharma Data
JANUARY 31, 2021
Clover plans to initiate a global Phase 2/3 efficacy trial of its protein-based S-Trimer COVID-19 vaccine candidate adjuvanted with Dynavax’s CpG 1018 plus alum in the first half of 2021 with an interim analysis for vaccine efficacy potentially in the middle of 2021. EMERYVILLE, Calif. and CHENGDU, China , Feb.

pharmaphorum
SEPTEMBER 15, 2020
Pharma’s go-to-market strategies have tended to be more traditional than those seen in non-regulated sectors, but there are some signs this is changing, with an acceleration in the pace of change forced by the COVID-19 pandemic that is allowing the industry to close its gap with faster, more nimble consumer brands. Jeremy Richter, Tag.

Rethinking Clinical Trials
FEBRUARY 9, 2023
The trial was designed before the COVID-19 pandemic, but it was conducted during the height of the pandemic in New York. Because of rebalancing of physicians, there were more COVID-19 patients in the intervention group. -What is the explanation for the excess deaths at 30 days in the intervention group?

Rethinking Clinical Trials
FEBRUARY 13, 2023
COVID-19 absorbed everyone in the middle of the trial. Tags #pctGR, @Collaboratory1 The post Grand Rounds February 3, 2023: Pragmatic Trials For Children With Congenital Heart Disease – Insights From The NITRIC Trial (Luregn Schlapbach, PhD, FCICM) appeared first on Rethinking Clinical Trials.

Rethinking Clinical Trials
SEPTEMBER 16, 2022
Has COVID-19 shifted the dynamic of Learning Health Systems, and if so, how? There might be an opportunity to learn from the lessons of Covid. It also means creating a pathway that ties faculty promotion to the contributions they make to the learning mission. Discussion Themes. Learn more. More info: [link]. Additional reading: [link].

Rethinking Clinical Trials
OCTOBER 13, 2022
The COVID situation was another aspect we could not control. . – What didn’t go as you expected and what were some of the lessons that you learned that would inform a follow-on trial to this trial? We had to the change the protocol once to correct a wording mistake. pctGR, @Collaboratory1.

Rethinking Clinical Trials
JUNE 7, 2023
Tracy Wang: It is safe to say that none of us predicted COVID, and it hit pretty soon after we started enrollment. We never had to make too many procedural operations because everything was set up as much as possible for remote recruitment.

Rethinking Clinical Trials
JULY 6, 2023
During the COVID-19 pandemic, several regulatory authorities around the world introduced guidance on DCTs. There has been a growing sense of hesitancy about whether regulators will remain equally receptive to these decentralized methods when the pandemic recedes.

Rethinking Clinical Trials
AUGUST 30, 2023
There were also major changes to the clinic workflows during the COVID-19 pandemic and issues with data completeness with clinic turnover. There are new medications now that were not showing up that required regular updates to the dashboard. There were also challenges with many software and security updates that impacted to the dashboard.

Rethinking Clinical Trials
NOVEMBER 21, 2022
– What challenges did you face due to COVID-19? We did not see specific site patterns, but we did see some cohort patterns. Many other trials in the country were behind and everyone else had to stop. We were fortunate that when the lockdowns were lifted everyone came back into action to complete recruitment during the time scheduled.

XTalks
JULY 16, 2021
A growing trend toward plant-based eating and distrust in the safety of meat brought on by the COVID-19 pandemic has transformed plant-based meat into a popular alternative rather than a niche product enjoyed only by vegans and vegetarians. But yet, the price tag is still higher than real meat. Turning a Profit.

pharmaphorum
AUGUST 17, 2020
In this article Joanna Carlish, managing director of financial services at Tag Americas , and Robb DeFilippis, Tag Americas’ managing director, life sciences, go head-to-head to discuss marketing within a regulated industry. Nevertheless, the advent of COVID-19 is forcing pharma, and many other sectors, to make some radical changes.

Rethinking Clinical Trials
DECEMBER 6, 2022
Enrollment halted before planned sample size of 200 due to COVID-19. There were two exploratory endpoints: change in mean daily step counts from baseline to 6 months and change in metabolomic profiling from baseline to 3 months. The trial enrolled 187 patients, with 35% women, 47% African American, 10% Hispanic, age 59 years.

Rethinking Clinical Trials
AUGUST 28, 2023
The work started before COVID-19, and the idea was to meet every six months. COVID-19 made us think about things differently, because one couldn’t apply rules, one had to apply principles. Discussion Themes – How much work was needed to get to this stage where the revisions are up for public comment?

Rethinking Clinical Trials
AUGUST 28, 2023
The work started before COVID-19, and the idea was to meet every six months. COVID-19 made us think about things differently, because one couldn’t apply rules, one had to apply principles. Discussion Themes – How much work was needed to get to this stage where the revisions are up for public comment?

XTalks
NOVEMBER 10, 2020
Despite enhanced safety measures brought on by the COVID-19 pandemic, consumers are still skeptical about the safety, source and quality of the foods they buy, according to a new survey. Implementing these technologies would require investing in handheld computers and tablets, barcode label printers and scanners, and special labels and tags.

Rethinking Clinical Trials
JULY 19, 2023
Key study limitations were the need for remote delivery due to the COVID-19 pandemic and the lack of total representation of the broader U.S. Tags #pctGR, @Collaboratory1 The post Grand Rounds July 14, 2023: Lessons From the COORDINATE-Diabetes Trial (Christopher B. or international population across the selected sites and patients.

pharmaphorum
OCTOBER 29, 2021
It is a key new product for Gilead as it faces pressure on its HIV franchise, which has been hit by reduced diagnoses and new therapy starts due to the COVID-19 pandemic, falling 8% to $4.2 COVID-19 therapy Veklury (remdesivir) continued to lead the charge at Gilead with a massive $1.9 billion for the full year.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content