DIA 2023 – a “patients included” global event
pharmaphorum
JUNE 29, 2023
DIA 2023 – a “patients included” global event Mike.Hammerton Thu, 29/06/2023 - 08:00 Bookmark this
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 patients event
patients event 
pharmaphorum
JUNE 29, 2023
DIA 2023 – a “patients included” global event Mike.Hammerton Thu, 29/06/2023 - 08:00 Bookmark this

Cloudbyz
JANUARY 4, 2024
A critical aspect of this journey is the reporting of adverse events (AEs), ensuring the safety of patients and contributing to the overall success of the device. Regulatory bodies may only necessitate the reporting of serious adverse events (SAEs).
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Medical Xpress
NOVEMBER 7, 2022
A new study by researchers at Intermountain Healthcare in Salt Lake City finds that following up a cardiac event, such as a heart attack, with a statin prescription and cholesterol measuring blood test, prevents 94% of patients from having or dying from a second cardiovascular event during the next three years.

Fierce Pharma
JULY 11, 2023
The study was assessing the drug in certain patients with previously untreated diffuse large B-cell lymphoma.

Advertisement
Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Outsourcing Pharma
FEBRUARY 28, 2022
The recent Outsourcing-Pharma webinar hosted a trio of industry experts highlighting challenges, opportunities, and innovations in the rare disease realm.

Pharmaceutical Technology
MARCH 11, 2024
Wegovy can now be used to reduce the risk of major cardiovascular events in obese or overweight patients with heart disease.

Medical Xpress
MARCH 6, 2023
Researchers from the University of Calgary, the Interdisciplinary Chronic Disease Collaboration (ICDC), and social impact creative agency Emergence Creative are announcing dramatic results that demonstrate a significant improvement in cardiovascular outcomes among patients who received access to a novel educational and support intervention called MOXIE. (..)

The Pharma Data
MARCH 2, 2021
BLINCYTO demonstrated significantly prolonged event-free survival (events were defined by relapse, death, second malignancy, or failure to achieve complete remission) compared with chemotherapy. months follow-up, 69% of patients treated with BLINCYTO were alive and event-free compared with 43% of patients treated with chemotherapy.

Bio Pharma Dive
MARCH 9, 2022
Patients with severe hemophilia A who were given the drug had, on average, less than one bleeding event over a year, results that Sanofi and partner Sobi believe will pass muster with regulators.
Pharma Times
NOVEMBER 8, 2021
Detailed results from the CheckMate-816 trial will be shared at an upcoming medical conference

BioTech 365
DECEMBER 5, 2021
Rexlemestrocel-L Shows Greatest Treatment Benefit on Major Adverse Cardiovascular Events in High-Risk Heart Failure Patients With Diabetes and/or Myocardial Ischemia Rexlemestrocel-L Shows Greatest Treatment Benefit on Major Adverse Cardiovascular Events in High-Risk Heart Failure Patients With Diabetes and/or Myocardial Ischemia … Continue reading (..)

Pharma Times
AUGUST 24, 2021
Data presented at ESC Congress 2021

Bio Pharma Dive
SEPTEMBER 1, 2021
The company is holding off dosing more patients with AT132, a potential treatment for a deadly neuromuscular disease, after one recently experienced a serious adverse event in the form of unusual liver function.

Rethinking Clinical Trials
MARCH 15, 2023
In this Friday’s PCT Grand Rounds, Ethan Basch of the University of North Carolina at Chapel Hill will present “Patient-Reported Outcomes for Symptom and Adverse Event Monitoring in Oncology.” ” The Grand Rounds session will be held on Friday, March 17, 2023, at 1:00 pm eastern. Basch is the Richard M.

Rethinking Clinical Trials
MARCH 25, 2024
The Patient-Centered Outcomes Research Institute (PCORI) announced the release of its new Foundational Expectations for Partnerships in Research. It builds on a growing body of evidence about engaging patients and other partners in patient-centered comparative effectiveness research. Register for the webinar.

BioSpace
DECEMBER 6, 2023
While almost half of multiple myeloma patients on linvoseltamab achieved a complete response or better, all experienced adverse events and 14 patients died due to treatment-emergent AEs.

Pharmaceutical Technology
OCTOBER 4, 2022
The type 2 diabetes (T2D) space recently witnessed a new approval: on 15 September, the European Commission (EC) authorised Eli Lilly’s Mounjaro (tirzepatide) for T2D patients. With respect to its safety profile, adverse events ranged from mild to moderate among subjects, and treatment discontinuation due to adverse events was 6.2%
Pharmaceutical Technology
FEBRUARY 10, 2023
4DMT had paused enrollment of patients to two of its trials for 4D-310 last month following a significant adverse event where three patients experienced kidney issues; however, these were treated and resolved in a four-week period.

BioTech 365
SEPTEMBER 28, 2021
AI-Powered Insights, Decentralized Clinical Trials, Diversity in Clinical Trials, and Patient Engagement Among the Critical Topics to Be Showcased at Medidata’s NEXT Global 2021 Event AI-Powered Insights, Decentralized Clinical Trials, Diversity in Clinical Trials, and Patient Engagement Among the Critical … Continue reading →

BioTech 365
JUNE 7, 2021
MagForce AG Supports World Brain Tumor Day and Informs about Various Patient Events to Raise Awareness for One of … Continue reading → DGAP-News: MagForce AG / Key word(s): Miscellaneous 07.06.2021 / 08:30 The issuer is solely responsible for the content of this announcement.

Pharma Times
JULY 7, 2021
During the event, the panel explored how partnerships innovated in the face of challenges

Rethinking Clinical Trials
NOVEMBER 22, 2022
A concordance analysis from ADAPTABLE, a large pragmatic, comparative effectiveness trial, found low to moderate agreement between patient-reported health data and data derived from the electronic health record (EHR). The findings highlight the need for better integration of patient-reported health data into pragmatic research studies.

BioTech 365
NOVEMBER 19, 2020
BeiGene Announces the Approval of XGEVA® (Denosumab) in China for the Prevention of Skeletal-Related Events in Patients With Bone Metastases From Solid Tumors and in Patients With Multiple Myeloma BeiGene Announces the Approval of XGEVA® (Denosumab) in China for the … Continue reading →

Pharma Times
JUNE 24, 2021
‘What’s next for patient advocacy?’ ’ will take place on Tuesday 6th July at 8.30am
Pharmaceutical Technology
APRIL 20, 2023
Zucara Therapeutics has received clearance from the US Food and Drug Administration (FDA) for its investigational new drug application for ZT-01 to prevent night-time (nocturnal) hypoglycaemia in type 1 diabetes (T1D) patients. The rate of nocturnal hypoglycaemic events is the primary endpoint of the trial.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 17, 2023
Adverse drug reactions (ADRs) reporting with the help of digital technologies will give us the opportunity to evaluate our capability to detect signals of adverse events.

Pharmaceutical Technology
JUNE 8, 2023
The use of 4D Molecular Therapeutics’ (4DMT) aerosolised gene therapy 4D-710 has improved the quality-of-life and spirometry-measured outcomes in three cystic fibrosis patients , based on early results from a Phase I/II study presented at this year’s annual meeting of the European Cystic Fibrosis Society (ECFS).

BioTech 365
SEPTEMBER 14, 2020
14, 2020 (GLOBE NEWSWIRE) — ATLANTA – The American College of Rheumatology (ACR) will hold its first virtual Advocates for Arthritis event on Tuesday, Sept. 15, where more than 120 rheumatologists, rheumatology health professionals, and patient advocates will … Continue reading → ATLANTA, Sept.

BioSpace
NOVEMBER 12, 2023
Novo Nordisk posted new data at AHA 2023 showing that its blockbuster weight loss drug could cut the risk of major cardiovascular events—including heart attack—across patient subgroups.

BioTech 365
NOVEMBER 11, 2021
Merck Announces Initiation of Phase 3 Study Evaluating VERQUVO® (vericiguat) in Patients with Chronic Heart Failure and Reduced Ejection Fraction Who Have Not Had a Recent Worsening Heart Failure Event Merck Announces Initiation of Phase 3 Study Evaluating VERQUVO® (vericiguat) … Continue reading →

Intouch Solutions
NOVEMBER 9, 2023
The contributions raised by this event fund lifesaving research, advocacy and support for blood cancer patients and their families. We were proud to walk side-by-side with patients, providers, and clients supporting the fight against blood cancers. Catch a glimpse of the event in the pictures and video below.

Fierce Pharma
DECEMBER 9, 2023
BCMA-targeted therapies are transforming care for multiple myeloma patients. A new study examined side effect reports of BMCA immunotherapies from the FDA Adverse Event Reporting System, hoping to help inform doctors in their treatment decisions. But these immunotherapies also come with various potentially dangerous side effects. |

Fierce Pharma
JULY 21, 2023
A week after ADC Therapeutics paused its Zynlonta trial to review seven deaths and five other respiratory events in patients who received the drug, the company has scrapped the study altogether fol | After severe respiratory events and seven deaths held up ADC's Zynlonta study in unfit or frail patients with previously untreated diffuse large (..)
STAT News
MARCH 20, 2023
Editor’s note: A livestream on the event will be embedded below at 1 p.m. Patients are having a difficult time seeing doctors quickly and understanding what specialists they can see. Physicians are burned out and overworked. Read the rest…

Medical Xpress
MAY 10, 2023
In the wake of an opioid-related event such as an overdose, infection, or detox admission, white patients received medication for opioid use disorder (OUD) up to 80% more frequently than Black patients and up to 25% more frequently than Hispanic patients, according to a new study led by researchers at Harvard T.H.

pharmaphorum
DECEMBER 1, 2023
AstraZeneca has decided to abandon two phase 3 trials of its drug to reduce high levels of potassium in the blood (hyperkalaemia) – Lokelma – in patients with kidney disease.
Pharmaceutical Technology
AUGUST 8, 2022
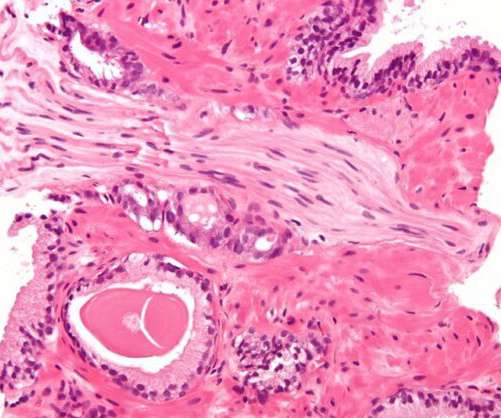
The US Food and Drug Administration (FDA) has granted approval for a supplemental New Drug Application (sNDA) of Orion and its partner Bayer ’s Nubeqa (darolutamide) plus docetaxel to treat metastatic hormone-sensitive prostate cancer (mHSPC) patients. Overall survival was the trial’s primary endpoint.

Pharmaceutical Technology
MAY 12, 2023
Marketed as Brilinta by AstraZeneca, ticagrelor has been developed for heart attack or stroke patients and those with acute coronary syndromes and coronary artery disease (CAD). It was found to be well-tolerated, with only minor adverse events observed. The US FDA granted breakthrough therapy designation to bentracimab in 2019.

Worldwide Clinical Trials
APRIL 5, 2024
At Worldwide, we are dedicated to amplifying the voices of those directly impacted by rare diseases — patients, caregivers, family members, advocates, and healthcare professionals alike. Overall, 35 people from across 19 unique patient organizations, sponsors, and industry organizations joined in to share their rare moments.

Rethinking Clinical Trials
APRIL 4, 2024
Miguel Vazquez, principal investigator for ICD-Pieces A primary care intervention for patients with coexisting chronic kidney disease, type 2 diabetes, and hypertension did not reduce the hospitalization rate for these patients when compared to usual care, according to the ICD-Pieces study. Read the full article.

Rethinking Clinical Trials
JUNE 7, 2023
The trial was designed to randomize 1,000 patients who had an On-X aortic valve replacement at least 3 months prior to randomization with either a standard dose of Apixaban (5mg) or continued warfarin (the standard of care). The drugs were administered open label and all patients were given a low dose of aspirin.
Pharmaceutical Technology
JUNE 4, 2023
Early-stage non-small cell lung cancer (NSCLC) is typically treated with surgery followed by adjuvant therapy (chemo ± radiation) in patients deemed to be at higher risk. months, the two-year event-free survival (EFS) was 62.4% The discontinuation rate due to adverse events was 12.5% At a median duration of follow-up of 25.2
Pharmaceutical Technology
MAY 17, 2023
By replacing the missing or deficient ADAMTS13 enzyme, TAK-755 offers targeted therapy for addressing an unmet medical need for thrombotic thrombocytopenic purpura patients. We continue to be encouraged by the data and are working closely with the US FDA and other global regulatory bodies with the goal to bring this treatment to patients.”
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content