New HIV Prevention Drug Shows 100% Efficacy in Clinical Trial
AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 14, 2024
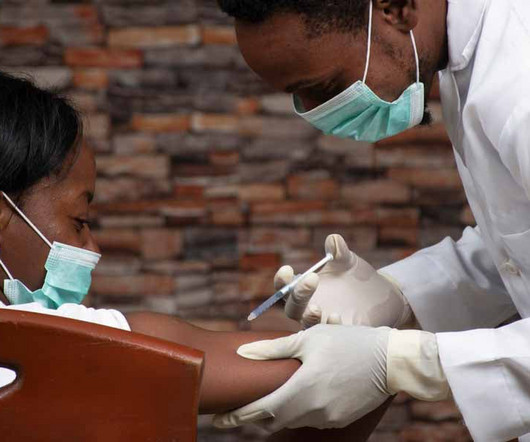
A large clinical trial in South Africa and Uganda has shown that a twice-yearly injection of a new pre-exposure prophylaxis drug gives young women total protection from HIV infection. All three medications […]


















































Let's personalize your content