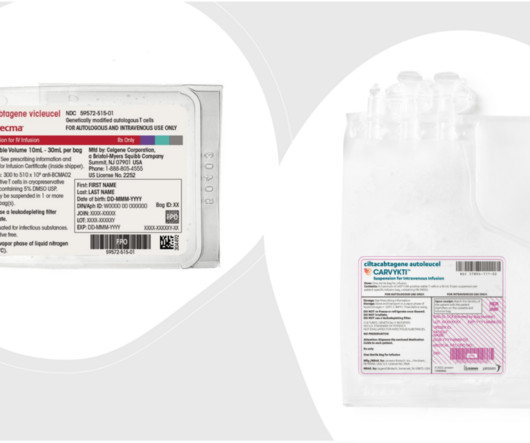
J&J’s Carvykti and BMS’ Abecma Win FDA Approvals for Earlier Use in Multiple Myeloma
XTalks
APRIL 11, 2024
In Abecma’s expanded approval, the therapy has a new recommended dose range of 300 to 510 x 10 6 CAR-positive T cells compared to a maximum of 460 x 10 6 in its original approval. In J&J’s CARTITUDE-4 trial, a higher proportion of patients in the Carvykti cohort died within the first ten months compared with the standard therapy group.








Let's personalize your content