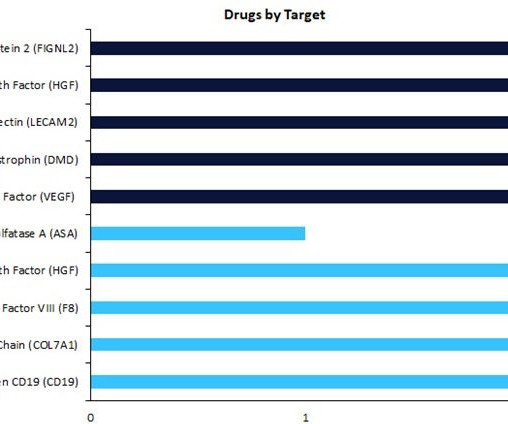
Top three trends in precision medicine
Pharmaceutical Technology
SEPTEMBER 19, 2024
An innovative approach to healthcare, precision medicine is a new medical model that takes into account individual differences in genes, environments, and lifestyles to drive better outcomes through medicine.













































Let's personalize your content