Using bispecific antibodies to advance cancer immunotherapy
pharmaphorum
NOVEMBER 2, 2021
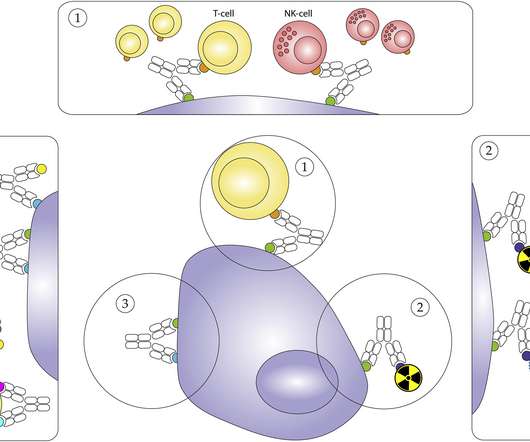
Clinical interest in bsAbs gained momentum following the promising data and market success of blinatumomab (BLINCYTO®, Amgen), a fragment-based bispecific T cell engager (BiTE), which first received FDA approval in 2014 and EMA approval in 2015. Simplified schematic overview of the proposed mechanisms of action for bsAbs.








Let's personalize your content