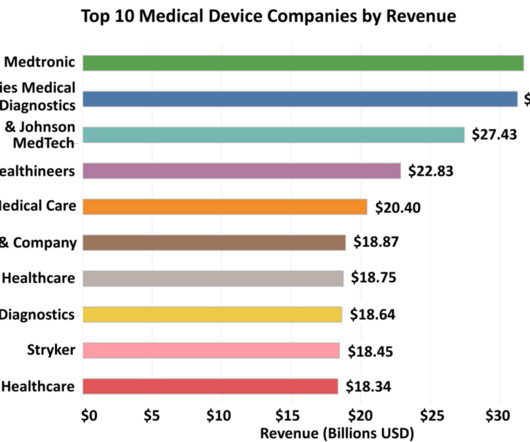
Top 10 Medical Device Companies in 2023 by Revenue Statistics and Trends
XTalks
JUNE 12, 2023
In Medtronic’s annual report, CEO Geoff Martha attributes their success in this area to their comprehensive suite of products and solutions, which equip clinicians with the tools necessary for optimal atrial fibrillation care. Note: When it comes to companies that report in foreign currencies, the conversion to U.S.












Let's personalize your content