“Game-changing” personalised cancer vaccine enters UK clinical trials
Drug Discovery World
APRIL 26, 2024
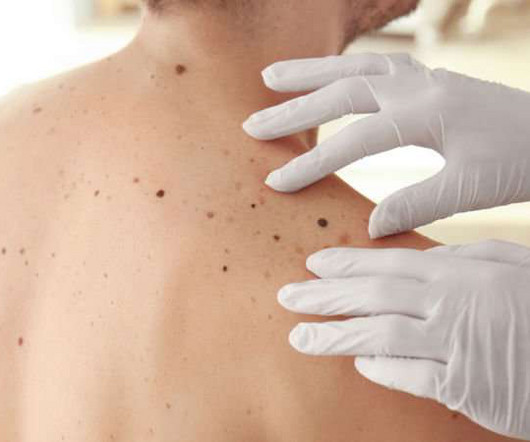
A clinical trial of a personalised mRNA cancer vaccine for melanoma patients has been launched in the UK. The vaccines are personalised to each patient after a tumour sample is removed during surgery and then analysed using DNA sequencing. in the combination arm and 62.2%























Let's personalize your content