TAKHZYRO® (lanadelumab) for Hereditary Angioedema Patients to be Presented at European Academy of Allergy and Clinical Immunology (EAACI) Hybrid Congress
The Pharma Data
JULY 11, 2021
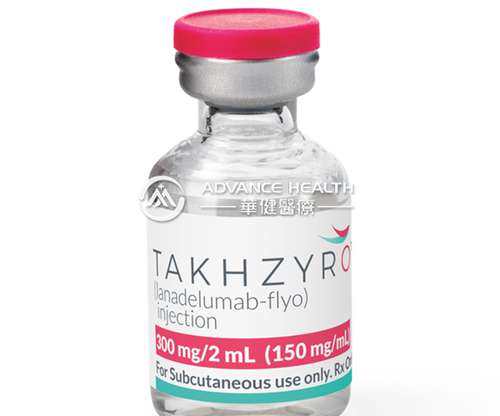
Department of Dermatology, Venereology and Allergology, Charité – Universitätsmedizin in Berlin, Germany. TAKHZYRO is produced in Chinese Hamster Ovary (CHO) cells by recombinant DNA technology. Depending on regional marketing authorization, TAKHZYRO is available as a 300 mg dose in a vial or pre-filled syringe.








Let's personalize your content