Sunlenca (lenacapavir) Receives FDA Approval as HIV Treatment Option for Multi-Drug Resistant Infection
XTalks
JANUARY 9, 2023
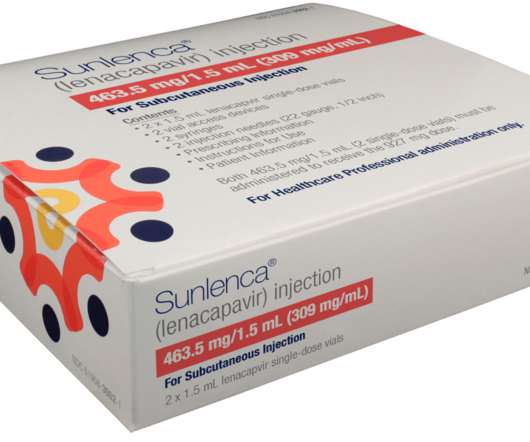
Gilead Sciences, Inc. recently announced in a press release that Sunlenca (lenacapavir) received approval from the US Food and Drug Administration (FDA) for use as a twice-yearly treatment option for people living with multi-drug resistant HIV (human immunodeficiency virus).











Let's personalize your content