Patient dies in Pfizer study of Duchenne gene therapy
Bio Pharma Dive
MAY 7, 2024
Pfizer said the patient, a young boy who was treated earlier last year, had died suddenly. The company is working with trial researchers to investigate further.

Bio Pharma Dive
MAY 7, 2024
Pfizer said the patient, a young boy who was treated earlier last year, had died suddenly. The company is working with trial researchers to investigate further.

Pharmaceutical Technology
MAY 3, 2024
UK liquid biopsy company, Angle, has agreed a supplier agreement with AstraZeneca to develop a Parsotix-based Androgen Receptor (AR) assay for use in prostate cancer studies.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
MAY 1, 2024
AstraZeneca has maintained that while the vaccine may, in “very rare” cases, cause TTS, the casual mechanism for this effect remains unknown.

Bio Pharma Dive
MAY 6, 2024
A 12-year-old boy in the Washington, D.C., area is set become the first patient treated with Lyfgenia since its U.S. approval last December.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 1, 2024
Scientists have developed a vaccine against a notorious drug-resistant superbug, targeting molecules on its surface that are also found on other bacteria and fungi.

Rethinking Clinical Trials
MAY 1, 2024
Dr. Gregory Tasian In this Friday’s PCT Grand Rounds, Gregory Tasian of the Children’s Hospital of Philadelphia will present “Comparative Effectiveness of Kidney Stone Surgery in Pediatric Patients: The PKIDS Trial.” The Grand Rounds session will be held on Friday, May 3, 2024, at 1:00 pm eastern. The Pediatric Kidney Stone (PKIDS) Care Improvement Network Trial is a patient-centered, pragmatic clinical trial comparing the effectiveness of stone clearance for 3 surgical m
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Bio Pharma Dive
MAY 2, 2024
The funding will help Reunion pay for a mid-stage study testing its most advanced medicine — essentially a synthetic version of the hallucinogenic psilocin — in women with postpartum depression.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 2, 2024
The hypertension drug rilmenidine has been shown to slow down aging in worms, an effect that in humans could hypothetically help us live longer and keep us healthier in our latter years. Previous research has shown rilmenidine mimics the effects of caloric restriction on a cellular level.

Fierce Pharma
MAY 6, 2024
As BioNTech continues to endure a sharp decline in COVID-19 vaccine sales, the German mRNA specialist is looking ahead to the next leg of its commercial journey. | With plans to have at least 10 potentially registrational trials underway by the end of 2024, BioNTech is plotting the first wave of its market debut in oncology from 2026 onward.

BioSpace
MAY 6, 2024
Adeno-associated viruses have long been go-to vectors for gene therapies. How AAVs are improving will be among the cell and gene therapy topics to be covered in Baltimore this week.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Pharmaceutical Technology
MAY 2, 2024
From Merck & Co to Sanofi, Pharmaceutical Technology lists the leading pharmaceutical companies spending the most on R&D in 2023.

Bio Pharma Dive
MAY 3, 2024
The company said data for its Wegovy competitor was promising enough to move the drug into late-stage testing, triggering a stock jump that added billions to Amgen’s market value.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 5, 2024
As the world’s aging population grows, and dementia, cardiovascular disease, and osteoporosis reach epidemic levels, people of all ages want to know how they can live healthier, not just longer, lives. For women in their 40s and 50s, it’s not too late to take action.

Fierce Pharma
MAY 1, 2024
Just about two months into his CEO job at Emergent BioSolutions, Joseph Papa has laid out his turnaround plan for the CDMO-turned-biopharma company. | Merely about two months into his CEO job at Emergent BioSolutions, Joseph Papa has laid out his turnaround plan for the CDMO-turned-biopharma company. And it involves a major organizational restructuring.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

FDA Law Blog
MAY 6, 2024
By Lisa M. Baumhardt, Senior Medical Device Regulation Expert & Adrienne R. Lenz, Principal Medical Device Regulation Expert — FDA recently issued a draft guidance which would update the agency’s Cybersecurity in Medical Devices: Quality System Considerations and Content of Premarket Submissions guidance. The draft guidance provides recommendations on what is required to meet cybersecurity obligations under section 524B of the Food, Drug and Cosmetic Act (FD&C).

Pharmaceutical Technology
MAY 3, 2024
The FDA has awarded the designation following a review of initial safety and efficacy data from two Phase I/II clinical trials.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 6, 2024
Aimed at driving innovation and progress in the field of microbiology, the Indian Pharmacopoeia Commission (IPC) and the CSIR-Institute of Microbial Technology (IMTech), Chandigarh, have joined forces towards advancing microbiological research and development for the betterment of public health.

Fierce Pharma
MAY 6, 2024
Amneal Pharmaceuticals has agreed to pay $272.5 million to settle approximately 900 lawsuits brought by state, local and Native American jurisdictions claiming that the New Jersey generics producer | Amneal Pharmaceuticals has agreed to pay $272.5 million to settle approximately 900 lawsuits brought by state, local and Native American jurisdictions claiming that the New Jersey generics producer fueled the opioid crisis by failing to act on suspicious opioid orders.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

BioSpace
MAY 6, 2024
FDA Commissioner Robert Califf has said that advisory committee meetings should focus more on discussion and be “less about the outcome,” but other agency officials recently advocated for retaining the vote on whether to approve a product under review.

Pharmaceutical Technology
MAY 3, 2024
Moderna reported a net loss of $1.2bn in the first quarter (Q1) of 2024, a stark contrast to its net income of $79m in Q1 2023.

Bio Pharma Dive
MAY 1, 2024
Sales of the shingles vaccine Shingrix and the RSV shot Arexvy helped fuel quarterly revenue totals that surpassed analyst expectations, though the company warned momentum could slow in the months ahead.

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 1, 2024
India is now putting in place a mechanism to combat malaria as drug resistance poses a serious challenge. The treatment of malaria depends on timely diagnosis and efficient medication, especially artemisinin-based combination therapies (ACTs) that help in getting rid of parasites and relieve symptoms.

Advertiser: FourKites
A research study conducted by The Journal of Commerce and FourKites surveyed hundreds of international shippers, exploring how their usage of global supply chain visibility technology has evolved since the onset of global disruptions caused by COVID-19. For international shippers, ocean freight visibility has evolved from optional to essential and satisfaction with visibility varies greatly depending on how it is obtained and delivered.

pharmaphorum
MAY 1, 2024
Discover how digital tools can provide insights to help us. Digital technology could be a game-changer that offers the elusive promise of tackling the root cause, not just the conditions themselves, when it comes to human health.

Fierce Pharma
MAY 1, 2024
It’s no secret that U.S. biopharma companies rely on Chinese contractors to provide lab research and drug manufacturing services. | Facing a push for China-U.S. decoupling from Washington, the industry trade group BIO now wants to quantify the U.S.’s dependence on Chinese CDMOs through a survey.
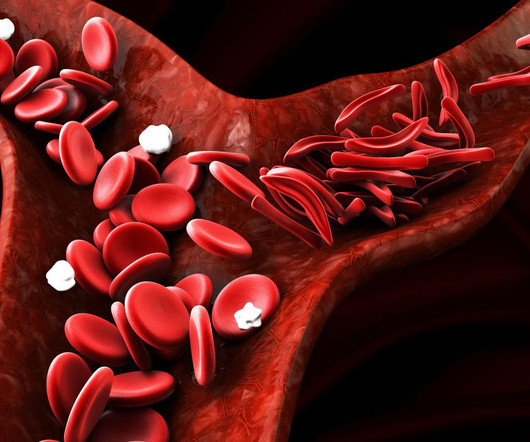
Pharmaceutical Technology
MAY 3, 2024
The NHS in the UK has announced plans to offer Pfizer's Voxelotor (Oxbryta), a new treatment option for sickle cell disease.

Bio Pharma Dive
MAY 1, 2024
Sales of the transthyretin amyloidosis treatment widely beat Wall Street forecasts in the first quarter. The company aims to extend its patent exclusivity.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

AuroBlog - Aurous Healthcare Clinical Trials blog
MAY 2, 2024
In a concerted effort to fortify regulatory capabilities and enhance pharmaceutical exports, the Central Drugs Standard Control Organisation (CDSCO) has orchestrated a series of impactful training initiatives in the fiscal year 2023-24.

BioSpace
MAY 6, 2024
Reeling from “endemic-level demand” for its COVID-19 vaccine, BioNTech on Monday reported a steep decline in revenues and a loss per share that fell short of analysts’ consensus estimates for the first quarter of 2024.

Fierce Pharma
MAY 6, 2024
“Our next question will come from Andrew Baum with Citi.” | Citi analyst Andrew Baum, after covering Pfizer for more than a decade, will join the Big Pharma company as chief strategy and innovation officer. In the role, he'll oversee the drugmaker's portfolio management and business development activities.

Pharmaceutical Technology
MAY 1, 2024
Eli Lilly has announced that its net income rose to $2.24bn in Q1 2024, a 67% increase from $1.34bn in the same period of 2023.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Let's personalize your content