Duchenne approval exposes FDA rift over Sarepta gene therapy
Bio Pharma Dive
JUNE 21, 2024
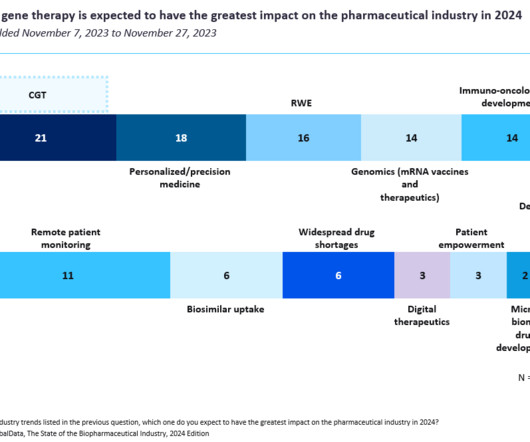
Peter Marks’ decision to override the objections of agency staff and broaden use of Elevidys could have a “lasting impact” on gene therapy as well as the FDA, one analyst wrote.








































Let's personalize your content