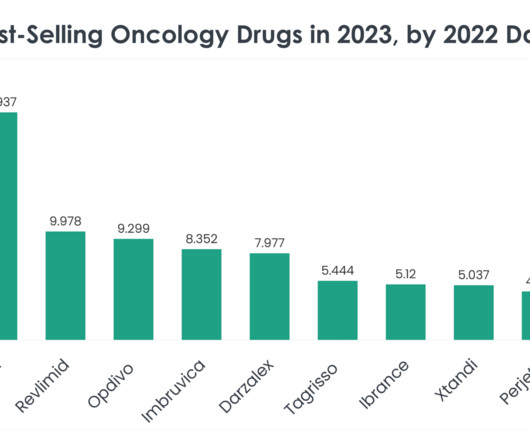
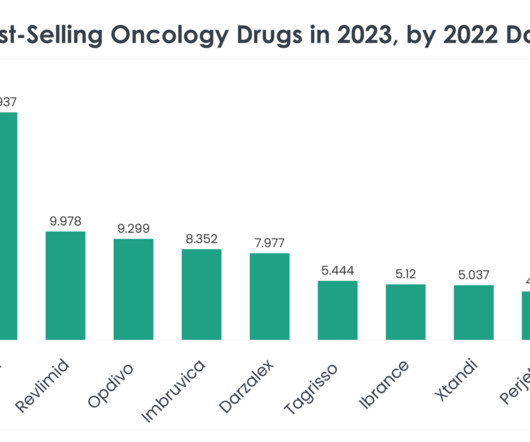
Top 40 Best-Selling Oncology Drugs in 2023 by 2022 Data
XTalks
FEBRUARY 5, 2024
Revlimid (Lenalidomide) Revlimid 2022 sales: $9.978 billion Company/Developer: Bristol Myers Squibb Date of first FDA approval: December 27, 2005 Indications Revlimid is FDA-approved for: Myelodysplastic disease (MDS), multiple myeloma, mantle cell lymphoma (MCL), follicular lymphoma and marginal zone lymphoma.



















Let's personalize your content