Almirall licenses IL-21 antibody from Novo Nordisk
Pharmaceutical Technology
FEBRUARY 19, 2024
Almirall has signed a licensing agreement for the acquisition of worldwide rights to Novo Nordisk’s IL21-hindering antibody NN-8828.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
FEBRUARY 19, 2024
Almirall has signed a licensing agreement for the acquisition of worldwide rights to Novo Nordisk’s IL21-hindering antibody NN-8828.

Pharmaceutical Technology
OCTOBER 13, 2023
Specialised Therapeutics has entered an agreement to license Korea-based CanariaBio’s oregovomab for treating ovarian cancer.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
FEBRUARY 28, 2024
AbbVie will pay $48m in upfront costs and up to $665m in milestone payments to license OSE’s monoclonal antibody OSE-230.

Pharma Mirror
APRIL 18, 2023
The license allows Sterling to manufacture antibody-drug conjugates (ADCs) for clinical use in its current Good Manufacturing Practices (cGMP) compliant manufacturing facility in Deeside, UK.

Pharmaceutical Technology
OCTOBER 12, 2023
Almirall will license EpimAb’s Fabs-In-Tandem Immunoglobulin (FIT-Ig) platform to develop up to three bispecific antibody targets.
Pharmaceutical Technology
OCTOBER 17, 2023
LegoChem Biosciences has entered an exclusive worldwide licensing agreement with Glycotope to develop an antibody-drug conjugate (ADC).
BioPharma Reporter
NOVEMBER 7, 2023
The German mRNA giant BioNTech has sealed a pact worth up to $1 billion with Biotheus to develop the Chinese biotech companyâs bispecific antibody cancer drug PM8002 in all territories except for Greater China.
Pharmaceutical Technology
APRIL 13, 2023
Biogen has exercised an option to license Denali Therapeutics ’ (Denali) programme to develop immunotherapies for the treatment of Alzheimer’s disease. ATV: Aβ uses Denali’s ATV technology to cross the blood-brain barrier (BBB) and increase brain exposure to antibodies.

Bio Pharma Dive
MAY 25, 2023
Study abstracts released Thursday give investors and analysts a better look at cancer treatments recently licensed by Merck and BioNTech as well as fresh details on other closely watched ADC candidates.

Pharmaceutical Technology
JULY 21, 2022
AstraZeneca has signed a deal with the Federal Office of Public Health (FOPH) of Switzerland to deliver over 1,200 doses of antibody therapy, tixagevimab and cilgavimab combination (AZD7442), for Covid-19 prevention and treatment. In June 2020, these antibodies, discovered at Vanderbilt University Medical Center, were licensed to AstraZeneca.

Pharmaceutical Technology
OCTOBER 21, 2022
AbbVie has announced the acquisition of UK-based biotechnology firm DJS Antibodies for nearly $255m in cash at closing. DJS focuses on the discovery and development of antibody therapies that act on difficult-to-drug disease-causing proteins, such as G protein-coupled receptors (GPCRs).

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.
Drug Discovery World
AUGUST 2, 2023
Renaissance Pharma has announced its first development programme focused on Hu14.18, a humanised anti-GD2 monoclonal antibody (mAb), licensed from St Jude Children’s Research Hospital for the treatment of newly diagnosed high-risk neuroblastoma. ” The post Renaissance Pharma licenses Hu14.18
Pharmaceutical Technology
JULY 4, 2022
Brii Biosciences (Brii Bio) has exercised an option for the acquisition of exclusive development and marketing rights for Vir Biotechnology’s investigational antibody, VIR-3434, for Hepatitis B in Greater China, under a partnership agreement. The mAb is presently in the Phase II development stage.

BioPharma Reporter
APRIL 2, 2024
Ipsen is expanding its oncology pipeline by securing the global licensing rights to an antibody-drug conjugate (ADC) for solid tumors from Sutro Biopharma, its first drug in the category.
Pharmaceutical Technology
DECEMBER 26, 2022
LegoChem Biosciences and Amgen have signed a multi-target research collaboration and license agreement to develop antibody-drug conjugates (ADC). Till date, the company has signed a total of 12 ADC licensing deals, worth more than $5bn.

BioSpace
OCTOBER 21, 2021
The license and collaboration agreement allows Janssen to use F-star’s proprietary technology in the research, development and commercialization of novel bispecific antibodies.
Pharmaceutical Technology
OCTOBER 19, 2022
Syncromune and Biocytogen Pharmaceuticals’ wholly owned subsidiary Eucure Biopharma have entered an exclusive global licence agreement for OX40 antibody YH002 and two other active ingredients. . Syncromune president and CEO Eamonn Hobbs said: “We are excited to enter into an exclusive licensing agreement with Eucure/Biocytogen.
Drug Discovery World
JULY 21, 2023
Cancer Research Horizons has signed a collaboration agreement to license the entire Twist Biopharma Solutions Library of Libraries. The post Partnership could enhance antibody discovery for cancer research appeared first on Drug Discovery World (DDW).
Pharmaceutical Technology
MARCH 28, 2023
On March 27, OncoVerity announced the acquisition of worldwide licensing rights of the anti-CD70 monoclonal antibody cusatuzumab from the biotech argenx. Alongside this, the company closed a $30 million Series A, which came from both argenx and the venture of UCHealth and the University License Equity Holdings.

BioSpace
DECEMBER 21, 2022
Merck entered into a license and collaboration agreement with Chinese company Kelun-Biotech to develop seven antibody-drug conjugates candidates against oncology targets.

Pharmaceutical Technology
FEBRUARY 14, 2023
Precision oncology firm Corbus Pharmaceuticals and CSPC Megalith Biopharmaceutical have entered an exclusive licensing agreement for the latter’s new clinical stage antibody drug conjugate (ADC), CRB-701 (SYS6002). CSPC Megalith is a subsidiary of CSPC Pharmaceutical Group.
Bio Pharma Dive
APRIL 13, 2023
The program is part of Denali’s “transport vehicle” platform, which uses drugmaking technology to shepherd large therapeutic molecules like antibodies across the so-called blood-brain barrier.

Pharmaceutical Technology
JANUARY 6, 2023
Danish biotech company Adcendo has entered into a licence agreement with Duality Biologics on the next-generation antibody-drug conjugates (ADC) platform. The post Adcendo and Duality Biologics sign license deal for ADC platform appeared first on Pharmaceutical Technology.

Pharmaceutical Technology
SEPTEMBER 16, 2022
The US Food and Drug Administration (FDA) has granted fast track designation to Jasper Therapeutics’ antibody, JSP191, for the treatment of severe combined immunodeficiency (SCID) patients who undergo allogeneic hematopoietic stem cell transplant. . The antibody was also analysed in a total of more than 110 healthy subjects and patients. .

BioPharma Reporter
OCTOBER 26, 2023
The biopharmaceutical industry witnessed a 400% growth in total deal value of antibody-drug conjugate (ADC) licensing agreements from 2017-2022 and reached a peak of $16.6 billion in 2022, reveals data and analytics company GlobalData.

Drug Discovery World
JUNE 1, 2023
Antibody-drug conjugates (ADCs) are biopharmaceutical products in which a monoclonal antibody (mAB) is linked to a small molecule drug with a stable linker 1. To overcome this challenge, Synaffix has developed an innovative technology platform that can rapidly and efficiently convert any antibody into an ADC.
Drug Discovery World
NOVEMBER 2, 2023
This brought us to intersect with AION Labs, which had issued a call for applicants to launch a new company leveraging AI-based solutions for the computational de novo design of high-affinity antibodies for targeted therapies. RA: How laborious and expensive are current antibody discovery efforts? KS: In short, incredibly so.

BioPharma Reporter
SEPTEMBER 2, 2021
Anti-CD25 antibodies have emerged as one of the most promising next-generation cancer therapeutics, says US plant-based manufacturer of therapeutics and vaccines, iBio, as it enters the field.
BioSpace
MARCH 14, 2024
Hansoh Pharma is licensing Biotheus’ anti-EGFR/cMet bispecific antibody to develop antibody-drug conjugates. The Chinese biotechs are broadening their collaboration.

BioTech 365
MAY 10, 2021
Hubrecht Institute Licenses Flow Eighteen38 Reagent Antibodies for Cancer Research Hubrecht Institute Licenses Flow Eighteen38 Reagent Antibodies for Cancer Research Antibodies generated by Flow Eighteen38 to be used as research tools by Hubrecht Institute.
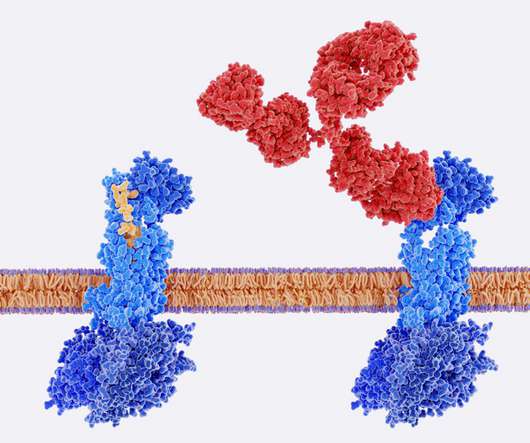
BioPharma Reporter
JUNE 1, 2021
Corbus Pharmaceuticals announced the expansion of its portfolio into immuno-oncology through licensing deals with the University of California San Francisco and Panorama Research Inc for two new monoclonal antibodies (mAbs).

pharmaphorum
MAY 18, 2022
Brand new UK startup RQ Biotechnology has been thrust into the spotlight after signing a $157 million licensing deal with AstraZeneca for monoclonal antibodies intended to protect vulnerable and immunosuppressed people from COVID-19. ” The post AstraZeneca buys into startup RQ Bio’s COVID antibodies appeared first on. .

BioPharma Reporter
JANUARY 6, 2023
Synaffix, a Netherlands based company providing clinical-stage platform technology for the development of antibody-drug conjugates (ADCs), has signed off on two new licensing deals this week.

Bio Pharma Dive
JANUARY 7, 2022
Neurimmune, which will license to AstraZeneca an early-stage treatment for a rare form of cardiomyopathy, is probably best known for its invention of the antibody that became Biogen's Alzheimer's drug Aduhelm.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?




Let's personalize your content