GSK antibody drug reduces COPD attacks in trial
Bio Pharma Dive
SEPTEMBER 6, 2024
New trial results could offer support for an expansion of Nucala’s label after U.S. regulators rejected GSK’s submission in 2018.

Bio Pharma Dive
SEPTEMBER 6, 2024
New trial results could offer support for an expansion of Nucala’s label after U.S. regulators rejected GSK’s submission in 2018.

Drug Discovery World
AUGUST 9, 2024
The first patient has been dosed in a Phase I clinical trial of AGX101, a novel TM4SF1-directed antibody-drug conjugate being developed for the treatment of solid cancers. We look forward to evaluating AGX101 in this trial.”
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
MAY 31, 2024
Just one week after Summit Therapeutics and Akeso’s bispecific antibody ivonescimab passed muster with Chinese regulators, the drug has pulled off another feat by staring down the PD-1 king Keytrud | In the phase 3 HARMONi-2 trial, solo ivonescimab delivered a statistically significant and clinically meaningful improvement in progression-free survival (..)

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.

Drug Discovery World
JULY 2, 2024
The National Institutes of Health (NIH) is sponsoring a clinical trial to evaluate the safety of an investigational monoclonal antibody to treat enterovirus D68 (EV-D68), which can cause severe respiratory and neurological diseases such as acute flaccid myelitis (AFM). Credit: NIAID and CDC.
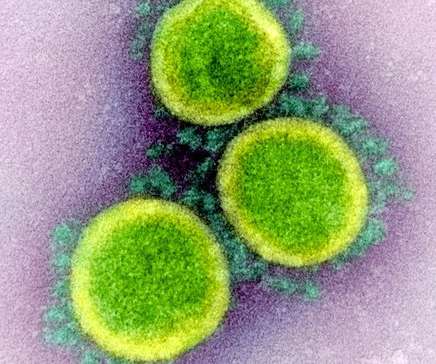
Bio Pharma Dive
JUNE 16, 2021
The biotech said it will seek an expanded FDA clearance after results from the RECOVERY trial showed its antibody can lower mortality among certain hospitalized COVID-19 patients.

Drug Discovery World
AUGUST 27, 2024
Skye Bioscience has started screening patients for a Phase II clinical trial (CBeyond) of its novel peripheral CB1 inhibitor, nimacimab, a negative allosteric modulating antibody. Assessing psychiatric effects of CB1 inhibition The clinical trial will enrol 120 patients across four treatment groups.
Let's personalize your content