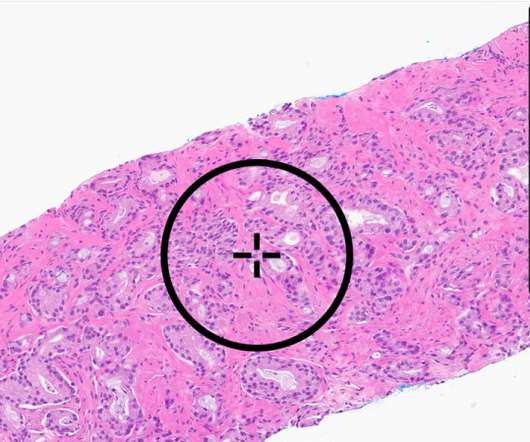
FDA clears AI-powered prostate cancer detection software
pharmaphorum
SEPTEMBER 22, 2021
The software – called Paige Prostate – is the first artificial intelligence-based to be approved by the FDA for this purpose, according to the US regulator. The results found that Paige Prostate – developed by Paige (formerly known as Paige.AI ) – improved detection of cancer on individual slide images by 7.3% That compared to 90.9%














Let's personalize your content