AI/ML-enabled Medical Devices Have Everyone’s Attention, Including FDA’s
FDA Law Blog
OCTOBER 16, 2023
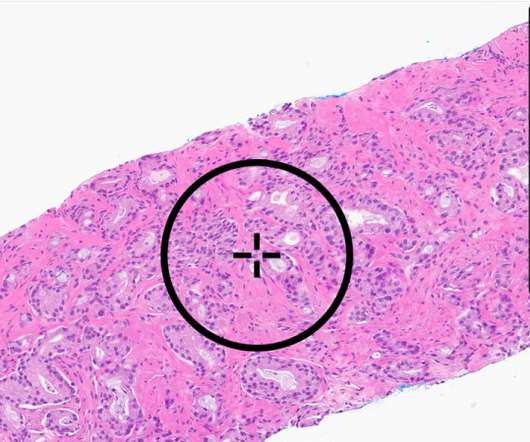
Three of these articles focus on medical applications of Artificial Intelligence and Machine Learning (AI/ML) and explore FDA’s role in regulating such products. Just a day earlier, FDA’s Center for Devices and Radiological Health (CDRH) released a list of the guidance documents the Center intends to publish in the upcoming 2024 fiscal year.










































Let's personalize your content