Novavax seeks FDA approval for updated Covid-19 vaccine
Pharmaceutical Technology
JUNE 17, 2024
Novavax has sought US FDA approval for an updated JN.1 1 version of its Covid-19 vaccine, NVX-CoV2705, for individuals aged 12 years and above.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
JUNE 17, 2024
Novavax has sought US FDA approval for an updated JN.1 1 version of its Covid-19 vaccine, NVX-CoV2705, for individuals aged 12 years and above.
Pharmaceutical Technology
MAY 10, 2024
The FDA approved WestGene’s mRNA therapeutic cancer vaccine as mRNA cancer vaccine development rises in popularity.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JUNE 3, 2024
Moderna has secured approval from the US Food and Drug Administration (FDA) for mRESVIA (respiratory syncytial virus vaccine).

Pharmaceutical Technology
JUNE 10, 2024
The US FDA approved GSK's Arexvy, an RSV vaccine for adults aged 50 to 59 years at higher lower respiratory tract disease (LRTD) risk.

Bio Pharma Dive
OCTOBER 23, 2023
The clearance of the pentavalent shot Penbraya adds to Pfizer’s infectious disease portfolio as it adjusts to slumping COVID-19 vaccine sales.
Pharmaceutical Technology
SEPTEMBER 12, 2023
The US FDA has approved Pfizer and BioNTech’s sBLA for Omicron XBB.1.5-adapted adapted monovalent Covid-19 vaccine, COMIRNATY 2023-2024 Formulation.
Bio Pharma Dive
MAY 31, 2023
The shot’s clearance comes several weeks after the regulator made GSK’s Arexvy the first vaccine for RSV in the U.S.

Pharmaceutical Technology
AUGUST 4, 2023
The US FDA has granted approval for an expanded indication of Merck’s Ebola vaccine, Ervebo, for usage in the paediatric population.
Pharmaceutical Technology
NOVEMBER 10, 2023
Following the three-month FDA review delay, Valneva’s live-attenuated chikungunya vaccine, Ixchiq received accelerated approval.

pharmaphorum
MAY 31, 2024
Moderna has secured FDA approval for its RSV vaccine, setting up a three-way market battle with GSK and Pfizer.
Bio Pharma Dive
MAY 7, 2021
The milestone filing could pave the way for the shot's use beyond the pandemic and give employers the legal heft to require vaccination, a key step toward herd immunity in the U.S.

Fierce Pharma
JUNE 17, 2024
The FDA has approved the world’s first pneumococcal disease vaccine designed for adults, signing off on Merck’s Capvaxive (formerly V116) and positioning it to become the primary shot used by senio | The FDA has approved the world’s first pneumococcal disease vaccine designed for adults, signing off on Merck’s Capvaxive (formerly V116) and positioning (..)
XTalks
JULY 25, 2023
Emergent BioSolutions, a multinational specialty biopharmaceutical company headquartered in Gaithersburg, Maryland, has achieved a significant milestone with the approval of its Cyfendus (Anthrax Vaccine Adsorbed, Adjuvanted) vaccine by the US Food and Drug Administration (FDA). How Does Cyfendus Work?
Fierce Pharma
OCTOBER 25, 2023
Five months after becoming the first company to secure FDA approval for a respiratory syncytial virus (RSV) vaccine, GSK is taking steps toward expanding its label for Arexvy.

Bio Pharma Dive
MAY 3, 2023
The decision represents the first fruits of a scientific breakthrough a decade ago that gave drugmakers, among them GSK, Pfizer and Moderna, a blueprint for an effective shot against the virus.
Bio Pharma Dive
SEPTEMBER 11, 2023
Pfizer and Moderna, which have seen slowing demand for their coronavirus vaccines, expect to make the reformulated shots available in the U.S. in the coming days.

Bio Pharma Dive
JULY 11, 2023
While the shot is approved in the EU, Takeda wasn’t able to address data collection issues raised by the US regulator in its current review cycle.

Pharma Times
SEPTEMBER 18, 2023
The updated vaccines more closely target current circulating variants - News - PharmaTimes

BioSpace
JUNE 21, 2022
On Wednesday, the FDA approved Merck's pneumococcal 15-valent conjugate vaccine for children 6 weeks through 17 years of age.

Fierce Pharma
NOVEMBER 28, 2023
Armed with a full FDA approval after four years supplying its anthrax vaccine under a pre-emergency use authorization, Emergent BioSolutions is expanding its countermeasure supply pact with the Uni | Armed with a full FDA approval after four years supplying its anthrax vaccine under a pre-emergency use authorization, Emergent BioSolutions is expanding (..)

Bio Pharma Dive
AUGUST 21, 2023
The expanded approval follows the FDA’s May clearance of the shot, called Abrysvo, in older adults.

Fierce Pharma
NOVEMBER 10, 2023
The FDA has approved the world’s first chikungunya vaccine, giving a thumbs-up to Valneva’s Ixchiq. The French company receives a priority review voucher from the FDA, which it said it will sell. It is an accelerated approval and subject to a confirmatory, real-world study. Valneva has won the race in the U.S. |

Drug Discovery World
JUNE 10, 2024
The US Food and Drug Administration (FDA) has approved mRESVIA (mRNA-1345), an mRNA respiratory syncytial virus (RSV) vaccine, to protect adults aged 60 years and older from lower respiratory tract disease caused by RSV infection. The vaccine uses the same lipid nanoparticles (LNPs) as the Moderna Covid-19 vaccines.

Fierce Pharma
OCTOBER 24, 2023
On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year. | On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year.
Drug Discovery World
JUNE 6, 2023
The US Food and Drug Administration (FDA) has approved Pfizer’s bivalent respiratory syncytial virus (RSV) prefusion F (RSVpreF) vaccine Abrysvo in individuals 60 years and older. A vaccine to help prevent RSV had been an elusive public health goal for more than half a century.
Pharmaceutical Technology
SEPTEMBER 15, 2022
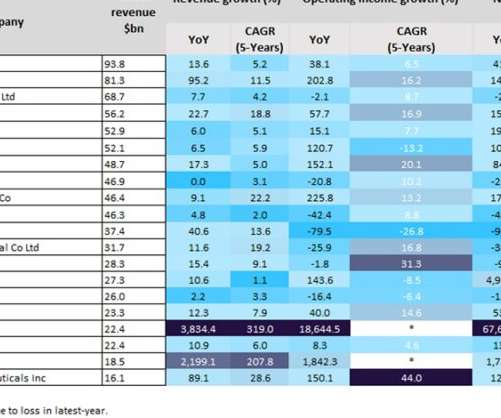
Last year was a positive year for biopharmaceutical companies, particularly those with Covid-19 vaccines. As a result of huge global sales of mRNA Covid-19 vaccines, the split in profits between Pfizer and BioNTech’s Comirnaty contributed towards revenues of $81.3bn and $22.4bn last year, respectively. YoY revenue growth.
XTalks
JULY 28, 2021
Merck has scored US Food and Drug Administration (FDA) approval for its next-generation pneumonia vaccine Vaxneuvance that covers 15 different strains of the pneumococcal bacteria that causes the infection. This is seven more strains than its current winning vaccine Prevnar 13, which registered $5.95

Pharmaceutical Technology
APRIL 28, 2023
The US Food and Drug Administration (FDA) has approved Pfizer’s 20-valent pneumococcal conjugate vaccine, PREVNAR 20 , to prevent invasive pneumococcal disease (IPD) in infants and children aged six weeks to 17 years. PREVNAR 20 has been developed on the basis of Pfizer’s approved PREVNAR 13 vaccine.

Drug Discovery World
AUGUST 23, 2023
The US Food and Drug Administration (FDA) has approved the first vaccine for pregnant individuals to prevent respiratory syncytial virus (RSV) in infants. The FDA approved Abrysvo in May for the prevention of LRTD caused by RSV in individuals 60 years of age and older.

Drug Discovery World
JUNE 6, 2024
Cancer vaccine mRNA-4157 (V940) in combination with Keytruda reduced the risk of recurrence or death by 49% in patients with resected high-risk melanoma, according to follow-up data presented at ASCO 2024. Ultimately it will contribute to survival rates improving continually over the next decades and more.” .

Pharmaceutical Technology
OCTOBER 28, 2022
On 24 October, American vaccine developer Vaxcyte shared positive topline data from a Phase I/II study of its multivalent conjugate pneumococcal vaccine VAX-24 , bringing the 24-valent pneumococcal jab one step closer to market. This is in addition to its long-available pneumococcal polysaccharide vaccine PPSV23.

BioSpace
JUNE 10, 2024
The FDA on Friday approved GSK’s application to use Arexvy to vaccinate adults aged 50 to 59 years who are at increased risk of developing severe respiratory syncytial virus.

JAMA Internal Medicine
APRIL 10, 2022
This survey study assesses vaccination intentions of unvaccinated US adults in response to full US Food and Drug Administration (FDA) approval of the BNT162b2 (Pfizer-BioNTech) COVID-19 vaccine and any demographic characteristics associated with those intentions.

Bio Pharma Dive
MARCH 29, 2021
A long-awaited reckoning for Biogen’s Alzheimer’s drug and the review of AstraZeneca’s coronavirus vaccine are among the top FDA decisions expected before the end of June.

Bio Pharma Dive
OCTOBER 1, 2021
Regulators face key decisions on COVID-19 shots for children and boosters for Moderna's and J&J's vaccines. Other closely watched drugs for multiple myeloma and depression are under review, too.

Fierce Pharma
JUNE 1, 2023
About a month after GSK won the world's first approval for a respiratory syncytial virus (RSV) vaccine, rival Pfizer has followed suit. After winning FDA approvals one month apart, Pfizer and GSK are set to launch competing RSV vaccines this fall.

BioSpace
JUNE 5, 2022
GSK's MMR vaccine is only the second to be approved in the United States for protection against measles, mumps and rubella. The first, licensed to Merck, was approved in 1971.

Pharmaceutical Technology
AUGUST 14, 2023
The PDUFA date has been pushed by three months to November to allow for more time for Phase IV activities.
Pharmaceutical Technology
AUGUST 23, 2022
Pfizer and BioNTech have filed an application seeking Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) for a booster dosage of an Omicron BA.4/BA.5-adapted 5-adapted bivalent vaccine for Covid-19 in people aged 12 years and above. 1-adapted vaccine. On obtaining the FDA EUA, the Omicron BA.4/BA.5-adapted

Bio Pharma Dive
SEPTEMBER 29, 2020
The world's attention will be on the FDA as it considers initial data from coronavirus vaccine developers. But several other important drugs, including a CAR-T therapy and an Ebola antibody, will also be on the agency's agenda.

XTalks
OCTOBER 31, 2023
GlaxoSmithKline (GSK) Canada has roped in both older and younger versions of Wayne Gretzky for a new ad for its respiratory syncytial virus (RSV) vaccine Arexvy. The RSV vaccine ad has the now 62-year-old hockey legend looking into a mirror and seeing his younger self, specifically an AI-generated 20-year-old version of himself.

Bio Pharma Dive
JUNE 30, 2022
But the drug may not meaningfully help people who are vaccinated, and concerns about its potency against emerging variants are growing. Converting Paxlovid’s emergency authorization to a standard clearance could further broaden its use.
BioSpace
MARCH 4, 2023
GSK Wins First-Ever FDA Approval for an RSV Vaccine 5/3/2023
XTalks
MAY 28, 2024
In a move signaling a major shift in influenza prevention strategies, CSL Seqirus, a global leader in flu vaccines, has transitioned from quadrivalent to trivalent formulations for the upcoming 2024/25 flu season in the US.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content