WestGene wins FDA approval for mRNA vaccine to treat certain cancers
Pharmaceutical Technology
MAY 10, 2024
The FDA approved WestGene’s mRNA therapeutic cancer vaccine as mRNA cancer vaccine development rises in popularity.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
MAY 10, 2024
The FDA approved WestGene’s mRNA therapeutic cancer vaccine as mRNA cancer vaccine development rises in popularity.

Bio Pharma Dive
OCTOBER 23, 2023
The clearance of the pentavalent shot Penbraya adds to Pfizer’s infectious disease portfolio as it adjusts to slumping COVID-19 vaccine sales.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Technology
SEPTEMBER 12, 2023
The US FDA has approved Pfizer and BioNTech’s sBLA for Omicron XBB.1.5-adapted adapted monovalent Covid-19 vaccine, COMIRNATY 2023-2024 Formulation.
Bio Pharma Dive
MAY 31, 2023
The shot’s clearance comes several weeks after the regulator made GSK’s Arexvy the first vaccine for RSV in the U.S.

Pharmaceutical Technology
AUGUST 4, 2023
The US FDA has granted approval for an expanded indication of Merck’s Ebola vaccine, Ervebo, for usage in the paediatric population.
Pharmaceutical Technology
NOVEMBER 10, 2023
Following the three-month FDA review delay, Valneva’s live-attenuated chikungunya vaccine, Ixchiq received accelerated approval.
Bio Pharma Dive
SEPTEMBER 11, 2023
Pfizer and Moderna, which have seen slowing demand for their coronavirus vaccines, expect to make the reformulated shots available in the U.S. in the coming days.

Bio Pharma Dive
MAY 3, 2023
The decision represents the first fruits of a scientific breakthrough a decade ago that gave drugmakers, among them GSK, Pfizer and Moderna, a blueprint for an effective shot against the virus.
XTalks
JULY 25, 2023
Emergent BioSolutions, a multinational specialty biopharmaceutical company headquartered in Gaithersburg, Maryland, has achieved a significant milestone with the approval of its Cyfendus (Anthrax Vaccine Adsorbed, Adjuvanted) vaccine by the US Food and Drug Administration (FDA). How Does Cyfendus Work?

Bio Pharma Dive
JULY 11, 2023
While the shot is approved in the EU, Takeda wasn’t able to address data collection issues raised by the US regulator in its current review cycle.
Bio Pharma Dive
MAY 7, 2021
The milestone filing could pave the way for the shot's use beyond the pandemic and give employers the legal heft to require vaccination, a key step toward herd immunity in the U.S.
Fierce Pharma
OCTOBER 25, 2023
Five months after becoming the first company to secure FDA approval for a respiratory syncytial virus (RSV) vaccine, GSK is taking steps toward expanding its label for Arexvy.

Bio Pharma Dive
AUGUST 21, 2023
The expanded approval follows the FDA’s May clearance of the shot, called Abrysvo, in older adults.
Drug Discovery World
NOVEMBER 13, 2023
The US Food and Drug Administration (FDA) has approved Ixchiq, Valneva’s single-dose, live-attenuated vaccine for the prevention of disease caused by chikungunya virus (CHIKV). The vaccine is indicated in individuals 18 years of age and older who are at increased risk of exposure to CHIKV.

BioSpace
JUNE 21, 2022
On Wednesday, the FDA approved Merck's pneumococcal 15-valent conjugate vaccine for children 6 weeks through 17 years of age.
XTalks
NOVEMBER 15, 2023
France-based biotech Valneva has won approval from the US Food and Drug Administration (FDA) for its chikungunya vaccine Ixchiq for the prevention of infection from the chikungunya virus. The vaccine is approved for adults 18 years of age and older who are at increased risk of exposure to the virus.

Pharma Times
SEPTEMBER 18, 2023
The updated vaccines more closely target current circulating variants - News - PharmaTimes
Drug Discovery World
JUNE 6, 2023
The US Food and Drug Administration (FDA) has approved Pfizer’s bivalent respiratory syncytial virus (RSV) prefusion F (RSVpreF) vaccine Abrysvo in individuals 60 years and older. A vaccine to help prevent RSV had been an elusive public health goal for more than half a century.
XTalks
JULY 28, 2021
Merck has scored US Food and Drug Administration (FDA) approval for its next-generation pneumonia vaccine Vaxneuvance that covers 15 different strains of the pneumococcal bacteria that causes the infection. This is seven more strains than its current winning vaccine Prevnar 13, which registered $5.95
Pharmaceutical Technology
JUNE 14, 2023
All six treatments are US Food and Drug Administration (FDA) approved. Blue Water bought the FDA-approved benign prostatic hyperplasia treatment from Veru for a total cost of $100m in April 2023. “We At market open, shares in Blue Water surged 90% higher than the previous day’s (13 June) market close.
XTalks
DECEMBER 7, 2021
VBI Vaccines got its first vaccine approval from the US Food and Drug Administration (FDA) for its hepatitis B shot PreHevbrio. PreHevbrio is approved for the prevention of hepatitis B infection caused by the hepatitis B virus (HBV) in adults 18 years of age and older.

Bio Pharma Dive
MARCH 29, 2021
A long-awaited reckoning for Biogen’s Alzheimer’s drug and the review of AstraZeneca’s coronavirus vaccine are among the top FDA decisions expected before the end of June.

Bio Pharma Dive
OCTOBER 1, 2021
Regulators face key decisions on COVID-19 shots for children and boosters for Moderna's and J&J's vaccines. Other closely watched drugs for multiple myeloma and depression are under review, too.

Drug Discovery World
AUGUST 23, 2023
The US Food and Drug Administration (FDA) has approved the first vaccine for pregnant individuals to prevent respiratory syncytial virus (RSV) in infants. The FDA approved Abrysvo in May for the prevention of LRTD caused by RSV in individuals 60 years of age and older.

Fierce Pharma
NOVEMBER 10, 2023
The FDA has approved the world’s first chikungunya vaccine, giving a thumbs-up to Valneva’s Ixchiq. The French company receives a priority review voucher from the FDA, which it said it will sell. It is an accelerated approval and subject to a confirmatory, real-world study. Valneva has won the race in the U.S. |

JAMA Internal Medicine
APRIL 10, 2022
This survey study assesses vaccination intentions of unvaccinated US adults in response to full US Food and Drug Administration (FDA) approval of the BNT162b2 (Pfizer-BioNTech) COVID-19 vaccine and any demographic characteristics associated with those intentions.

Bio Pharma Dive
SEPTEMBER 29, 2020
The world's attention will be on the FDA as it considers initial data from coronavirus vaccine developers. But several other important drugs, including a CAR-T therapy and an Ebola antibody, will also be on the agency's agenda.

Fierce Pharma
NOVEMBER 28, 2023
Armed with a full FDA approval after four years supplying its anthrax vaccine under a pre-emergency use authorization, Emergent BioSolutions is expanding its countermeasure supply pact with the Uni | Armed with a full FDA approval after four years supplying its anthrax vaccine under a pre-emergency use authorization, Emergent BioSolutions is expanding (..)

Fierce Pharma
OCTOBER 24, 2023
On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year. | On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year.

Pharmaceutical Technology
AUGUST 14, 2023
The PDUFA date has been pushed by three months to November to allow for more time for Phase IV activities.

XTalks
DECEMBER 12, 2023
After receiving US Food and Drug Administration (FDA) approval for Fabhalta (iptacopan) last week for the treatment of the rare blood disorder paroxysmal nocturnal hemoglobinuria (PNH), Novartis presented trial data yesterday showing the drug’s promise in another indication.

Bio Pharma Dive
JUNE 30, 2022
But the drug may not meaningfully help people who are vaccinated, and concerns about its potency against emerging variants are growing. Converting Paxlovid’s emergency authorization to a standard clearance could further broaden its use.
BioSpace
MARCH 4, 2023
GSK Wins First-Ever FDA Approval for an RSV Vaccine 5/3/2023
Pharmaceutical Technology
APRIL 11, 2023
In August 2021, Zydus secured emergency use authorisation (EUA) from the Drugs Controller General of India for its Covid-19 deoxyribonucleic acid (DNA) plasmid vaccine, ZyCoV-D. The vaccine, which generates the SARS-CoV-2 viral spike protein on administration, induces the immune system’s cellular and humoral arm-mediated immune response.

BioSpace
JUNE 5, 2022
GSK's MMR vaccine is only the second to be approved in the United States for protection against measles, mumps and rubella. The first, licensed to Merck, was approved in 1971.

World of DTC Marketing
NOVEMBER 12, 2020
Pfizer did not take government money to develop a COVID vaccine and said the release of preliminary vaccine data was based on receiving the data on Monday. That, however, didn’t stop a conspiracy loving imbecile from blaming the FDA and Pfizer. Of these, 4 (19.0%) received Accelerated Approval.

Pharmaceutical Technology
APRIL 28, 2023
The US Food and Drug Administration (FDA) has approved Pfizer’s 20-valent pneumococcal conjugate vaccine, PREVNAR 20 , to prevent invasive pneumococcal disease (IPD) in infants and children aged six weeks to 17 years. PREVNAR 20 has been developed on the basis of Pfizer’s approved PREVNAR 13 vaccine.

XTalks
DECEMBER 8, 2021
Moderna’s CEO sparked concerns when he said the current vaccines likely won’t be effective against Omicron, while Pfizer’s CEO said vaccines for COVID-19 will likely be needed for many years. The team also talked about a new hepatitis B vaccine from VBI Vaccines that received FDA approval for adults.
Drug Discovery World
MARCH 26, 2024
It is approved in adults and adolescents (12 years of age and older weighing at least 40kg) who are moderate-to-severely immune compromised due to certain medical conditions or receipt of certain immunosuppressive medications or treatments and are unlikely to mount an adequate immune response to Covid-19 vaccination.

Bio Pharma Dive
JANUARY 18, 2023
Moderna plans to follow those companies in asking for FDA approval. The biotech’s results appear competitive to past data for RSV shots being developed by GSK and Pfizer.

Pharma Times
AUGUST 23, 2023
The decision allows Abrysvo to be given to those who are 32 to 36 weeks into their pregnancy - News - PharmaTimes
XTalks
APRIL 24, 2024
ImmunityBio’s Anktiva (N-803, or nogapendekin alfa inbakicept-pmln) along with the Bacillus Calmette-Guérin (BCG) vaccine has won US Food and Drug Administration (FDA) approval for the treatment of non-muscle invasive bladder cancer (NMIBC). Anktiva is mixed together with the BCG vaccine before administration.
Bio Pharma Dive
FEBRUARY 19, 2021
Testing showed the vaccine could be safely stored at normal freezer temperatures, rather than the roughly minus 70 degrees Celsius now specified. The change, if cleared by the agency, will help distribution.
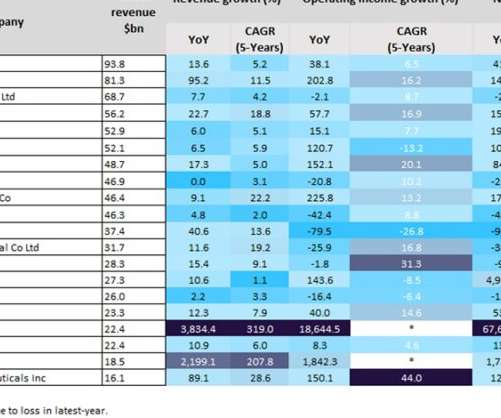
Pharmaceutical Technology
SEPTEMBER 15, 2022
Last year was a positive year for biopharmaceutical companies, particularly those with Covid-19 vaccines. As a result of huge global sales of mRNA Covid-19 vaccines, the split in profits between Pfizer and BioNTech’s Comirnaty contributed towards revenues of $81.3bn and $22.4bn last year, respectively. YoY revenue growth.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content