Sanofi signs $1.2bn licensing agreement for Novavax’s Covid-19 vaccine
Pharmaceutical Technology
MAY 10, 2024
Sanofi has signed a $1.2bn licensing agreement with Novavax to co-commercialise its stand-alone adjuvanted Covid-19 vaccine.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
MAY 10, 2024
Sanofi has signed a $1.2bn licensing agreement with Novavax to co-commercialise its stand-alone adjuvanted Covid-19 vaccine.

Bio Pharma Dive
OCTOBER 12, 2022
The drugmaker’s decision to grab rights to the shot deepens its ties with the COVID-19 vaccine developer and comes weeks before a key data release.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

pharmaphorum
MAY 10, 2024
Sanofi has licensed joint commercial rights to Novavax’s COVID-19 vaccine and will work with the biotech on the development of combined flu/COVID shots in a deal worth up to $1.2

Fierce Pharma
FEBRUARY 24, 2023
Moderna pays US government $400M 'catch-up payment' under new COVID-19 vaccine license esagonowsky Fri, 02/24/2023 - 09:18
Bio Pharma Dive
JULY 20, 2023
The startup will take on development of a shigellosis vaccine GSK inherited when it acquired LimmaTech’s predecessor, GlycoVaxyn, in 2015.
Pharmaceutical Commerce
DECEMBER 19, 2023
Merck’s Biologics License Application for V116, a 21-valent pneumococcal conjugate vaccine, was given a Prescription Drug User Fee Act (PDUFA) date of June 17, 2024.
Pharmaceutical Technology
APRIL 13, 2023
The expanded licence will include the development of vaccines and therapeutic proteins beyond Covid-19 for human and animal health markets in Africa. During the Covid-19 pandemic, vaccination rates of many countries in Africa were significantly trailing the rest of the world.

Pharmaceutical Technology
MAY 25, 2023
A new vaccine developed by the Serum Institute of India to fight meningococcal disease could help eliminate meningitis across Africa. The results from a trial, published in The New England Journal of Medicine , found the vaccine was associated with a strong immune response and good safety profile. percentage points (96% CI, −0.3

Pharmaceutical Technology
DECEMBER 2, 2022
India’s Central Drugs Standard Control Organisation (CDSCO) has granted approval for Bharat Biotech’s BBV154 vaccine for treating Covid-19. BBV154 is claimed to be the first intra-nasal vaccine for Covid-19 in the world.
Pharmaceutical Technology
MAY 16, 2023
Bavarian Nordic has completed the acquisition of Emergent BioSolutions ’ travel vaccine portfolio for an upfront payment of $274m, with the potential for the total value to rise to $384m. Following the completion of the deal, Bavarian Nordic now boasts the only oral vaccine licensed by the U.S.

Pharmaceutical Technology
AUGUST 1, 2022
GreenLight Biosciences has entered a partnership with the US National Institutes of Health (NIH) for the development of Covid-19 vaccines, which offer broader protection against new variants and with durable effects. They intend to develop vaccines that provide lasting immune responses compared to existing vaccines.
Pharmaceutical Technology
DECEMBER 6, 2022
Evaxion Biotech and ExpreS 2 ion Biotechnologies have entered a vaccine discovery collaboration agreement to co-develop a new cytomegalovirus (CMV) vaccine candidate. Until 2025, the companies will equally share the research and intellectual property licensing expenses under the partnership.

BioPharma Reporter
MAY 6, 2021
The next step will be to start a rolling submission for a Biologics License Application (BLA) for its COVID-19 vaccine with the FDA this month. Moderna has reported the first profitable quarter in the companyâs history: after 10 years of research and several billion dollars of investment in its mRNA platform.

STAT News
OCTOBER 12, 2022
Merck on Wednesday agreed to extend an ongoing collaboration with Moderna to develop a personalized vaccine for the treatment of patients with skin cancer. Moderna is getting $250 million from Merck to secure opt-in rights to the cancer vaccine candidate, called mRNA-4157.

BioPharma Reporter
DECEMBER 6, 2021
Australiaâs Noxopharm has in-licensed novel RNA tech developed by Hudson Institute of Medical Research: focusing on RNA drug discovery and mRNA vaccine manufacture via Noxopharmâs subsidiary Pharmorage.
World of DTC Marketing
SEPTEMBER 10, 2021
The government has the latitude to protect citizens from deadly conditions, especially when the science supporting vaccination is so clear” We, as a nation, can not allow ignorance and stupidity to endanger us all. It is reckless at this point for the government not to mandate vaccination.
STAT News
AUGUST 9, 2022
heath secretary recently declared monkeypox as a public health emergency , paving the way for emergency use authorizations of additional vaccines or vaccine regimens to fight the disease. The same day, Food and Drug Administration Commissioner Robert Califf said that the U.S. — by changing the way it is given.

BioPharma Reporter
FEBRUARY 21, 2023
The US Food and Drug Administration (FDA) has accepted Pfizerâs Biologics License Application (BLA) for its maternal RSV vaccine under priority review. If approved, the vaccine will become the first RSV vaccine for pregnant women to protect against RSV in infants.
Drug Discovery World
NOVEMBER 24, 2023
The University of Queensland’s re-engineered clamp platform has produced a vaccine that is equally safe and virus-neutralising as an approved Covid vaccine considered among the best in its class. He said: “CEPI is striving towards vaccines being developed within 100 days of a new virus emerging: a goal known as the 100 Days Mission.
Bio Pharma Dive
AUGUST 6, 2020
The Serum Institute, one of the world's largest vaccine producers, licensed Novavax's experimental shot, while Wockhardt signed on with the U.K. to help make AstraZeneca's candidate.

Fierce Pharma
OCTOBER 24, 2023
On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year. | On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year.
Bio Pharma Dive
MAY 13, 2022
The agreement, which covers three experimental vaccines as well as several key patents, will be royalty free for products sold in 49 low-income countries.

NY Times
AUGUST 20, 2021
Federal regulators are winding down the process of licensing Pfizer’s two-dose coronavirus vaccine, setting up an approval possibly by Monday and possibly kicking off a wave of new mandates.
Medical Xpress
FEBRUARY 3, 2023
A National Institutes of Health research group with extensive experience studying ebolavirus countermeasures has successfully developed a vaccine against Sudan virus (SUDV) based on the licensed Ebola virus (EBOV) vaccine. The new vaccine, VSV-SUDV, completely protected cynomolgus macaques against a lethal SUDV challenge.

BioSpace
JUNE 7, 2023
Promosome filed lawsuits Tuesday against Moderna and Pfizer/BioNTech, alleging that the vaccine developers used patent-protected mRNA technology without a license.
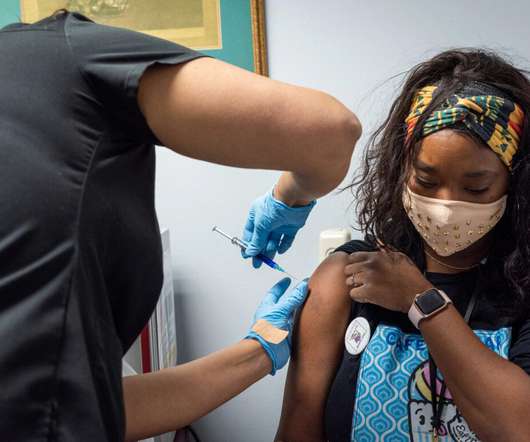
Pharmaceutical Technology
AUGUST 14, 2022
Novavax and its partner SK bioscience have obtained a Post Approval Change Application approval from the Korean Ministry of Food and Drug Safety (KMFDS) for the Covid-19 vaccine, Nuvaxovid (NVX-CoV2373), to be used in adolescents of the age 12 to 17 years. It enrolled 2,247 adolescents across 73 US sites.
Pharmaceutical Commerce
MAY 10, 2024
Parties sign co-exclusive licensing agreement, which includes a minority equity investment in Novavax, who will be receiving payments totaling as high as $1.2 billion when combining upfront payment and milestones.

pharmaphorum
NOVEMBER 23, 2021
Sanofi has bolstered its push into mRNA-based therapies with a new licensing deal – but not as might be expected with some up-and-coming biotech company. Sanofi is the first pharma company to partner with Baidu on its LinearDesign AI platform, which has been developed specifically to optimise the sequence of mRNA-based vaccines and drugs.

BioSpace
JULY 12, 2023
Following the regulator’s request for more data beyond the scope of its Phase III TIDES trial, Takeda decided to voluntarily withdraw the Biologics License Application for its TAK-003 dengue vaccine.

XTalks
FEBRUARY 19, 2024
Moderna has been testing an experimental mRNA vaccine aimed at combating the human cytomegalovirus (CMV), which poses significant health risks to newborns and immunocompromised patients. The team used the data and patient samples from a gB/MF59 Phase II trial in adolescent girls as a benchmark to assess the new mRNA-based vaccine.

BioPharma Reporter
MAY 7, 2021
Pfizer and BioNTech have started a rolling submission for a Biologics License Application (BLA) in the US for their COVID-19 vaccine.
BioPharma Reporter
APRIL 18, 2023
Dyadic extends its licensing agreement with Rubic for the use of its C1-cell protein expression platform.
BioPharma Reporter
DECEMBER 8, 2022
The US Food and Drug Administration (FDA) has accepted Pfizerâs RSV vaccine Biologics License Application (BLA) for priority review: with a decision expected in May.

STAT News
APRIL 24, 2023
Marburg could soon become the second virus in the past year to have experimental vaccine candidates ready for testing under an emergency use listing. Until this point, no licensed vaccines or treatments are available for Marburg. Read the rest…
BioPharma Reporter
APRIL 13, 2023
The R21/Matrix-M malaria vaccine, developed by the University of Oxford, has received its first regulatory clearance.

Drug Discovery World
FEBRUARY 20, 2024
A five-year contract totalling up to $31 million including programme options has been awarded to Ginkgo Bioworks to discover and develop next-generation vaccine adjuvants. Licensing for human use Adjuvants are components of vaccines that help to enhance the magnitude, breadth, and duration of the immune response to vaccination.
Pharmaceutical Technology
AUGUST 23, 2022
5-adapted bivalent vaccine for Covid-19 in people aged 12 years and above. 1-adapted vaccine. The FDA also sought the vaccine’s pre-clinical and manufacturing data for addressing the SARS-CoV-2 virus’ evolution. 5-adapted bivalent vaccine will be made readily available for shipping. 5-adapted bivalent vaccine.

BioTech 365
JANUARY 20, 2021
Gritstone and Genevant Sciences Announce License Agreement for COVID-19 Vaccine Gritstone and Genevant Sciences Announce License Agreement for COVID-19 Vaccine — Deal provides nonexclusive access to Genevant’s leading LNP technology for use in Gritstone’s self-amplifying RNA COVID-19 vaccine program; NIH-sponsored Phase … Continue reading → (..)
Pharmaceutical Technology
AUGUST 26, 2022
Amid a shortage of monkeypox vaccine doses, the WHO and FDA have declared public health emergencies, and the FDA has granted its first Emergency Use Authorization (EUA) to a monkeypox vaccine and opened the door for more. On the same day, the FDA granted a EUA to Bavarian Nordic’s (Copenhagen, Denmark) Jynneos vaccine.

Pharmaceutical Technology
OCTOBER 28, 2022
On 24 October, American vaccine developer Vaxcyte shared positive topline data from a Phase I/II study of its multivalent conjugate pneumococcal vaccine VAX-24 , bringing the 24-valent pneumococcal jab one step closer to market. This is in addition to its long-available pneumococcal polysaccharide vaccine PPSV23.
Drug Discovery World
JANUARY 16, 2024
A new study will compare whether giving tuberculosis vaccine by inhalation is better at protecting against tuberculosis (TB) than injection into the skin. The Jenner Institute at the University of Oxford is conducting the study using Bacille Calmette-Guérin (BCG), the current licensed vaccine against TB.
Drug Discovery World
MARCH 27, 2024
The vaccines currently in late-stage development hold promise in tackling the disease burden of tuberculosis (TB), says data and analytics company GlobalData on World Tuberculosis Day 2024. According to GlobalData, there are 11 vaccines in late-stage development for TB globally. TB remains a global pandemic, with 1.8

The Pharma Data
DECEMBER 2, 2020
This License Agreement represents an important corporate milestone. The License provides EYAM with the exclusive right to develop several proprietary COVID-19 vaccine candidates in a self-amplifying mRNA vaccine platform. EYAM anticipates rapid commencement of animal trials for these COVID-19 vaccine candidates.

BioSpace
JUNE 5, 2022
GSK's MMR vaccine is only the second to be approved in the United States for protection against measles, mumps and rubella. The first, licensed to Merck, was approved in 1971.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content