Twist and Cancer Research Horizons sign antibody library platform deal
Pharmaceutical Technology
JULY 19, 2023
Twist Bioscience and CRUK innovation arm Cancer Research Horizons have signed an agreement for licensing a library of libraries.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
APRIL 21, 2023
Twist Bioscience and Astellas Pharma have entered a collaboration to help the latter to discover antibodies for immunotherapies. In May 2022, they signed a research partnership and exclusive option licence agreement for the development of antibodies to reduce tumour microenvironment-mediated immunosuppression.
Drug Discovery World
JULY 21, 2023
Cancer Research Horizons has signed a collaboration agreement to license the entire Twist Biopharma Solutions Library of Libraries. The post Partnership could enhance antibody discovery for cancer research appeared first on Drug Discovery World (DDW).

Pharmaceutical Technology
OCTOBER 21, 2022
AbbVie has announced the acquisition of UK-based biotechnology firm DJS Antibodies for nearly $255m in cash at closing. DJS focuses on the discovery and development of antibody therapies that act on difficult-to-drug disease-causing proteins, such as G protein-coupled receptors (GPCRs).

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.
Pharmaceutical Technology
JULY 1, 2022
Ildong Pharmaceutical will licence a suite of Twist Bioscience's VHH antibody libraries after the two companies entered a partnership agreement. Twist's VHH libraries, used for discovering and developing antibodies for use in immuno-oncology, will be licensed by Ildong for three years for research and development works. .
Drug Discovery World
AUGUST 2, 2023
Renaissance Pharma has announced its first development programme focused on Hu14.18, a humanised anti-GD2 monoclonal antibody (mAb), licensed from St Jude Children’s Research Hospital for the treatment of newly diagnosed high-risk neuroblastoma. ” The post Renaissance Pharma licenses Hu14.18
Pharmaceutical Technology
DECEMBER 26, 2022
LegoChem Biosciences and Amgen have signed a multi-target research collaboration and license agreement to develop antibody-drug conjugates (ADC). Till date, the company has signed a total of 12 ADC licensing deals, worth more than $5bn.

BioSpace
OCTOBER 21, 2021
The license and collaboration agreement allows Janssen to use F-star’s proprietary technology in the research, development and commercialization of novel bispecific antibodies.

BioTech 365
MAY 10, 2021
Hubrecht Institute Licenses Flow Eighteen38 Reagent Antibodies for Cancer Research Hubrecht Institute Licenses Flow Eighteen38 Reagent Antibodies for Cancer Research Antibodies generated by Flow Eighteen38 to be used as research tools by Hubrecht Institute.
Pharmaceutical Technology
OCTOBER 18, 2022
Gilead Sciences has entered an exclusive option and partnership agreement with MacroGenics for developing bispecific antibodies. Under the deal, the companies will leverage MacroGenics’ DART platform to develop MGD024 as well as two further bispecific research programmes.

Drug Discovery World
JUNE 1, 2023
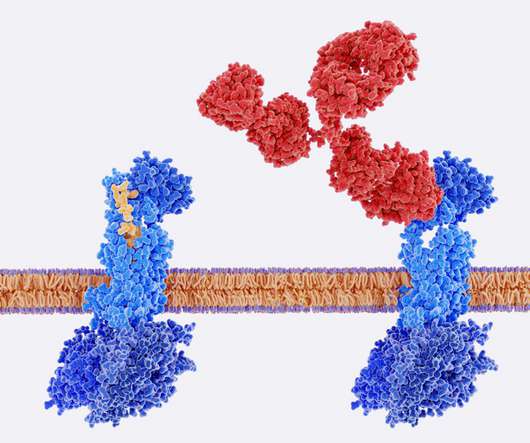
Antibody-drug conjugates (ADCs) are biopharmaceutical products in which a monoclonal antibody (mAB) is linked to a small molecule drug with a stable linker 1. To overcome this challenge, Synaffix has developed an innovative technology platform that can rapidly and efficiently convert any antibody into an ADC.
BioPharma Reporter
JUNE 1, 2021
Corbus Pharmaceuticals announced the expansion of its portfolio into immuno-oncology through licensing deals with the University of California San Francisco and Panorama Research Inc for two new monoclonal antibodies (mAbs).

BioPharma Reporter
AUGUST 1, 2023
Renaissance Pharma, a company focused on the development of life changing therapies in pediatric rare disease, has entered into an exclusive license agreement with St. Jude Childrenâs Research Hospital for Hu14.18, a humanised antibody in development by the hospital for the treatment of newly diagnosed high-risk neuroblastoma.

pharmaphorum
MAY 18, 2022
Brand new UK startup RQ Biotechnology has been thrust into the spotlight after signing a $157 million licensing deal with AstraZeneca for monoclonal antibodies intended to protect vulnerable and immunosuppressed people from COVID-19. ” The post AstraZeneca buys into startup RQ Bio’s COVID antibodies appeared first on. .
Drug Discovery World
JULY 26, 2023
A Cancer Research UK-funded clinical trial has shown, for the first time, that a new class of antibody could benefit cancer patients whose existing treatments have stopped working. The drug, MOv18 IgE, was developed by researchers at King’s College London.
pharmaphorum
DECEMBER 16, 2020
AbbVie is to begin clinical development of an antibody designed to neutralise the SARS-CoV-2 coronavirus after licensing the therapy in from Harbour BioMed and Utrecht University. AbbVie has begun a phase 1 clinical trial of the antibody, with clinical development beginning in the US and expanding into Europe.

The Pharma Data
JUNE 2, 2022
Boehringer Ingelheim and the Agency for Science, Technology and Research (A*STAR) today announced a global licensing agreement under which Boehringer Ingelheim will obtain exclusive worldwide rights to research, develop and commercialize products based on a panel of innovative, tumor-specific antibodies from A*STAR.

BioTech 365
MAY 4, 2021
Ablexis Announces Multi-Target License Agreement with Tanabe Research Laboratories U.S.A. Ablexis Announces Multi-Target License Agreement with Tanabe Research Laboratories U.S.A.

Drug Discovery World
NOVEMBER 10, 2023
Megan Thomas shines a spotlight on Adele Hannigan , CBO for Antibody Analytics. AH : I am Chief Business Officer for Antibody Analytics, a collaborative immunology CRO, specialising in supporting immunotherapy developers at the discovery to pre-clinical stages of the drug development process. MT: What has you career path looked like?

The Pharma Data
MARCH 31, 2023
AstraZeneca has completed an exclusive global license agreement with KYM Biosciences Inc. i for CMG901, a potential first-in-class antibody drug conjugate (ADC) targeting Claudin 18.2, Notes CMG901 CMG901 is a novel antibody drug conjugate targeting Claudin 18.2, a promising therapeutic target in gastric cancers.

BioSpace
OCTOBER 21, 2021
Atreca announced it is licensing its preclinical monoclonal antibody for the prevention of malaria to the Gates Medical Research Institute (MRI) for development.

BioTech 365
FEBRUARY 1, 2022
Abcam Collaborates With Twist Bioscience to Enhance Antibody Discovery for Diagnostic and Research Applications Abcam Collaborates With Twist Bioscience to Enhance Antibody Discovery for Diagnostic and Research Applications Abcam to license in VHH phage library(1) from Twist, complementing in-house recombinant … Continue reading →

The Pharma Data
JANUARY 11, 2021
a San Diego-based biotechnology company with an array of technology platforms for antibody discovery and optimization, and novel NK and T cell engager generation, today announced licensing of a panel of its anti-SARS-CoV-2 antibody clones to IGM Biosciences for COVID-19 therapy development.
Drug Discovery World
OCTOBER 11, 2023
Collaborators Boehringer Ingelheim and CDR-Life have commenced a Phase I evaluation of BI 771716, their antibody fragment-based treatment developed to preserve the vision of people with geographic atrophy (GA). The company’s first therapeutic candidate, CDR404, targets MAGE-A4 and is expected to enter the clinic in 2024.
Drug Discovery World
JULY 24, 2023
Talquetamab is a subcutaneous bispecific antibody that binds G protein-coupled receptor class C group 5 member D (GPRC5D), a novel target on multiple myeloma cells, and CD3, on T-cells. The post CHMP recommendation for bispecific antibody talquetamab appeared first on Drug Discovery World (DDW).
Drug Discovery World
OCTOBER 21, 2022
Under an exclusive agreement, Jazz Pharmaceuticals will acquire development and commercialisation rights to Zymeworks’ zanidatamab across all indications and all territories except for those Asia/Pacific territories previously licensed by Zymeworks. Zanidatamab is a HER2-targeted bispecific antibody with novel mechanisms of action.

Drug Discovery World
JANUARY 31, 2024
Onward Therapeutics has announced the Phase I clinical trial of OT-A201, a first-in-class bispecific antibody targeting two immune checkpoints is in progress.
Drug Discovery World
FEBRUARY 28, 2024
Lu Rahman selects some of the year’s interesting and noteworthy advances in cancer research drug discovery and development. In the UK, according to Cancer Research UK, around 167,000 people die from cancer in the UK each year. Research into new drugs and therapeutics is on-going and is helping drive mortality rates down.
Drug Discovery World
OCTOBER 28, 2022
A panel of investigational monoclonal antibodies (mAbs) targeting different sites of the Epstein-Barr virus (EBV) have been shown to block infection when tested in human cells in a laboratory setting. . There is no licensed vaccine to protect against the virus. . EBV is one of the most common human viruses.

Roots Analysis
JANUARY 21, 2024
With fourteen approved drugs and several drug candidates being evaluated under different stages of development, antibody drug conjugates (ADCs) are now recognized as a potent class of targeted therapeutics. In this context, the role of the linker molecule and the conjugation technology used is pivotal.

Pharmaceutical Technology
JANUARY 6, 2023
Dutch biotechnology company Synaffix and Amgen have entered a licensing agreement for the development of next-generation antibody-drug conjugates (ADCs). For four future programmes, Amgen will also have an option for exercising exclusive licenses for research and commercialisation.
The Pharma Data
MARCH 9, 2021
. (“Biolojic”), a biotechnology company that computationally designs functional antibodies, today announced a research collaboration and license agreement that will leverage Biolojic’s AI-based multibody platform to discover and develop a potential novel antibody-based therapy for the treatment of diabetes.
Drug Discovery World
JUNE 22, 2023
IOME Bio continues to collaborate with the Sharpe and Kasper labs through sponsored research and has an exclusive license to intellectual property stemming from their discoveries. Professor Gordon Freeman at the Dana-Faber Cancer Institute is also a key collaborator and contributor.

pharmaphorum
SEPTEMBER 2, 2020
The drug, JTX-1811, is a monoclonal antibody designed to selectively deplete immunosuppressive tumour-infiltrating T regulatory (TITR) cells. . When JTX-1811 binds to CCR8, it targets TITR cells for depletion by an enhanced antibody-dependent cellular cytotoxicity mechanism. . In 2016 Celgene signed a $2.6

Delveinsight
JANUARY 12, 2021
Medivir, IGM Biosciences enters into an exclusive licensing agreement for Birinapant. Medivir AB has entered into an exclusive licensing agreement with IGM Biosciences to receive global, exclusive development rights for Birinapant. AvantGen Enters into a Licensing Agreement for its Anti-SARS-CoV-2 Antibodies with IGM Biosciences.

pharmaphorum
JANUARY 27, 2021
AstraZeneca is playing catch up with Eli Lilly and Regeneron with its antibody therapy for COVID-19, but aims to narrow the gap with the help of decentralised clinical trial specialist Care Access Research. The two antibodies were discovered by Vanderbilt University Medical Center and licensed to AZ in June 2020.

pharmaphorum
JANUARY 29, 2021
US drugmaker Eli Lilly – still waiting for an FDA decision on one non-opioid pain drug – has just added another to its pipeline via a licensing agreement with Japan’s Asahi Kasei worth up to $410 million. While tanezumab is an antibody and has to be delivered by subcutaneous injection, AK1780 is orally bioavailable.

Pharmaceutical Technology
DECEMBER 19, 2022
With the latest development, Sanofi will licence a NK cell engager programme that acts on B7H3 from the antibody-based NK cell engager therapeutics (ANKET) platform of Innate. Initially, the companies signed a research collaboration and licensing agreement in 2016 to develop and assess up to two bispecific NK cell engagers.

Drug Discovery World
JULY 28, 2023
To coincide with the publication of the DDW eBook ‘ Antibody discovery: Breakthroughs accelerating therapeutic antibodies ’, our news highlights this week all focus on antibodies, particularly the recommendation by the EU’s CHMP for two bispecific antibodies that treat multiple myeloma and lymphoma.

XTalks
JULY 25, 2022
Byondis announced that the US Food and Drug Administration (FDA) accepted their Biologics License Application (BLA) for [vic-]trastuzumab duocarmazine (SYD985), an investigational therapy being studied to treat HER2-positive unresectable locally advanced or metastatic breast cancer. Vic-]Trastuzumab Duocarmazine Mechanism of Action.

Delveinsight
AUGUST 18, 2020
Sanofi had earlier licensed-in Principia’s experimental multiple sclerosis treatment ‘168; the late-stage pemphigus treatment Rilzabrutinib; and PRN473, a topical BTK inhibitor. Administered orally, Enspryng is a humanized monoclonal antibody. The drug is approved in Canada, Japan, and Switzerland for NMOSD.

The Pharma Data
DECEMBER 14, 2020
Harbour BioMed (HBM) announced with Utrecht University on Monday that they have licensed their fully human SARS-CoV-2 neutralizing antibody, 47D11, and its program to AbbVie. AbbVie has initiated a Phase I clinical trial of the antibody, and it will conduct the initial clinical program in the U.S.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?



Let's personalize your content