How to plan for uncertainty while maximizing the efficiency of your clinical sup…
The Pharma Data
SEPTEMBER 14, 2020
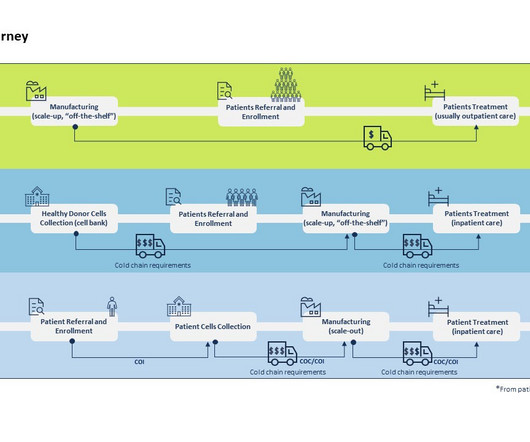
Supply chain uncertainty is a challenge every clinical supply chain manager wrestles with – stemming from increasing product demand and quality, the growing complexity of clinical trial designs and a lack of visibility in the supply chain. Accelerating your clinical trial supply through digitalization.















Let's personalize your content