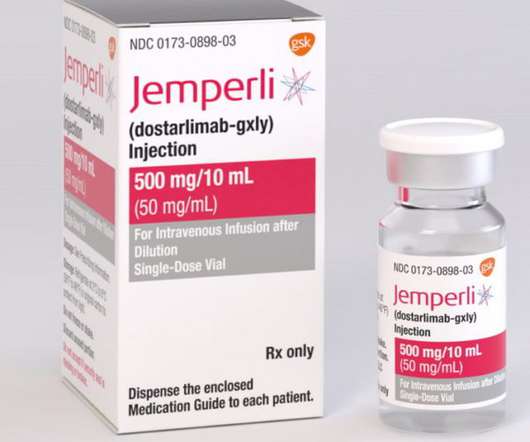
GSK receives FDA accelerated approval for JEMPERLI (dostarlimab-gxly) for adult patients with mismatch repair-deficient (dMMR) recurrent or advanced solid tumours
The Pharma Data
AUGUST 17, 2021
Mismatch repair-deficient tumours contain abnormalities that affect the proper repair of DNA when copied in a cell [i]. GSK is responsible for the ongoing research, development, commercialization, and manufacture of each of these Products under the Agreement. Initiate hormone replacement as clinically indicated.














Let's personalize your content