CanSino, Canada abandon plans for a coronavirus vaccine trial
Bio Pharma Dive
AUGUST 27, 2020
Canada was set to help CanSino produce its experimental shot as part of a deal to run what would've been the country's first coronavirus vaccine study.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 coronavirus
coronavirus 
Bio Pharma Dive
AUGUST 27, 2020
Canada was set to help CanSino produce its experimental shot as part of a deal to run what would've been the country's first coronavirus vaccine study.

Bio Pharma Dive
OCTOBER 6, 2020
The review criteria outlined in the document, which the White House had held up for weeks, make the early approval of a coronavirus shot before the Nov. 3 election less likely.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
OCTOBER 8, 2020
The biotech, a frontrunner in the coronavirus vaccine race, said it won't tie up resources defending the intellectual property covering its experimental shot as long as the pandemic continues.
Bio Pharma Dive
JANUARY 26, 2021
The French drugmaker, having hit delays with its own coronavirus shot, agreed to manufacture over 100 million doses of Pfizer and BioNTech's vaccine for supply in Europe.
Pharmaceutical Technology
FEBRUARY 15, 2023
In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Coronavirus vaccine components. Sanofi is the leading patent filer in coronavirus vaccine components.

Bio Pharma Dive
SEPTEMBER 17, 2020
The biotech, among the furthest along in coronavirus vaccine development, is the first developer to share the so-called study protocol of its large Phase 3 trial.
Bio Pharma Dive
NOVEMBER 11, 2020
The companies' early success helps validate an industry-wide bet to target the coronavirus' spike protein. But differences between each vaccine could still prove crucial.
Bio Pharma Dive
AUGUST 28, 2020
The reported talks between CanSino and several countries highlight the urgency with which governments are operating to secure supply of experimental coronavirus vaccines.

Bio Pharma Dive
NOVEMBER 12, 2020
Coronavirus' fast accelerating spread in the U.S. has sped up Moderna's timeline for its late-stage vaccine study, which now has sufficient COVID-19 cases for a first look at results.
Bio Pharma Dive
SEPTEMBER 15, 2020
The Serum Institute of India, a go-to partner for coronavirus vaccine makers, will help Novavax boost capacity to levels matching its larger rivals.
Bio Pharma Dive
NOVEMBER 17, 2020
Positive results from Pfizer and Moderna offer the strongest validation yet of researchers' approach to coronavirus vaccines, but critical questions remain.
Bio Pharma Dive
JANUARY 19, 2021
As worries about coronavirus mutations grow, Gritstone Oncology will work with the NIH to develop a shot capable of stimulating a broad immune response.

Bio Pharma Dive
NOVEMBER 23, 2020
The large drugmaker will pay $425 million to acquire OncoImmune, which is developing a treatment for severe coronavirus disease.
Bio Pharma Dive
SEPTEMBER 17, 2020
Both companies believe the messenger RNA technology they're using in their coronavirus vaccines could be applied to create a better flu shot.

Bio Pharma Dive
OCTOBER 20, 2020
The co-leader of an NIH network of coronavirus prevention studies spoke with BioPharma Dive about the FDA's big vaccine meeting this week and what will come next.
Bio Pharma Dive
AUGUST 13, 2020
The biotech, which has quickly advanced its coronavirus shot to late-stage trials, raised nearly $750 million through the listing.
Medical Xpress
JANUARY 26, 2023
The coronavirus keeps evolving, and in so doing it evades our immune defenses. But does the entire coronavirus evolve, or do some portions remain unchanged?

Bio Pharma Dive
MAY 24, 2021
One month after Lilly's first coronavirus antibody was pulled from market, the U.S. government stopped distributing its other treatment in two states.
Bio Pharma Dive
NOVEMBER 3, 2020
Clover Biopharmaceuticals, one of several China-based groups advancing a coronavirus shot, has now received one of the largest investments to date from the vaccine development alliance group CEPI.
Bio Pharma Dive
OCTOBER 28, 2020
The two drugmakers are aiding an international effort to ensure low- and middle-income countries can get access to successful coronavirus shots.
BioSpace
JUNE 28, 2022
BioNTech announced it is partnering with Pfizer to begin testing a next-generation universal vaccine against coronaviruses, including SARS-CoV-2, the coronavirus that causes COVID-19.

Pharmaceutical Technology
APRIL 15, 2024
Discover ImmunityBio Inc's groundbreaking patent for a cross-reactive SARS-CoV2 vaccine targeting multiple variants. Learn how this innovative method boosts immune response and memory cells for enhanced protection.
STAT News
JANUARY 26, 2023
The evolution of the coronavirus has knocked out another treatment. The Food and Drug Administration on Thursday withdrew the authorization of Evusheld, the latest antibody therapy to be rendered ineffective by the mutations the virus has picked up.

NPR Health - Shots
JANUARY 29, 2023
30, 2020, the World Health Organization declared the "novel coronavirus" sweeping through China to be a global health emergency. Here's how NPR covered the story at that point in time. Image credit: Stringer/Getty Images)
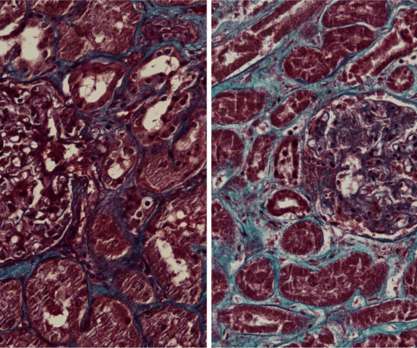
Scienmag
DECEMBER 27, 2021
The Coronavirus SARS-CoV-2 infects the kidneys and contributes to tissue scarring, as shown by researchers from the RWTH Uniklinik Aachen, Germany, and Radboudumc, The Netherlands. Credit: Jitske Jansen and Bart Smeets, Radboudumc The Coronavirus SARS-CoV-2 […].
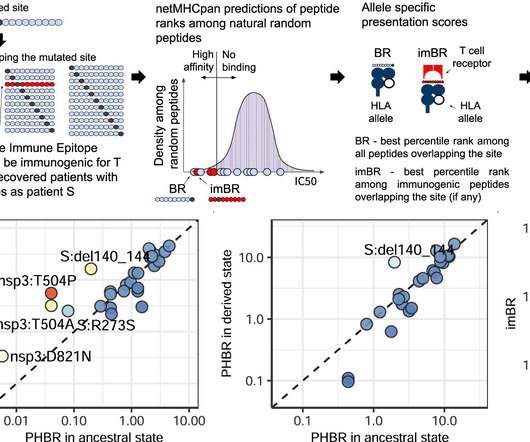
Medical Xpress
FEBRUARY 14, 2023
The immune system fights the coronavirus with antibodies and T cells. Scientists have a fairly good idea of how the virus avoids antibodies, including those promoted by vaccination, which is how variants of concern such as the omicron are known to appear. T cell evasion, on the other hand, has remained poorly understood.
Bio Pharma Dive
JANUARY 27, 2021
As coronavirus variants spread, the White House aims to up U.S. vaccine supply by exercising purchase options in contracts with Pfizer and Moderna.
Bio Pharma Dive
DECEMBER 16, 2021
Days after the FDA strengthened safety warnings for the company's shot, a CDC panel recommended Pfizer's and Moderna's vaccines be "preferred" over J&J's.
Medical Xpress
FEBRUARY 28, 2023
A team of scientists from Scripps Research and the University of North Carolina (UNC) has found antibodies in the blood of certain COVID-19 donors that can block infection from a broad set of coronaviruses—specifically, in people who have recovered from the virus and were then vaccinated.
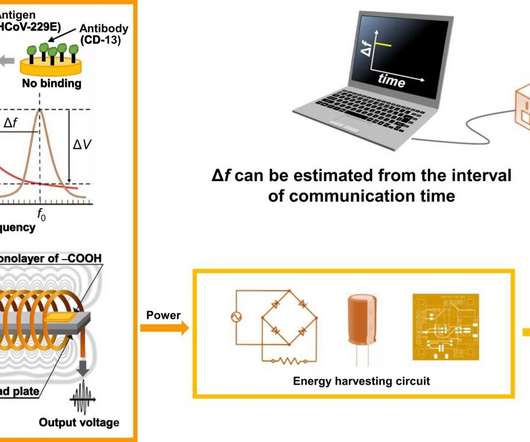
Medical Xpress
DECEMBER 2, 2022
A research group has engineered a batteryless, self-powering device that can wirelessly transmit the detection of coronavirus in the air. What if you could tell if your surroundings contained COVID-19 particles or droplets the moment they or you entered the vicinity? This is now closer to reality.
pharmaphorum
AUGUST 18, 2020
million euro project funded by European Union cash and European pharma companies is a work stream that aims to find drugs that work against coronavirus strains that may emerge in the future. The trick is to look for proteins that are used across all coronavirus types and tend not to vary much from one virus species to another.

Bio Pharma Dive
JANUARY 25, 2021
The drugmaker, one of the largest vaccine developers globally, won't move forward with either of two candidates it's been testing after disappointing data in early trials.

NY Times
NOVEMBER 21, 2020
The furious race to develop a coronavirus vaccine played out against a presidential election, between a pharmaceutical giant and a biotech upstart, with the stakes as high as they could get.
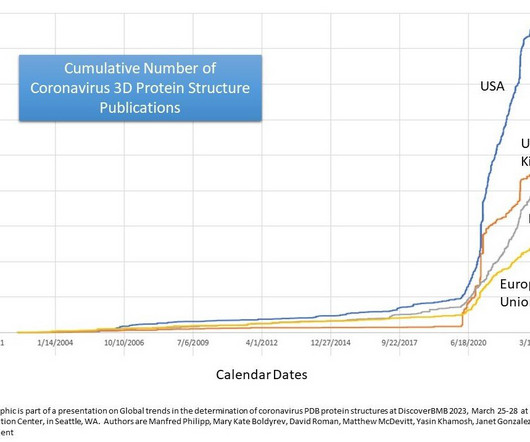
Medical Xpress
MARCH 27, 2023
In a new study, researchers examined how a country's number of published 3D protein structures for coronaviruses, including the one responsible for COVID-19, correlated with its economic output and population.

pharmaphorum
AUGUST 7, 2020
Pharma’s reputation has soared due to the COVID-19 pandemic – but coronavirus is starting to bite companies’ sales. Roche, AstraZeneca, Novo Nordisk, and Sanofi all shot up the rankings in the first edition of the report since the start of the coronavirus crisis, but the big loser was Gilead Sciences, falling 71 places to 74 in the list.

BioSpace
NOVEMBER 9, 2021
A research team at the University of North Carolina – Chapel Hill (UNC) and Duke University, in Durham, NC identified an antibody that attacks SARS-CoV-2 and its variants and other types of coronaviruses.
Bio Pharma Dive
FEBRUARY 27, 2021
The agency's emergency clearance makes J&J's vaccine the third available in the U.S., adding much-needed reinforcements at a critical time.

pharmaphorum
AUGUST 21, 2020
New insights into smell and taste loss symptoms, artificial intelligence and further encouraging data from a vaccine candidate hit the headlines this week as the COVID-19 coronavirus pandemic continues. Here we highlight the biggest R&D, market access and digital coronavirus news of the past week.

BioSpace
OCTOBER 27, 2020
Please check out the biopharma industry coronavirus (COVID-19) stories that are trending for October 27, 2020.
Bio Pharma Dive
MAY 10, 2021
The emergency clearance greatly expands the pool of people who can be vaccinated in the U.S. just as some states are beginning to report waning demand.
pharmaphorum
NOVEMBER 19, 2020
The coronavirus vaccine being developed by Oxford University and AstraZeneca produces a strong immune response in higher risk older patients, a study has found. The post Encouraging data from AZ/Oxford coronavirus shot appeared first on. Booster regimes were given 28 days apart and dosing regimen varied according to age group.
NPR Health - Shots
OCTOBER 4, 2022
There is a lot of information about coronavirus vaccines out there, and some of it seems vague or contradictory. We talked to experts to help answer some of the most common and confusing questions. Image credit: Stephanie Carter/Getty Images/Imagezoo)

BioSpace
AUGUST 20, 2020
Following approval of its coronavirus vaccine, the first in the world, Russia plans to initiate a new study involving 40,000 people as the country looks to boost production and distribute the drug to patients in that country, as well as to patients in countries it has struck deals with.
pharmaphorum
SEPTEMBER 14, 2020
AstraZeneca has resumed UK trials for its coronavirus vaccine, after the country’s medicines regulator gave the all-clear following a safety scare. The vaccine is top of the list of 35 potential coronavirus vaccines in clinical development, according to a list maintained by the World Health Organization.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content