Almirall and Microsoft partner for dermatological drug development
Pharmaceutical Technology
FEBRUARY 1, 2024
Almirall has entered a strategic collaboration with Microsoft to steer innovation and digital transformation in dermatology.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
FEBRUARY 1, 2024
Almirall has entered a strategic collaboration with Microsoft to steer innovation and digital transformation in dermatology.

Pharmaceutical Technology
NOVEMBER 15, 2023
Almirall has entered into a collaboration with Absci for the development and commercialisation of new treatments for dermatological ailments.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

pharmaphorum
FEBRUARY 1, 2024
Almirall has formed a three-year alliance with Microsoft designed to accelerate the discovery and development of personalised dermatology therapies.

Pharma Mirror
APRIL 8, 2022
BARCELONA, Spain and Dundee, UK, a global biopharmaceutical company focused on skin health, today announced a research collaboration with the University of Dundee, a top-ranked university in the UK in biological sciences, to develop a novel approach against chronic and debilitating dermatology diseases where itch, or pruritus, plays a dominant role.

pharmaphorum
OCTOBER 9, 2023
Champion Innovation at the 7th Dermatology Drug Development Summit Mike.Hammerton Mon, 09/10/2023 - 14:39 Bookmark this

pharmaphorum
MAY 9, 2023
4th Dermatology Drug Development Summit Europe Mike.Hammerton Tue, 05/09/2023 - 20:11 Bookmark this

Medical Xpress
JANUARY 24, 2023
A dermatology program first developed by the University of Missouri in 2015 can bring life-saving diagnoses of skin conditions to communities without adequate dermatologic care.
Drug Discovery World
MARCH 23, 2023
Aldena Therapeutics, a biotech company that is pioneering siRNA-based therapies for dermatological indications, has updated its immuno-dermatology pipeline, appointed a Scientific Advisory Board (SAB), and announced $30 million financing by Medicxi. appeared first on Drug Discovery World (DDW).

Outsourcing Pharma
MAY 23, 2022
With Veeva Link for Key People, LEO Pharma is looking to deepen its efforts to engage with knowledgeable medical professionals in the dermatology community.

pharmaphorum
NOVEMBER 11, 2021
KNUTSFORD, United Kingdom — Fishawack Health welcomes FIDE, an independent organization providing its clients with insight from clinical key opinion leaders in inflammatory dermatology. Kenneth B Gordon, MD, Professor and Chair of Dermatology at the Medical College of Wisconsin. November 10, 2021, UK.

WCG Clinical
SEPTEMBER 20, 2023
Welcome to the recorded session of ‘Optimizing Endpoint Assessments for Dermatology Studies.’ In this insightful webinar, we take a deep dive into dermatology trials and discuss how standardizing rater qualification and training can solve common challenges and protect the quality and integrity of endpoint data.

WCG Clinical
OCTOBER 9, 2023
The dermatology market is embarking on tremendous growth and sponsors need to be ready. Over the last 12 months, dermatology-focused studies have increased by roughly 20%, with top focus areas including psoriasis, atopic dermatitis, and alopecia. This rapid growth brings new challenges. Efficiency and speed are critical.

BioTech 365
DECEMBER 3, 2020
DermTech Presents Updates in Precision Medicine at 2020 Dermatology Drug Development Summit DermTech Presents Updates in Precision Medicine at 2020 Dermatology Drug Development Summit LA JOLLA, Calif.–(BUSINESS –(BUSINESS WIRE)–DermTech, Inc.
Drug Discovery World
MARCH 13, 2023
LEO Pharma and ICON have formed a strategic partnership that will enable LEO to offer clinical trials within medical dermatology that are patient-centric and cost effective. The partnership will operate under the acronym of PACE, reflecting the requirement to move quickly to address today’s clinical development challenges.
WCG Clinical
MARCH 30, 2023
WCG’s InvestigatorSpace® offers customizable training solutions to bring accurate and detailed training to life while reducing the time spent on developing assets by an average of 60%. WCG collaborated with study teams and clinical experts to develop the training modules.

BioTech 365
MAY 20, 2021
Pyramid Biosciences Announces First Patients Dosed in Phase 1 Clinical Program for Dermatology Pyramid Biosciences Announces First Patients Dosed in Phase 1 Clinical Program for Dermatology PBI-100 is a topical treatment being developed for people living with mild to moderate … Continue reading →

Outsourcing Pharma
DECEMBER 13, 2021
The pharmaceutical company currently has several promising treatments in development for gastroenterological, dermatological, and cardiological conditions.

Pharmaceutical Technology
JANUARY 19, 2023
Concert’s patent portfolio includes oral Janus kinases JAK1/2 inhibitor, deuruxolitinib, to treat Alopecia Areata, an autoimmune dermatological disease. The company also noted that the deal will strengthen its global dermatology franchise with the addition of a late-stage product in an area with significant unmet need.
Pharmaceutical Commerce
FEBRUARY 20, 2024
As part of the agreement, Novo will be paid both development and commercial milestone payments for NN-8828, is currently in Phase II of non-dermatological indications.
XTalks
AUGUST 2, 2023
Verrica Pharmaceuticals is a dermatology therapeutics company developing medications for skin diseases that require medical interventions. Tune into this episode to learn more about YCANTH and its approval, as well as the changing landscape of dermatology, from Dr. Eichenfield.

BioTech 365
SEPTEMBER 23, 2020
23, 2020 (GLOBE NEWSWIRE) — Verrica Pharmaceuticals Inc. (“Verrica”) (Nasdaq: VRCA), a dermatology therapeutics company developing medications for skin diseases requiring medical interventions, today announced that positive results from the two pivotal Phase III CAMP (Cantharidin … Continue reading →
Medical Xpress
MARCH 28, 2023
Adults with atopic dermatitis (AD) have an increased risk for developing melanoma, basal cell carcinoma (BCC), and squamous cell carcinoma (SCC), with significantly higher risks seen for moderate-to-severe versus mild AD, according to a study presented at the annual meeting of the American Academy of Dermatology, held from March 17 to 21 in New Orleans. (..)

Outsourcing Pharma
MARCH 14, 2023
A strategic partnership between LEO Pharma and ICON plc, a clinical development company, was announced on March 10.
Pharmaceutical Technology
APRIL 19, 2023
The drug is taken once a day and is currently being developed for the treatment of inflammatory dermatoses, mainly in hair-bearing body areas such as the face, the trunk and the scalp. The regulator has set 16 December 2023 as a prescription drug user fee act (PDUFA) target action date for the decision on the application.

Pharmaceutical Technology
FEBRUARY 28, 2023
KFA-115 is under clinical development by Novartis and currently in Phase I for Melanoma. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.
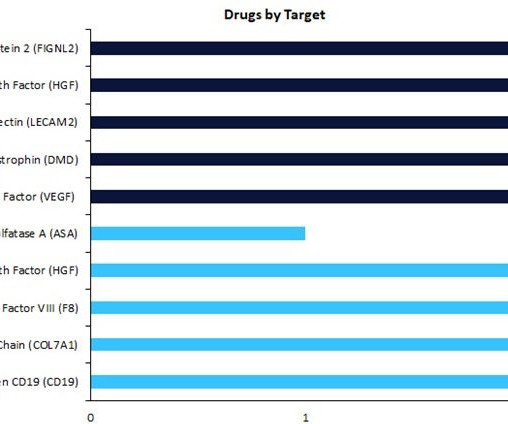
Pharmaceutical Technology
MARCH 27, 2023
Regenerative medicines in early-stage development (preclinical, discovery, or investigational new drug [IND]/ clinical trial application [CTA] filed status) have seen a change in drug targets compared to therapies in late-stage development (Phase II to pre-registration stage).

The Pharma Data
OCTOBER 29, 2020
30, 2020 /PRNewswire/ — Eli Lilly and Company (NYSE: LLY) and Incyte (NASDAQ: INCY) announced today new data for baricitinib (marketed as OLUMIANT ® ) will be presented at the annual Fall Clinical Dermatology meeting taking place virtually October 29-November 1, 2020. vice president of immunology development at Lilly.

pharmaphorum
APRIL 29, 2022
An artificial intelligence-powered digital tool for diagnosing skin cancers developed by Skin Analytics has been cleared in the UK as a Class IIA medical device, setting up broader use of the technology in patient assessments.
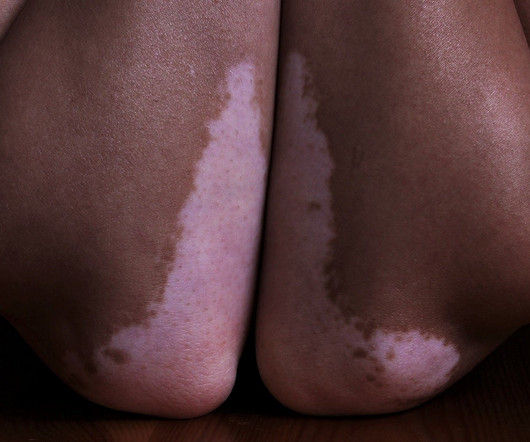
Pharmaceutical Technology
APRIL 17, 2023
The company will provide regulatory advice to Incannex, and manage clinical trials to develop CannQuit and ReneCann products to treat addiction and immune-disordered skin diseases. The company will help in managing the clinical trials of the products once the regulators provide approvals for the proposed research and development programmes.
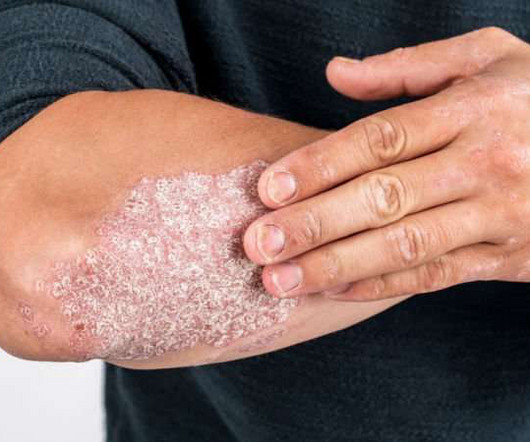
Drug Discovery World
MARCH 18, 2024
IL-23 plays a critical role in pathogenic T-cell activation in moderate-to-severe PsO and underpins the inflammatory response in PsO and other dermatological, rheumatological and gastroenterological IL-23-mediated diseases. JNJ-2113 is the first and only investigational targeted oral peptide designed to block the IL-23 receptor.

pharmaphorum
MARCH 24, 2021
After 27 years at Bayer, Jörg Möller has moved to LEO Pharma as the company’s EVP, global research and development. The post LEO Pharma’s new R&D lead on driving agility in drug development appeared first on. Is it a disease that has a high symptomatic disease burden, but is otherwise more of a nuisance?”
The Pharma Data
OCTOBER 1, 2021
The research, which spans clinical, health economics and outcomes research, translational, clinical pharmacology and preclinical presentations, highlights the breadth and depth of the company’s data on deucravacitinib, a first-in-class, oral, selective tyrosine kinase 2 (TYK2) inhibitor, also because the emerging dermatology pipeline.

XTalks
APRIL 17, 2023
These results are very promising for patients with atopic dermatitis,” said Jonathan Silverberg, MD, professor of dermatology at George Washington University School of Medicine & Health Sciences and co-investigator of the studies, in the company’s press release.

Pharmaceutical Technology
DECEMBER 5, 2022
Incyte has signed a partnership and licence agreement with CMS subsidiary CMS Aesthetics to develop and market ruxolitinib cream to treat autoimmune and inflammatory dermatologic ailments in Greater China and some Southeast Asian countries. Incyte will also receive royalty payments on the net product sales in the territory of CMS.

Pharmaceutical Technology
FEBRUARY 28, 2023
KFA-115 is under clinical development by Novartis and currently in Phase I for Malignant Mesothelioma. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.
pharmaphorum
SEPTEMBER 16, 2022
Atopic dermatitis (AD) is a highly prevalent chronic dermatological disease, with symptoms which include itching, dry skin, severe pain, and inflammation – all of which create considerable stress in the daily lives of patients and often negatively affect their day-to-day and long-term well-being. Findings presented at EADVC 2022.

ACRP blog
OCTOBER 3, 2023
Edited by Gary Cramer The post Internship Program Boosts Clinical Research Workforce Development in Pennsylvania appeared first on ACRP. Every department that had an intern this past summer is asking for the opportunity to have an intern next year, Gardner said, and other departments are asking to participate. “It
XTalks
APRIL 26, 2023
He began his career in business development and commercial strategy roles at Genentech. Poster Presentation, Innovations in Dermatology Conference, Las Vegas, NV, November 3-5, 2022. Dermatology. Dermatology. Hear more about the innovative technology and application of DermaSensor in this episode. 2004;208:27-31.

Pharmaceutical Technology
FEBRUARY 28, 2023
Iptacopan hydrochloride is under clinical development by Novartis and currently in Phase III for Paroxysmal Nocturnal Hemoglobinuria. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.

Pharmaceutical Technology
FEBRUARY 28, 2023
Iptacopan hydrochloride is under clinical development by Novartis and currently in Phase III for Glomerulonephritis. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.

Pharmaceutical Technology
FEBRUARY 24, 2023
(Lopinavir + ritonavir) is under clinical development by Douglas Pharmaceuticals and currently in Phase I for Genital Warts (Condylomata Acuminata). GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.

Pharmaceutical Technology
FEBRUARY 24, 2023
(Lopinavir + ritonavir) is under clinical development by Douglas Pharmaceuticals and currently in Phase I for Cervical Intraepithelial Neoplasia (CIN). GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data.

pharmaphorum
DECEMBER 1, 2021
The post Sanofi snaps up acne vaccine developer Origimm Bio appeared first on. It will highlight vaccines for pneumococcal disease, meningitis, respiratory syncytial virus (RSV), influenza, and chlamydia. Photo by Scott Webb on Unsplash.

pharmaphorum
SEPTEMBER 8, 2022
Furthering DiME’s previous work on digital clinical measures, nocturnal scratch’s more widespread implementation aims to establish a firmer digital groundwork for future clinical research, technology development, and reimbursement decisions in the dermatological field, and thereby improve the lives of AD patients and caregivers.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content