Cancer Research UK and CRIS fund lung cancer vaccine research
Pharmaceutical Technology
MARCH 25, 2024
Cancer Research UK and the CRIS Cancer Foundation have awarded a £1.7m ($2.1m) grant for developing the lung cancer vaccine LungVax.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
MARCH 25, 2024
Cancer Research UK and the CRIS Cancer Foundation have awarded a £1.7m ($2.1m) grant for developing the lung cancer vaccine LungVax.
Pharmaceutical Technology
JUNE 6, 2023
Oragenics has secured funding from the Canadian bioresearch consortium CQDM to develop a variant-agnostic protein antigen for use in its Covid-19 intranasal vaccine. The project is a partnership with Inspirevax and the National Research Council of Canada (NRC).
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
NOVEMBER 1, 2022
What is Research and Development (R&D)? This category includes organisations that have demonstrated a commitment to the research and development of new products and/or new processes. This category highlights companies that have conducted research into the use of AI and technology for the development of new treatments.

Pharmaceutical Technology
MARCH 20, 2023
RVAC Medicines has announced a research collaboration with the University of Pennsylvania (Penn) for the discovery and development of mRNA vaccines. The mRNA vaccine candidates will help reduce the chances of autoimmune responses that might lead to allergic conditions or serious autoimmune diseases.
Pharmaceutical Technology
OCTOBER 14, 2022
German pharmaceutical firm Merck has extended its partnership with Moderna to jointly develop and sell mRNA-4157/V940, an investigational personalised cancer vaccine (PCV). In 2016, the companies entered a strategic partnership to develop novel messenger RNA (mRNA) based PCVs.

Pharmaceutical Technology
NOVEMBER 25, 2022
XtalPi has entered a strategic partnership with CK Life Sciences for artificial intelligence (AI)-driven tumour vaccine research and development (R&D). Under the collaboration, the companies will utilise their capabilities to co-develop a new AI tumour vaccine R&D platform.

Pharmaceutical Technology
AUGUST 1, 2022
GreenLight Biosciences has entered a partnership with the US National Institutes of Health (NIH) for the development of Covid-19 vaccines, which offer broader protection against new variants and with durable effects. They intend to develop vaccines that provide lasting immune responses compared to existing vaccines.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 22, 2024
The Indian Institute of Science (IISc) is now developing a heat-tolerant vaccine that can offer protection against different strains of SARS-CoV-2 – both current and future variants. The research […]
Pharmaceutical Technology
MAY 18, 2023
International non-profit research organisation IAVI and Zendal Group’s subsidiary Biofabri have signed an expanded deal to develop MTBVAC, a tuberculosis (TB) vaccine candidate. The organisation and the company initially collaborated in 2021 to advance the vaccine candidate into efficacy trials.

Pharmaceutical Technology
JUNE 28, 2023
SK bioscience and the Peter Doherty Institute have signed a research collaboration deal for the development of a influenza vaccine.

Drug Discovery World
MAY 1, 2024
The round was heavily oversubscribed, also drawing in funds from Cancer Research Horizons, Cancer Research Institute, CRIS Cancer Foundation, Kindred Capital, Manta Ray, Martlet Capital, Meltwind Advisory, Saras Capital, Wilbe Capital, and expert angel investors. seed financing appeared first on Drug Discovery World (DDW).
Pharmaceutical Technology
NOVEMBER 30, 2022
in a project agreement from the US government for developing self-amplifying RNA (saRNA) vaccine technology against advanced and emergent viral threats. Development of vaccines to Phase I trials under the five-year $59m prototype project comprises additional $28.4m in milestone payments.
Pharmaceutical Technology
DECEMBER 6, 2022
Evaxion Biotech and ExpreS 2 ion Biotechnologies have entered a vaccine discovery collaboration agreement to co-develop a new cytomegalovirus (CMV) vaccine candidate. Additionally, this project will be part of the development pipeline of Evaxion under EVX-V1. A herpesvirus family member, CMV is spread through body fluids.
Pharmaceutical Technology
MAY 11, 2023
Results published in Nature for a personalised pancreatic cancer vaccine that uses neoantigens from patients’ tumours have lent further support to early positive signals. The vaccine, developed by BioNTech, led to half of the patients with pancreatic cancer in the Phase I trial remaining cancer-free 18 months later.

Pharmaceutical Technology
OCTOBER 26, 2022
SK bioscience has signed a new collaboration agreement with the Coalition for Epidemic Preparedness Innovations (CEPI) to develop messenger ribonucleic acid (mRNA) vaccines for infectious diseases. The company has obtained the newest vaccine platform technologies such as bacterial culture, cell culture and genetic recombination.
Pharmaceutical Technology
FEBRUARY 3, 2023
Only a few weeks into the new year, the prospect of getting a successful advanced HIV vaccine shrank after the discontinuation of yet another late-stage trial. On January 18, Janssen, a Johnson & Johnson (J&J) subsidiary, stated that its vaccine was not effective in preventing HIV infections.
Drug Discovery World
MAY 2, 2024
A study led by the University of Oxford has successfully investigated human immunity against Covid-19 in people who already have antibodies, with the aim of advancing future vaccines and treatments. The results suggest that previous infection, together with vaccination, offers strong protection against the original Covid-19 strain.

Pharmaceutical Technology
APRIL 14, 2023
Ghana’s Food and Drug Authority (FDA) has approved R21/Matrix-M malaria vaccine in children aged 5 to 36 months, marking the first regulatory clearance for the University of Oxford-developed vaccine in any country in the world. Children between the ages of five and 36 months are at highest risk of death from malaria.

Medical Xpress
APRIL 7, 2023
After helping to develop and test new mRNA technologies for COVID-19 vaccines, University of Maryland School of Medicine (UMSOM) researchers and scientists are turning their attention to utilizing this innovative technology to ward off other infectious diseases like malaria and influenza.
Drug Discovery World
APRIL 15, 2024
Naobios has signed a partnership agreement with Sumagen, a Korean-Canadian biotechnology company developing an HIV-1 vaccine candidate, to manage the manufacturing process during its Phase II clinical trials. At the end of 2022, the World Health Organization (WHO) reported that approximately 39 million people were living with HIV.

AuroBlog - Aurous Healthcare Clinical Trials blog
SEPTEMBER 22, 2022
The Indian Institute of Science (IISc), Centre for BioSystems Science and Engineering, has designed a new method to deliver a vaccine candidate for tuberculosis (TB). The research involves using spherical vesicles secreted by bacteria coated on gold nanoparticles which can then be delivered to the immune cells.
Drug Discovery World
APRIL 18, 2024
For the first time, a controlled human infection model for tuberculosis (TB) has been developed and could help with the future development of TB vaccines. The team states that it’s the first step towards establishing a challenge model that can be used to test new vaccines for TB.

BioPharma Reporter
SEPTEMBER 1, 2022
from the Biomedical Advanced Research and Development Authority (BARDA): allowing the company to expand its current influenza vaccine program to include development of a pandemic influenza vaccine based on its proprietary self-amplifying mRNA platform. Arcturus Therapeutics has obtained an award for up to $63.2m

Bio Pharma Dive
DECEMBER 18, 2023
The newly established initiative, which consists of a research center and an accelerator, will focus on developing vaccines for tuberculosis, influenza and group A streptococcus.
BioSpace
JULY 7, 2022
Facing the looming threat of AMR, researchers finally have a new clue as to why attempts to develop an effective S. aureus vaccine have failed. BioSpace spoke with the team leaders.

Bio Pharma Dive
FEBRUARY 1, 2021
Gritstone Oncology, known for its cancer research, is drawing attention in infectious disease, signing a deal with Gilead two weeks after announcing plans to develop a coronavirus vaccine.

Scienmag
FEBRUARY 24, 2022
To address climbing economic losses from swine that contract the porcine epidemic diarrhea virus, Virginia Tech researchers in the College of Agriculture and Life Sciences and Virginia-Maryland College of Veterinary Medicine are developing a vaccine to combat the disease that has a near 100 percent mortality rate in newborn piglets.

Pharmaceutical Technology
DECEMBER 5, 2022
Sanofi has signed an agreement with the Queensland Government in Australia to establish a $190m (A$280m) research facility in Brisbane. With the establishment of this Translational Science Hub, Queensland is set to become an international messenger ribonucleic acid (mRNA) vaccine hub.
Pharmaceutical Technology
JULY 1, 2022
Some months after the first Covid-19 vaccines became available, the World Health Organization (WHO) set a target of vaccinating 70% of each country’s population by the end of June 2022. The post Magazine-How delays derailed Covid-19 vaccination in Africa appeared first on Pharmaceutical Technology.
AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 8, 2023
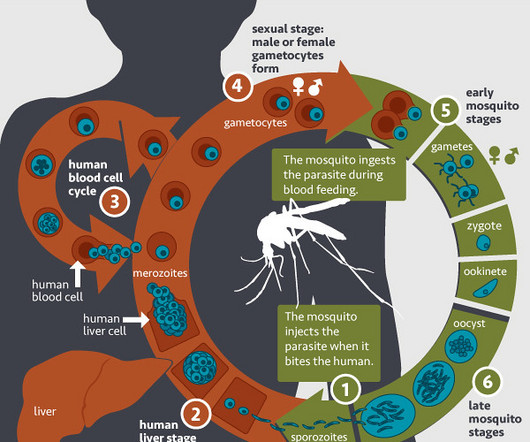
The World Health Organization has approved a new vaccine that scientists argue will be a game-changer in the fight against malaria, which kills half a million people in Africa every year.
pharmaphorum
NOVEMBER 20, 2022
One standout success we have all benefitted from is vaccine development, with notable vaccines from Pfizer/BioNTech, AstraZeneca, and Moderna. As the world stopped, researchers and stakeholders from across the industry turned their attention to tackling the SARS-CoV-2 virus. New hurdles from COVID-19 vaccine development.
Pharmaceutical Technology
MARCH 21, 2023
Pre-clinical-stage biotechnology firm InvisiShield Technologies has partnered with Gladstone Institutes to develop intranasal preventatives against airborne viral infections including influenza, respiratory syncytial virus (RSV) and SARS-CoV-2.

Medical Xpress
MARCH 22, 2023
A UCLA-led team has developed an inexpensive, universal oral COVID-19 vaccine that prevented severe respiratory illness and weight loss when tested in hamsters, which are naturally susceptible to SARS-CoV-2. It proved as effective as vaccines administered by injection or intranasally in the research.

Pharmaceutical Technology
DECEMBER 2, 2022
India’s Central Drugs Standard Control Organisation (CDSCO) has granted approval for Bharat Biotech’s BBV154 vaccine for treating Covid-19. BBV154 is claimed to be the first intra-nasal vaccine for Covid-19 in the world.

Pharma Mirror
AUGUST 28, 2021
ADELAIDE- Agilex Biolabs, the Australian specialist bioanalytical and toxicology laboratory facilities for clinical trials is partnering with Endpoints News to share the latest on “Non-clinical and clinical pathways for rapid vaccine development in Australia”, in a webinar hosted by Endpoints News Editor Arsalan Arif.
Pharmaceutical Technology
JUNE 1, 2023
A new intracellular drug delivery centre will be established in the UK to support potential ribonucleic acid (RNA) vaccines and therapeutics , as well as the development of innovative drug delivery technologies. It will focus on studying and developing new lipid nanoparticle (LNP) formulations for the delivery of RNA medicine.

Drug Discovery World
OCTOBER 14, 2022
Merck (MSD) and Moderna have revealed plans to work together on the development and commercialisation of an investigational personalised cancer vaccine. The post Merck and Moderna to develop a personalised cancer vaccine appeared first on Drug Discovery World (DDW).

Pharma Mirror
APRIL 24, 2022
and NEC Corporation, today announced the execution of a strategic research collaboration agreement for the development of a novel hepatitis B therapeutic vaccine. NEC OncoImmunity, an NEC subsidiary that specializes in artificial intelligence-driven biotechnology, is also participating in this research collaboration.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 13, 2023
COVID sent us into a series of lockdowns in a bid to control the spread of the virus until a vaccine could be developed. Most of us knew that a vaccine would be the key to our post-pandemic […] The world changed for all of us when we were suddenly plunged into a pandemic in 2020.
Pharmaceutical Technology
FEBRUARY 14, 2023
The US Department of Health and Human Services (HHS) and the Department of Defense have modified their existing agreement with Novavax to secure additional doses of the Covid-19 vaccine, Adjuvanted (NVX-CoV2373). million additional doses of its Covid-19 vaccine. It was developed using the recombinant nanoparticle technology of Novavax.
pharmaphorum
NOVEMBER 25, 2022
Scientists in the US say they have developed an mRNA-based vaccine that encompasses all 20 known subtypes of influenza that could form the basis of a future universal jab. The goals of the programme are also similar to those in COVID vaccine development.

VirTrial
OCTOBER 25, 2023
Vaccine studies can be large, multinational endeavors with sensitive timeframes, extended participation follow-up periods, multiphase designs, and critical data management needs. In recent years, Signant has been a part of more than 95 vaccine studies involving 140,000+ patients in more than 30 countries.
Bio Pharma Dive
AUGUST 11, 2020
The shot, developed by the state-backed Gamaleya Research Institute, hasn't yet been tested in the large, placebo-controlled tests scientists emphasize are needed to prove a vaccine is protective.

Drug Discovery World
JUNE 6, 2024
Read more: Immunotherapy prior to surgery a ‘game-changer’ for bowel cancer Long-lasting effect from melanoma vaccine Cancer vaccine mRNA-4157 (V940) in combination with Keytruda reduced the risk of recurrence or death by 49% in patients with resected high-risk melanoma.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content