BMS agrees to acquire Karuna Therapeutics for $14bn
Pharmaceutical Technology
DECEMBER 26, 2023
Bristol Myers Squibb (BMS) has signed a definitive agreement for the acquisition of Karuna Therapeutics for $14bn in cash.

Pharmaceutical Technology
DECEMBER 26, 2023
Bristol Myers Squibb (BMS) has signed a definitive agreement for the acquisition of Karuna Therapeutics for $14bn in cash.

XTalks
DECEMBER 26, 2023
Karuna Therapeutics, a biopharmaceutical company dedicated to uncovering, advancing and providing groundbreaking medicines for individuals facing psychiatric and neurological conditions, has recently disclosed encouraging outcomes from the Phase III EMERGENT-2 trial of KarXT (xanomeline-trospium) in adults diagnosed with schizophrenia. “New treatments and novel mechanisms are urgently needed for people with schizophrenia because many don’t respond to their therapy and others only have a partial
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
DECEMBER 26, 2023
ABBV-400 is under clinical development by AbbVie and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

pharmaphorum
DECEMBER 26, 2023
Exploring the powerful collaborations between scientists, investors, and the medical community in finding breakthrough treatments for major diseases, and the notion of 'healthspan'

Pharmaceutical Technology
DECEMBER 26, 2023
Exarafenib is under clinical development by Kinnate Biopharma and currently in Phase I for Non-Small Cell Lung Cancer.

pharmaphorum
DECEMBER 26, 2023
In the first of a five-part festive instalment of the pharmaphorum podcast, web editor Nicole Raleigh speaks with Advarra’s chief product officer, Elisa Cascade, about the reasons behind increasing burdens on clinical research sites over the last five years.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Drug Patent Watch
DECEMBER 26, 2023
Annual Drug Patent Expirations for HYSINGLA+ER Hysingla Er is a drug marketed by Purdue Pharma Lp and is included in one NDA. It is available from one supplier. There are… The post New patent for Purdue Pharma drug HYSINGLA ER appeared first on DrugPatentWatch - Make Better Decisions.

Pharmaceutical Technology
DECEMBER 26, 2023
In 2023, investors pursued pharmaceutical assets with value amidst an unstable economic environment.
Drug Patent Watch
DECEMBER 26, 2023
Annual Drug Patent Expirations for AYVAKIT Ayvakit is a drug marketed by Blueprint Medicines and is included in one NDA. It is available from one supplier. There are three patents… The post New patent for Blueprint Medicines drug AYVAKIT appeared first on DrugPatentWatch - Make Better Decisions.

Pharmaceutical Technology
DECEMBER 26, 2023
NVG-111 is under clinical development by NovalGen and currently in Phase I for Mantle Cell Lymphoma.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
Drug Patent Watch
DECEMBER 26, 2023
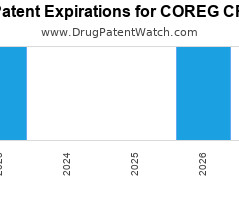
Annual Drug Patent Expirations for COREG+CR Coreg Cr is a drug marketed by Woodward and is included in one NDA. It is available from four suppliers. There are two patents… The post New patent expiration for Woodward drug COREG CR appeared first on DrugPatentWatch - Make Better Decisions.

Pharmaceutical Technology
DECEMBER 26, 2023
AWT-020 is under clinical development by Anwita Biosciences and currently in Phase II for Sarcomas.

Drug Discovery World
DECEMBER 26, 2023
Otsuka Pharmaceutical and Ionis Pharmaceuticals Otsuka Pharmaceutical and RNA therapy company Ionis have entered into a licence agreement for Otsuka to acquire exclusive marketing rights to Ionis’ hereditary angioedema (HAE) drug candidate donidalorsen in Europe. Donidalorsen is an investigational ligand-conjugated antisense (LICA) medicine designed to target the prekallikrein, or PKK, pathway.

Pharmaceutical Technology
DECEMBER 26, 2023
Sacituzumab tirumotecan is under clinical development by Merck and currently in Phase II for Human Epidermal Growth Factor Receptor 2 Negative Breast Cancer (HER2- Breast Cancer).

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

pharmaphorum
DECEMBER 26, 2023
AstraZeneca agrees a $1.

Pharmaceutical Technology
DECEMBER 26, 2023
KSQ-4279 is under clinical development by F. Hoffmann-La Roche and currently in Phase I for Ovarian Cancer.

Pharmaceutical Commerce
DECEMBER 26, 2023
Items in this sector ranged touched on topics such as pharma shipping, container technology, and sustainability.

Pharmaceutical Technology
DECEMBER 26, 2023
BI-765179 is under clinical development by Boehringer Ingelheim International and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Drug Patent Watch
DECEMBER 26, 2023
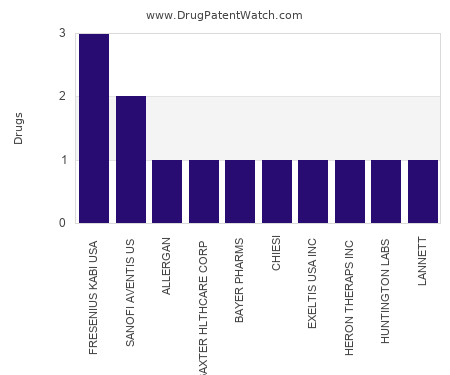
This chart shows the pharmaceutical companies with the most emulsion dosed drugs. For a different perspective, see the most popular dosage types. The companies with the most emulsion dosed drugs… The post Which pharmaceutical companies have the most emulsion dosed drugs? appeared first on DrugPatentWatch - Make Better Decisions.

Pharmaceutical Technology
DECEMBER 26, 2023
BI-765179 is under clinical development by Boehringer Ingelheim International and currently in Phase I for Cutaneous Squamous Cell Carcinoma (cSCC).

Drug Discovery World
DECEMBER 26, 2023
DDW’s Diana Spencer takes a look at the breaking news stories that were most popular with DDW readers over the last year. Representing a dominant field of drug discovery, it isn’t surprising that breakthroughs in cancer research were popular with DDW readers in 2023, particularly strategies to overcome treatment resistance and prevent the spread of disease.

Pharmaceutical Technology
DECEMBER 26, 2023
BR-790 is under clinical development by Jiangxi Qingfeng Pharmaceutical and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Pharmaceutical Technology
DECEMBER 26, 2023
Sacituzumab tirumotecan is under clinical development by Merck and currently in Phase II for Metastatic Transitional (Urothelial) Tract Cancer.

Pharmaceutical Technology
DECEMBER 26, 2023
Sacituzumab tirumotecan is under clinical development by Merck and currently in Phase II for Adenocarcinoma Of The Gastroesophageal Junction.

Pharmaceutical Technology
DECEMBER 26, 2023
AZD-9550 is under clinical development by AstraZeneca and currently in Phase I for Metabolic Dysfunction-Associated Steatohepatitis (MASH).

Pharmaceutical Technology
DECEMBER 26, 2023
ADRX-0706 is under clinical development by Adcentrx Therapeutics and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Pharmaceutical Technology
DECEMBER 26, 2023
HEC-88473 is under clinical development by HEC Pharma and currently in Phase I for Metabolic Dysfunction-Associated Steatohepatitis (MASH).

Pharmaceutical Technology
DECEMBER 26, 2023
TAK-500 is under clinical development by Takeda Pharmaceutical and currently in Phase II for Head And Neck Squamous Cell Carcinoma (HNSC).

Pharmaceutical Technology
DECEMBER 26, 2023
NT-175 is under clinical development by Neogene Therapeutics and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

Pharmaceutical Technology
DECEMBER 26, 2023
ORB-011 is under clinical development by Orionis Biosciences and currently in Phase I for Head And Neck Squamous Cell Carcinoma (HNSC).

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content